A Circuit Mechanism Linking Past And Future Learning Through Shifts in Perception Part 2
Sep 28, 2023
Elucidating the control circuit which drives the shift in perception
Next, to characterize the circuit mechanisms that mediate this learning-induced shift in perception, we measured activity in two higher-order interneurons, cerebral ventral 1a (CV1a) and pattern-reversing neuron (PRN). These cells are command-like neurons in the ingestion and egestion circuits, respectively, and, when active, are sufficient to drive their respective motor programs (16, 31). Intracellular recordings showed that CV1a activity was significantly up-regulated at 4 hours after strong training versus naïve animals, while PRN activity was significantly down-regulated (Fig. 3, A to E).
Perceptual change is a change in the way of thinking and an adjustment in mentality. It includes awareness of the surrounding environment, awareness of self-identity, understanding of personal experiences, and the pursuit of goals and aspirations. Perceptual changes can encourage people to take a more positive approach to life and better cope with challenges. And this positive attitude and mentality can improve people's memory.
First, a shift in perception allows people to focus more on the positives in their lives. As "Looking for Optimistic Faces" says, there are often many positive factors in life, but we often fail to discover them. And through changes in perception, we can be more keenly aware of these positive factors and give them enough attention and importance. These optimistic factors can not only accumulate our happiness but also promote us to better grasp the information in life, thereby improving memory.
Secondly, perception change can also help people better understand themselves, including their strengths, weaknesses, values, etc. This deepening of self-awareness allows people to better manage their emotions and stress, and become more motivated to pursue their goals and desires. This kind of ambition and motivation can stimulate people's brains to be more creative and thinking, and further improve their memory.
Finally, perceptual shifts can also lead people to pay more attention to their own experiences and growth. Whether it is success or setbacks, every experience is a valuable asset. Through changes in perception, we can better reflect and summarize, not only better discover our growth points and shortcomings, but also better absorb and remember experiences and lessons.
In short, a shift in perception, while simply a mental adjustment, can have a profoundly positive impact on our lives and learning. With a positive and sunny attitude, we can better cope with challenges, pursue growth, and better master information and memorize content. Let us actively invest in the transformation of perception and continuously improve our growth and quality. It can be seen that we need to improve our memory. Cistanche deserticola can significantly improve memory, because Cistanche deserticola can also regulate the balance of neurotransmitters, such as increasing the levels of acetylcholine and growth factors. These substances are very important for memory and learning. In addition, meat can also improve blood flow and promote oxygen delivery, which can ensure that the brain receives sufficient nutrients and energy, thus improving brain vitality and endurance.

