Therapeutic Potential Of Phenylethanoid Glycosides: A Systematic Review
Sep 29, 2022
Abstract:
Phenylethanoid glycosides (PhGs) are generally water-soluble phenolic compounds that occur in many medicinal plants. Until June 2020, more than 572 PhGs have been isolated and identified. PhGs possessantioxidant, neuroprotective, anti-inflammatory, antibacterial, antiviral, antidiabetic, anticancer, and antiobesity properties. Despite of these promising benefits, PhGs have failed to fulfill its their therapeutic applications due to their poor bioavailability. The attempts to understand their metabolic pathways to improve their bioavailability are investigated. In this review article, we will first summarize the number of PhGs compounds which is not accurate in the literature. The latest information on the biological activities, structure-activity relationships, mechanisms and especially the clinical applications of PhGs will be reviewed. The bioavailability of PhGs will be summarized and factors leading to the low bioavailability will be analyzed. Recent advances in methods such as bioenhancers and nanotechnology to improve the bioavailability of PhGs are also summarized. The existing scientific gaps of PhGs in knowledge are also discussed, highlighting research directions in the future. Keywords: Phenylethanoid glycosides; Bioavailability; Acteoside; Salidroside; Echinacoside
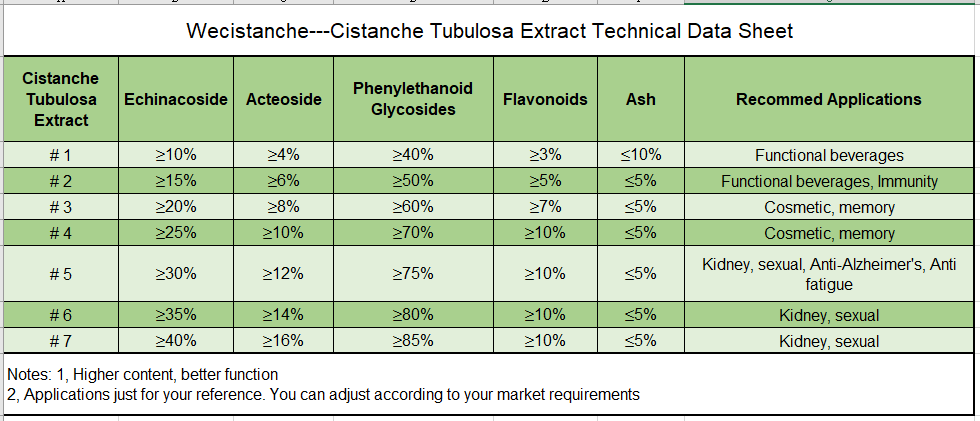
Phenylethanoid glycosides Specification in Cistanche Herba
Introduction
Phenylethanoid glycosides (PhGs) are generally water-soluble phenolic compounds that occur in many medicinal plants. PhGs have been isolated from the roots, stems, bark, leaves, flowers, fruits and seeds of medical plants, as well as from suspension cell cultures, callus tissues and hairy roots cultures. They are also found in various plant-based foods such as edible flowers and tea. However, their accumulations in each plant organ may vary considerably1-3 . The main PhGs are reported from the families Acanthaceae, Berberidaceae, Asteraceae, Gesneriaceae, Lamiaceae, Loganiaceae, Magnoliaceae, Oleaceae, Orobanchaceae, Plantaginaceae, Portulacaceae, Rosaceae, Scrophulariaceae, and Verbenaceae4 . For example, a total of 69, 51, 21 and 16 PhGs have been isolated from Cistanche herba5 , Forsythiae fructus6 , Magnoliae officinalis7 and Houttuynia cordata8 , respectively. Acteoside (also known as acteoside), one of the representative PhGs, is widely distributed in the family Lamiaceae,Plantaginaceae, Scrophulariaceae, and Orobanchaceae9 . In1994, Jiménez and Riguera summarized the structures and biological activities of 155 PhGs reported before 19922 . In 2008, Fu et al. provided an overview in the advances on 190 new PhGs isolated from 1997 to 200710 . The detailed information of 116 new PhGs identified during 2009-2016 was given in 20163 . In the present review, we summarized the 111 PhGs11-56 that have not been reviewed previously (1993-1997, 2007-2009 and 2016-present) in Table 1 and Table 2. The latest new PhGs (Ginkgoside C and D) was published on 16 June 2020. Up to 572 PhGs were identified from nature until June 2020. These 572 PhGs are distributed in 21 orders and 35 families of the plant kingdom (Figure 1). It should be noted that some PhGs identified were not published in English. Thus, the actual number identified must be over 572. In general, the basic structure of PhGs consists of a hydroxyphenylethyl unit as an aglycone which is attached to a sugar moiety mostly a β-D-glucopyranose through a glycosidic bond at the C-1 site. In most cases, the glucose moiety is esterified with a hydroxycinnamic acid derivative such as caffeic acid, coumaric acid, cinnamic acid, and ferulic acid. Rhamnose, xylose, arabinose, allose, galactose, and apiose, among others, may also be attached to the glucose residue (Figure 2, Table 1 and 2). The diversity of sugar and hydroxyphenylethyl moieties make the plentiful variation of PhGs. Generally, the number of sugars ranges from one to three. However, four-sugar and five-sugar residues are also found occasionally. According to the number of the sugars bonded to hydroxyphenylethyl moieties, PhGs can be classified into monosaccharidic PhGs, dissaccharidic PhGs, trisaccharidic PhGs, tetrasaccharidic PhGs, and pentasaccharidic PhGs4 . To date, there are 10 tetrasaccharidic PhGs reported, namely, magnolosides C57 , ballotetroside58, trichosanthoside B59, marruboside60, velutinosides I61, velutinosides II61 , lunariifolioside62 ,raduloside63, barlerinoside64, and poliumoside B65. Only one pentasaccharidic PhGs named yulanoside A from M.salicifolia was reported in 201566
Most purified PhGs are white, buff or yellow amorphous powders with high polarity. They are soluble in polar solvents but insoluble in non-polar organic solvents67 . As the characteristics of the strong ultraviolet (UV) absorption in PhGs, it is easy to monitor these compounds by UV spectrophotometer. The specific UV spectra of each PhGs can also serve as an index to deduce the structure. For example, the UV absorption peaks of Acteoside and isoActeoside are 232, 246, 289, 332 nm, and 232, 246, 286, 328 nm, respectively68 . And that of echinacoside are 236, 288, 330 nm69 . PhGs and the extracts rich in PhGs exhibited various benefits, such as antioxidant activity, neuroprotective effect, anti-inflammatory activity, antibacterial activity, antivirus activity, anti-diabetic activity, anti-cancer activity, and anti-obesity activity3, 9. Figure 3 shows the number of papers and times cited of papers indexed in the Web of Science related to “PhGs”, illustrating a significant increase in publication in this area. Although over 572 PhGs have been isolated and identified, only a few of them are extensively studied. For example, the number of papers on salidroside, Acteoside, echinacoside, forsythoside and isoActeoside are 1746, 1258, 538, 370 and 230, respectively. And the numbers of citations of the papers about Acteoside, salidroside, echinacoside, forsythoside and isoActeoside are 19356, 14352, 6468, 3234 and 4098, respectively. Other PhGs have fewer than 100 papers published. The number of papers published and number of citation of the papers of specific PhGs are also shown in Figure 3. Despite of the many promising biological activities, PhGs have failed to fulfill the therapeutic applications due to poor oral bioavailability3 . The bioavailability of Acteoside was found to be 0.12% in rats after Acteoside was given at the dosages of 100 mg/kg oral administration (p.o.) and 3 mg/kg intravenous injection (i.v.) 70 , but the bioavailability of vActeosidein dogs was around 4% after vActeosidewas given at 40 mg/kg intragastric administration (i.g.) and 5 mg/kg i.v.71 . The bioavailability of echinacoside, and angoroside C in rats at the dose of 100 mg/kg i.g. and 5 mg/kg i.v., was reported to be 0.83%72 and 2.1%73 , respectively. The bioavailability of forsythiaside (100 mg/kg p.o. and 5 mg/kg i.v.) and poliumoside (200 mg/kg p.o. and 10 mg/kg i.v.) in rats was 0.5%74 and 0.69%75 , respectively. Feng et al. compared the pharmacokinetic and bioavailability characteristics of savaside A, vActeoside and isovActeosidein rats after the compounds were given at the dosages of 1000 mg/kg p.o. and 5 mg/kg i.v.. The bioavailability order of the three PhGs appears to be vActeoside> isovActeoside> savaside A76 . Zhang et al investigated the pharmacokinetic of four PhGs (vActeoside isovActeoside martynoside, and crenatoside) after orally administrated 10.0 g crude Acanthus ilicifolius herb /kg to rats. Although the four PhGs share similar molecular structures, they displayed different elimination half-lives (T1/2), and different areas under the curves (AUC0–t), ranging from 3.4 to 9.0 h, and 1826.3 to 23.6 μg/L×h, respectively77 . Different dosages and administrative patterns might affect the bioavailability of PhGs. However, there is one exception. The bioavailability of salidroside was reported to be 51.97%78 . As for the reasons why the bioavailability of salidroside was significantly higher than other PhGs, this may be ascribed to its relatively simple structure (Figure. 2). Salidroside belongs to monosaccharidic PhGs consisting of phenylethanol and sucrose, and the relatively large polarity allowed it to be easily excreted from the urine without complicated metabolic processes. The higher absorption of salidroside may also lead to its obviously higher bioavailability than other PhGs (section 5.1). Numerous approaches such as bioenhancers, β-cyclodextrin encapsulation, liposomal PhGs, nanoparticles and phospholipid complex have been applied to improve the bioavailability of PhGs. There have been a number of reviews on PhGs since the 90’s. As early as 1994, Jiménez and Riguera reviewed the isolation, purification, as well as structure and biological activity of PhGs2 . Pan et al. highlighted the pharmacological activities of natural PhGs in 200379 . Fu et al. summarized the phytochemistry and bioactivity of PhGs in 200810. Radev et al. published a mini review on pharmacological effects of PhGs in 201080 . Xue and Yang summarized advances in the phytochemistry, pharmacology and pharmacokinetics of PhGs in 20163 . Alipieva et al. reviewed the biosynthesis and pharmacological significance of vActeoside the most popular phenylethanoid glycoside in 20149 . Liu et al. generalized the distribution, extraction methods, poor pharmacokinetics and therapeutic uses of echinacoside in 201881 . Tao et al. gave a detailed summary of chemical, pharmacological, toxicological, and clinical studies of various Rhodiola species with salidroside as the characteristic chemical constituents in 201982 . However, there are no comprehensive reviews concerning the stability, biotransformation, clinical application and bioavailability of PhGs. This review will summarize the latest information on the chemistry, pharmacology, stability, clinical application, pharmacokinetics, metabolites and biotransformation of PhGs. Recent advances in methods such as bioenhancers and nanotechnology to improve the bioavailability of PhGs will also be summarized. The existing scientific gaps of PhGs in knowledge are also discussed, highlighting research directions in the future
2. Pharmacology of PhGs
PhGs have been reported to have various bioactivities in cell and animal models. Herein, the potential health benefits of PhGs are summarized, and the structure-activity relationship and mechanisms of PhGs’ pharmacology are highlighted.
2.1 Antioxidant and free radical scavenging activity of Phenylethanoid glycosides
Many PhGs and extracts rich in PhGs have shown powerful antioxidant activity. Two new PhGs named macrophylloside E and macrophylloside F, together with eight known PhGs (jionoside C, forsythoside B, alyssonoside, Acteoside, isoActeoside, martinoside, isomartinoside and leucosceptoside) were isolated from Callicarpa macrophylla. All the ten PhGs showed high to moderate antioxidant effect with the IC50 from 2.72 to 38.65 μM in the DPPH assay43 . Acteoside isolated from Plantago major can significantly scavenge both DPPH radical (IC50, 11.27 μM) and superoxide radicals (IC50, 1.51 μM). Acteoside can also inhibit lipopolysaccharide induced production of nitric oxide in RAW264.7 macrophages (IC50, 75.0 μM) 83. Seven PhGs (plantalide A, vActeoside plantamajoside, martynoside, himaloside B, desrhamnosyl isovActeosideand plantainoside D) discovered from P. asiatica showed DPPH radical scavenging activity with the IC50 values ranging from 22.9–88.5 μM. While other 22 compounds from P. asiatica showed weak antioxidant activity85. In addition, VActeosidevActeosideand salidroside were demonstrated to be two major PhGs contributing to the great antioxidant capacities of Osmanthus fragrans flowers85 . All nine PhGs (magnolosides Ia, Ib, Ic, IIa, IIb, IIIa, Iva, and Va and crassifolioside) from M. officinalis were found to possess strong free radical scavenging potential with the IC50 ranging from 11.79 to 20.99 μM, and magnoloside Ia (IC50, 11.79 μM) was the strongest one86 . The DPPH radical scavenging capacity of crassifolioside (IC50, 21.38 μM), magnoloside IIa (22.94 μM), and magnoloside IIb (24.62 μM) was weaker than that of magnoloside Ia (11.79 μM), magnoloside Ic (12.99 μM), magnoloside Ib (16.23 μM), and magnoloside Va (20.99 μM). As we can see from the structures of these compounds, crassifolioside, magnoloside IIa and magnoloside IIb contained three sugars while magnoloside Ia, magnoloside Ic, magnoloside Ib, and magnoloside Va contained two sugars. More sugars mean larger steric hindrance in compounds and prevent them from easily approaching the free radicals, finally causing the weaker DPPH radical scavenging capacity. In addition, compared with the other seven PhGs, magnoloside IIIa (32.18 μM) and magnoloside IV (35.17 μM) with two adjacent phenolic groups only in one side exhibited poor activity86 . Furthermore, benzene ring plane conjugation in PhGs can be increased by the α, β-conjugated unsaturated ester structures and allow electron delocalization to inhibit free radicals86.
2.2 Neuroprotective effect of Phenylethanoid glycosides
Acteoside, salidroside, and echinacoside exhibited antioxidant and neuroprotective activities in hydrogen peroxide induced apoptosis in PC12 cells via the nuclear factor erythroid 2-related pathway87 . CaleolariosideB, paraboside B, and paraboside II isolated from Paraboea martinii effectively protected PC12 cells from H2O2-induced damage by upregulating HO-1 88 . It is believed that β amyloid peptide (Aβ) is a major cause of Alzheimer’s disease89 . Total PhGs extracted from C. Herba at concentrations of 5, 25 and 50 μg/mLincreased the viability and decreased LDH and MDA release by PC12 cells injured with Aβ1‑4290. Torenoside B and Savatiside savatiside A were demonstrated to improve the enzyme activity of GSH‑Px and SOD, decrease the content of MDA and ROS, and downregulate intracellular Ca2+ concentrations and Calnexin expression in Aβ25–35 induced SH‑SY5Y cells91 . VActeoside salidroside, and PhGs from C. Herba have significant protective potential against oxidative stress induced by Aβ 92, 93. The characteristic pathology in Parkinson’s disease is the degeneration of dopamine neurons in the substantia nigra pars compacta94 . Campneoide and tubuloside B can protect neurons from 1-methyl-4-phenylpyridinium induced apoptosis in vivo 95, 96 . VActeosidehas potential therapeutic value against PD through attenuating the oxidative stress and activating the Nrf2/ARE signaling pathway97 . SAMP8 mice, a model for AD, were administered by PhGs extracted from C. Herba daily intraperitoneally at 25, 50, or 100 mg/kg/day for 30 days. PhGs were found to improve cognitive deficits in SAMP8 mice by improving synaptogenesis and synaptic plasticity98. It has been reported that the mean lifespan of caenorhabditis elegans was extended by 13.64% and 15.82% after treated with 200 μM and 300 μM ECH, respectively. The protective effect of ECH on Aβ-induced toxicity in C. elegans was almost equal to that of ginkgolide A, a well-known agent with positive effects for AD99 . Liu et al. synthesized eight PhGs derivatives based on calceolarioside A, and studied their neuroprotective effects in PC12 cells. The results showed that seven compounds could protect the cell damage or death from the free radical damage except the chloro-substituted analog. The structure-activity relationship indicated that the catechol moiety might not monopolize the bioactivity but probably could play an important role in neuroprotection and the glucose moiety seemed not important for the neuroprotection100 . The findings were consistent with the recent structure-activity of caffeic acid phenethyl ester analogs101, 102

Click here to know more info about
How Cistanche Take An Effect on Human Health
2.3 Hepatoprotective effect of Phenylethanoid glycosides
Acteoside, echinacoside, tubuloside B, cistanoside A and 2-acetylacteosid possess offer hepatoprotective effects via multiple mechanisms including strengthening antioxidant defense system, free radicals scavenging, and blocking cytochrome P450 biotransformation103 . Leucoseceptoside A, crenatoside, martynoside, and 3-O-methylcrenatoside extracted from Incarvillea compacta alleviated CCl4-induced hepatotoxicity by enhancing the activity of superoxide dismutase, decreasing the intracellular ROS and malondialdehyde content as well as activating NF-κB pathway104 . 14 Fourteen PhGs isolated from Forsythia suspensa were evaluated for their hepatoprotective effects on HepG2 cells damage induced by APAP. It was found that forsythoside N, forsythoside O, forsythenside A and forsythenside B exerted significant hepatoprotective activities28 with the cell survival rates from 52.48% to 67.15%, 67.61%, and 64.88% at the concentration of 10 μM, respectively. Cistanoside A (125, 250, and 500 mg/kg/day) could alleviate ethanol-induced hepatotoxicity in mice by improving the activities of the activities of energy metabolism enzymes (Ca2+ -Mg2+ -ATPase, ATPase, and Na+ -K+ -ATPase), mitochondrial antioxidant enzymes (SOD, GST and CAT), and antioxidant defense system105 . Besides, cistanoside A (100, 75, 50, and 25 μg/mL) suppressed the apoptosis of hepatocytes by increasing the expression of Bcl-2 and supressing cfos105 . Echinacoside (60 mg/kg, i.p.) could significantly protect LPS and D-galactosamine induced acute liver injury in mice due to its anti-apoptotic and anti-inflammatory activities106 . PhGs from C. deserticola was assessed for their hepatoprotective activity in vitro and in vivo. Concentrations of 0.33, 1.00, 3.00 mg/mL PhGs can could improve the HepG2 cells viability to almost 10%, 22% and 35%, respectively. After orally administered with PhGs at 200, 600 or 1800 mg/kg for 31 consecutive days, ICR mice with liver injury induced by alcohol showed improved hepatic indicators (superoxide dismutase, glutathione Stransferase, glutathione, glutathione peroxidase, malondialdehyde and triglyceride) levels107 . Structure–activity relationship indicated that the catechol moiety on PhGs was important for the hepatoprotective activity108 . Acteoside (IC50, 4.6 μM), 2ʹ-acetylActeoside (4.8 μM), isoActeoside (5.3 μM), tubuloside A (8.6 μM) and echinacoside (10.2 μM) inhibited D-GalN-induced death of hepatocytes109 . Acteoside (IC50, 4.6 μM) showed significantly stronger activity than kankanose (>100 μM), and echinacoside (10.2 μM) showed significantly stronger activity than cistanoside F (>100 μM), which indicated that aglycone was an important group for the activity109. As the activity of isovActeoside(5.3 μM) was higher than kankanoside G (14.8 μM), it can be concluded that aglycone with the 4-hydroxy group showed weaker activity than that having 3,4-dihydroxy group109. The 8-O-β-D-glucopyranosyl part with 6ʹ-O-caffeol group (Tubuloside B, 14.6 μM) showed weaker activity than that with 4ʹ-O-caffeoyl group (2ʹ-acetylvActeoside 4.8 μM)109. The introduction of 6-O-β-D-glucopyranosyl (echinacoside < vActeoside and 2ʹ-O-acetyl moiety (2ʹ-acetylvActeosidevActeoside could reduce the protective

2.4 Anti-cancer activity of Phenylethanoid glycosides
In a recent study, echinacoside was reported to possess antiproliferative activities (20 μg/mL, 9.57 %; 50 μg/mL, 26.67%; 100 μg/mL, 37.20%) on HepG2 cells by inactivating AKT pathway and decreasing TREM2 expression110 . Acteoside, echinacoside, cistantubuloside A, cistanoside A, and 2´- acetylActeoside inhibited the proliferation of mouse skin melanoma cancer cell line KML with the inhibition rate ranging from 33% to 93%111 . Pretreatment with 5, 10, 20, 40 and 50 μM salidroside for 48 h can inhibit the proliferation of human breast cancer MCF-7 cells to almost 70%, 60%, 55%, 45% and 30%, respectively. The mechanism maybe related with increasing caspase activity, down-regulating the Bcl-2 expression, and up-regulating the Bax expression. Moreover, salidroside treatment inhibited tumor growth in a xenograft tumor model. Compared with control group, after treated with salidroside (50 mg/kg body weight) on alternate days for 3 weeks, the weight and volume of tumor was decreased by 0.7 g and 300 mm3 , respectively112 . Salidroside was reported to possess anti-tumor activity against Wilms' tumor113 , breast cancer114, ovarian cancer115, gastric cancer116, skin cancer117, renal cell carcinoma118 and colorectal cancer119 . Li et al. investigated the effects of PhGs extract from C. tubulosa (CTPG) on the inhibition of melanoma cell (B16-F10) growth. In vitro, 100 μg/mL of CTPG for 48 h or 200 μg/mL of CTPG for 72 h treatment inhibited the growth rates of B16-F10 cell to higher than 60% and 90%, respectively. CTPG can up-regulate the expressions of BAX, down-regulate the expressions of BCL-2, increase the generation of ROS, and reduce the mitochondrial membrane potential in vitro. Furthermore, subcutaneously administering 400 mg/kg CTPG in mice every 2 days for up to 15 days lasted the survival of mice from 8.3% to 41.7% 120 . Acteoside from Pedicularis striata can could inhibit cancer cell growth and cell cycle in G2/M phase, induce apoptosis and inhibition of telomerase activity and reduced telomere length121 . It should be noted that not all PhGs exhibit anticancer properties. For example, Kirmizibekmez et al. tested the cytotoxic activity of four PhGs (plantainoside D, calceolarioside D, neocalceolarioside D and lugrandoside) against a series cancer cell lines, namely SH-SY5Y, T98G, A375, HT29, MCF-7, PC3. All the four compounds showed no toxicity against the six cancer cell lines at the concentration of 1–50 μM122 . A number of structure-activity relationships proved that the caffeic acid moiety and catechol group are essential for the cytotoxicity of PhGs. The number of acetyl moieties and their position in the aliphatic rings also play an important role in the anti-proliferative activities of PhGs123-125 . The antiproliferative activity of Acteoside was almost twice as that of echinacoside and calceolarioside. The similar cytotoxic activity of calceorioside A and vActeosidesuggest that rhamnose substitution does not influence the cytotoxic activity of PhGs126 . VActeosideinhibited about 23%–30% of the proliferation activity of the cancer cells, which is almost twice as many as echinacoside (10%–18%), calceolarioside A (13%–18%), and calceolarioside B (5%–15%). The higher antiproliferative activity may be related to the α-Rha-(1→3)- Glc disaccharide unit and the 4-caffeoyl function in vActeoside27 . The structure-cytotoxicity relationships among 14 PhGs compounds indicated that the fewer sugar units they have, the stronger activities they may have. Furthermore, the position of phenolic acid does not affect the activity. Besides, methylation of the phenolic hydroxyl groups has an adverse impact on the activity128 .
2.5 Anti-inflammatory activity of Phenylethanoid glycosides
The anti-inflammatory activity of PhGs is often connected to suppression of MAPK, NF-κB, and JAKSTATs pathways and activation of Nrf2 pathway129 . Wu et al. confirmed that PhGs (Acteoside, parvifloroside A, syringalide A, 3′-α-L-rhanmnopyranoside, forsythoside B, poliumoside and alyssonoside) from C. kwangtungensis provided protection against LPS-induced inflammatory response in RAW 264.7 macrophages by activating Keap1/Nrf2/HO-1 signaling pathway130 . Echinacoside attenuated LPS-induced inflammation in rat intestine epithelial cells by suppressing the mTOR/STAT3 pathway131 . Acteoside can inhibit the release of β-hexosaminidase, arachidonic acid and histamine in RBL-2H3 cells through inhibiting MAPK and JNK pathways and Ca2+ independent phospholipase132-134 . Acteoside (30, or 60 mg/kg) was shown to decrease inflammatory response against LPS-induced acute lung injury in mice by inhibiting NF-κB signaling pathway135. Gao et al. investigated the anti-inflammatory effects of vActeoside isovActeoside torenoside B and savaside A and found that isovActeoside(80 μM), possessed the strongest activity on inhibiting the expression of iNOS and COX-2 136. IsovActeosideexerts anti-inflammatory via modifying NF-κB and MAPK pathways136 . Forsythiaside A was reported to have protective potential on LPS-induced inflammation in BV2 microglia cells and primary microglia cells via increasing Nrf2 and HO-1 levels and suppressing NF-κB pathway137 . Forsythiaside A could attenuate inflammation in acute liver injury animals by activating Nrf2 and inhibiting NF-κB pathway138 . PhGs from Phlomis younghusbandii exerted anti-inflammatory properties on acute hypobaric hypoxia-stimulated HACE in rats by rehabilitating the oxidative stress levels and inhibiting the expression of pro-inflammatory cytokines regulated by the NF-κB signaling pathways139 . The anti-inflammatory activity of seven PhGs on inhibiting NO production showed that leucosceptoside A (IC50, 9.0 μM ), lipedoside A-I (11.6 μM ), vActeoside(12.8 μM ), isovActeoside(13.7 μM ), and campneoside II (22.1 μM ) possessed stronger activity than martynoside (>100.0 μM) and angoroside C (>100.0 μM). This indicated that the two adjacent hydroxide groups in PhGs may be related to their antiinflammatory activity140. Yang et al. demonstrated that PhGs with two sugar groups possessed weaker activities than others141 .

2.6 Antiviral, the antibacterial and antiprotozoal activity of PhGs
Two new PhGs, Llippiarubelloside A and lippiarubelloside B, together with four known PhGs, Acteoside forsythoside A, and podium side, isolated from Lippia rubella could strongly inhibit the growth of Cryptococcus neoformans at the concentrations of 15-125 μg/mL32 . Total PhGs extract from Monochasma savatieri showed significant anti-bacterial effects at a concentration from 0.0625 to 16 mg/mL142. VeActeosidend forsythoside B showed high antibacterial activities against five strains of Staphylococcus aureus from 64 g/L to 256 g/L, which were comparable to that of norfloxacin143 . When used alone at the dose of 200 μg/mL, veActeosidead inhibitory activity against clinical isolate of Escherichia. coli and Staphylococcus. aureus. However, co-administration of veActeosidend gentamicin showed a synergistic effect against E. coli and S. aureus.. This indicated that veActeosideould be applied to overcome bacterial resistance caused by traditional medicines144 . Isoforsythiaside and forsythiaside are the main antibacterial constituents in Forsythia suspense, which is often applied to treat the infection in upper respiratory tract. Isoforsythiaside and forsythiaside well inhibited the growth of E. coli, P. aeruginosa and S. aureus145, 146 . In addition, forsythoside H exhibited strong inhibitory effects against B. vulgare, B. dysenteriae, M. pneumonia, and A. bacillus147 . VeActeosideas anti-viral activity in vitro and antiinfluenza activity in vivo. And the anti-viral mechanism of veActeosideas related to the activation of ERK and enhancement of IFN-γ production148 . Forsythiaside and calceolarioside B showed significant antiviral potential on respiratory syncytial virus in vitro149 . Forsythiaside inhibited the infectivity of avian infectious bronchitis virus 150 . Taraffinisoside A, a new PhGs isolated from Tarphochlamys affinis, showed antihepatitis B activity with IC50 values of 0.50 and 0.93 mM against hepatitis B surface antigen and hepatitis B eantigen, respectively67 . Forsythoside A from F. suspensa decreased the viral titers of different influenza virus subtypes in cell cultures at the dose of 160 μM. Forsythoside A also increased the survival rate of the mice in an influenza virus infection model at 5 or 10 μg/g body weight 151. Hu et al. evaluated the anti-influenza virus effects of PhGs in vitro and in vivo. PhGs at 0.5 mg/mL could inhibit the influenza A virus H1N1 type infection of Madin Darby canine kidney cell in vitro. PhGs at 300 and 900 mg/kg significantly reduced the mouse lung index (p<0.05), alleviated influenza-induced lethality and clinical symptoms, and prolonged mouse survival time (p<0.05). The mechanism maybe related to up-regulating IFN-γ 152 . It has been reported that veActeosideossessed antiprotozoal activity against Trypanosoma brucei rhodesiense, Leishmania infantum, L. donovani, and L. amazonensis153, 154 . VeActeosidehowed an EC50 of 19 μM against L. promastigotes and is a competitive arginase inhibitor with Ki of 0.7 μM155 . Among seven PhGs extracted from Tecoma mollis, luteoside B and luteoside A showed the strongest antileishmanial activity with the IC50 values of 6.7 and 15.1 μg/mL, respectively156 . Little information is available about the structure-activity relationship of PhGs in its antiviral and antibacterial activities. Kyriakpoulou et al. discovered that samioside is more active than veActeosidegainst four strains of bacteria, indicating that an additional sugar moiety (apiose) at C-4 of rhamnose could contribute to the antibacterial activity157. Although phlinoside C and forsythoside B have a similar structure, phlinoside C hardly inhibit multi-drug-resistant strains of S. aureus. This indicated that introducing the third glycoside (rhamnose) to forsythoside B might cause its inactivity143 .
2.7 Antidiabetic activity of PhGs
A new PhGs named flavaioside from Scrophularia flava showed α-glucosidase inhibitory activities with IC50 value of 6.50 μg/mL. In addition, flavaioside possessed a significant inhibitory activity on the αglucosidase enzyme, and the inhibitory activity (91.85%) was comparable with the known anti-type 2 diabetic drug, acarbose (92.87%)158 . The in vitro experiments showed that veActeoside echinacoside, isoveActeoside2'-acetylveActeosidetubulosides A, tubulosides B, syringalide A' 3-O- rhamnose, campneoside I, and kankanoside J1 from C. tubulosa could offer strong inhibition against lens aldose reductase with their IC50 of 3.1, 1.2, 4.6, 0.071, 8.8, 4.0, 11.1, 0.53, and 9.3 μM, respectively. Especially, 2'-acetylveActeosidehowed the similar activity with epalrestat, a clinical aldose reductase inhibitor159 . VeActeosidend echinacoside were demonstrated to improve glucose tolerance and decrease glucose level in mice at doses of 250-500 mg/kg159 . VeActeosidend echinacoside could suppress the increased postprandial blood glucose level by inhibiting glucose transporter 1-mediated glucose uptake160 . Isocampneoside II isolated from P. coreana could significantly inhibit recombinant human aldose reductase with the IC50 of 9.72 μM. Furthermore, veActeosideisoveActeosideisocampneoside II and cistanoside F effectively inhibited sorbitol accumulation in a rat lens incubated with a high concentration of glucose by almost 70.6, 47.9, 71.3, and 31.7% at 50 μM, respectively161 . Compared with control group, three weeks oral administration of veActeoside10, 20, and 40 mg/kg) caused a significant reduction of blood glucose to 111.30, 74.88, and 75.15 mg/dL, respectively, in diabetic rats. Regarding serum insulin levels, oral treatment with veActeoside10, 20, and 40 mg/kg) elevated the serum insulin level to be 3.23, 5.38, and 6.80 μIU/mL, respectively, in diabetic rats162 .

2.8 Other activities of PhGs
Wu et al. investigated the anti-obesity properties of PhGs form Ligustrum purpurascens. The results showed that PhGs inhibited α-chymotrypsin, trypsin and pepsin with the IC50 values of 0.42, 0.38, and 0.68 mg/mL, respectively. VeActeosidexerted the anti-obesity effects by inhibiting pancreatic lipase. VeActeosideounded to lipase at Ka = 1.88×104 /l mol163. The anti-obesity effect of PhGs from L. purpurascens against fatty diet-fed mice was associated with the up-regulating of mRNA and protein levels of adipose leptin164 .Echinacoside (0.01-10 nmol/L) was reported to boost bone regeneration in MC3T3-E1 cells by enhancing receptor activator of NF-κB ligand (RANKL)165 . Similarly, 12 weeks’ daily i.g. administration ofechinacoside (30, 90, and 270 mg/kg/day) to ovariectomized (OVX) rats significantly increased osteoprotegerin (OPG) level and decreased RANKL level166 . Compared to OVX group, 270 mg/kg/dayechinacoside treatment caused the highest levels of OPG and OPG/RANKL ratios (150.14% and 197.64%)166 . After 12 weeks’ daily orally administration of echinacoside (30, 90, 270 mg/kg/day) in OVX rats, the urine concentration of calcium, inorganic phosphorus, and hydroxyproline was increased by 92.23%, 66.67% and 36.41%, respectively, in 270 mg/kg/day group167 . Cistanoside A (p.o., 20, 40 and 80 mg/kg/day for 12 weeks) was found to promote bone formation and prevent bone resorption in OVX rats by downregulating TRAF6, coordinating the inhibition of NF-kB pathway and stimulating PI3K/Akt pathway168 .

If you have any question, please send us Email: wallencesuen@wecistanche.com