Click know ways to improve brain function
We reasoned that this learning-driven antagonistic activity relationship could be explained by a common circuit motif, namely, an inhibition between competing circuits (32). In this scenario, an upregulation of activity in one network would serve to drive a suppression in the antagonistic one. In support of this, CV1a receives large inhibitory inputs throughout the protraction phase during both spontaneous and PRN-driven egestion cycles (Fig. 3F). These inhibitory inputs did not arise directly from PRN, suggesting that downstream neurons were recruited.
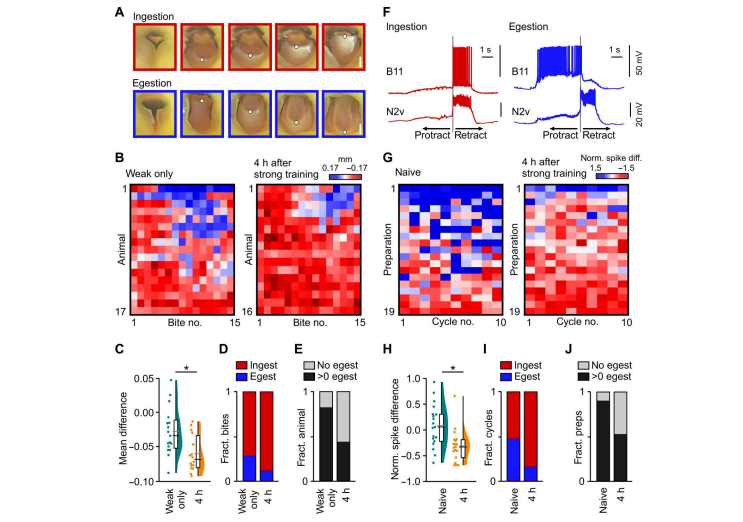
Fig. 2. Previous learning alters the perception of future training by shifting the feeding network state. (A) Example frames showing mouth movements during ingestion or egestion. Frames are color-matched to (B) (red: ingestion, blue: egestion). White dots indicate the distal tip of the radula tracked during bite classification. Scale bar, 0.5 mm. (B) Heat plots of radula movements during the first 15 bites in response to the CS + US during weak training in animals receiving weak training only versus animals receiving vital training 4 hours earlier. Red-white-blue lookup table represents radula movements. Positive (blue) is egestion; negative (red) is ingestion. (C) Statistical summary of (B) shows a significant change in the mean difference in radula movements between conditions (two-tailed t-test, P < 0.01, t = 3.1). (D) The plot of the fraction of ingestion/egestion bites produced during weak training shows a significant difference between conditions(Fisher’s exact test, P < 0.01). (E) The plot of fraction of animals performing no egestion bites versus >0 egestion bites shows a significant difference between conditions(Fisher’s exact test, P < 0.001). (F) B11 and N2v activity in an in vitro preparation during ingestion and egestion cycles. B11 is predominantly active in the retraction phase during ingestion and the protraction phase during egestion.
N2v activity does not change during ingestion and egestion cycles. B11 is therefore a readout of ingestion versus egestion. Gray lines represent the retraction phase onset. (G) Heat plots of B11 activity during fictive feeding cycles. Lookup table colors are normalized B11 spike differences. Positive (blue) is egestion; negative (red) is ingestion. (H) Statistical summary of (G) shows a significant change in the 11 spike difference between conditions (Mann-Whitney test, P < 0.01, U = 70). (I) The plotof fraction of ingestion/egestion cycles shows a significant difference between conditions (Fisher’s exact test, P < 0.001). (J) The plot of the fraction of preparations producing no egestion cycles versus >0 egestion cycles shows a significant difference between conditions (Fisher’s exact test, P < 0.001). h, hours.
Next, to identify the source of these inputs, we carried out an extensive search for a neuron type that would fulfill the following criteria: (i) It should inhibit CV1a when active, (ii) it should be active during the protraction phase of egestion cycles, and (iii) it should be excited by PRN activity. Using a fluorescence labeling approach to reveal neurons projecting from the buccal ganglia where most of the feeding circuitry is housed (33), we identified a single candidate neuron type, pattern switch 1 (PS1) (Fig. 4A), that satisfied all three criteria. First, artificial stimulation of PS1 monosynaptically inhibited the ipsilateral CV1a (Fig. 4, B and C). Second, this neuron was strongly active during the protraction phase of both PRN-driven and stimulus-driven egestion cycles (Fig. 4D and fFig.S9A) when CV1a was inhibited. Third, PRN activity excited PS1 monosynaptically, observed as 1:1 excitatory postsynaptic potentials (EPSPs) (Fig. 4, E and F). To ascertain whether PS1 was the sole source of inhibition to CV1a during egestion feeding behavior, we artificially manipulated its activity during PRN-driven cycles. When hyperpolarized, there was now a significant increase in CV1a activity (Fig. 4, G, and H) with no evidence for the phasic inhibitory synaptic inputs it normally receives. Moreover, PS1 hyperpolarization was also sufficient to increase CV1a activity during sensory-driven egestion cycles (fig. S9B). Thus, this pivotal inhibitory neuron type acts as a switch during action selection, preventing disruptive activation of the ingestion command centers during egestion.

Since CV1a does not have a monosynaptic connection with PRN, how does it ensure the suppression of egestion when it is active? We were able to answer this question by identifying the second component of the control circuit: a buccal interneuron type, PS2, which was strongly electrically coupled to PRN and sufficient to drive robust egestion cycles (fig. S9, C, and D). It receives strong facilitating inhibition from CV1a and thus leads to the suppression of egestion when ingestion cycles are generated (fig. S9, E, and F). Moreover, artificial activation of PS2 caused polysynaptic inhibitory inputs on CV1a, arising through its monosynaptic excitatory connection onto PS1 (fig. S9, G, and H). Together, these results demonstrate that mutual inhibition is used to prevent the activation of competitive circuits and that this circuit motif provides a control point on which plasticity could act (fFigS9I). Next, we explored the possible role of suppression of the egestion circuit in biasing the perception of learning events.

Manipulation of perceptual control circuit enables new learning in vivo
Given that it favors the expression of egestion behavior, we reasoned
that a reduction in PRN → PS1 activity might be sufficient to alter
an animal’s perception of weak training and thus enhance memory
acquisition. To investigate this, we developed a pharmacological
strategy that allowed us to manipulate the PRN output pathway.
We have previously shown that this neuron is dopaminergic and
that the D2 receptor blocker, sulpiride, is highly effective in inhibiting its action on follower motoneurons (16). Here, we confirmed
that the PRN → PS1 connection is also sulpiride sensitive (Fig. 5, A
and B), causing a significant reduction in the amplitude of the
PRN → PS1 EPSP. Next, we tested whether blocking this connection
could mimic the increase in CV1a activity observed 4 hours after
strong training. We found that sulpiride caused a robust elevation
in CV1a cycle activity versus pretreatment (Fig. 5, C, and D), consistent with our previous work showing that sulpiride application
biases activity toward ingestion events (16). Thus, sulpiride can robustly alter the network state, substituting for the effects of strong
training. As such, this agent provides the opportunity to test in vivo
whether this pathway underlies the animal’s altered perception of
the weak training. Animals were injected with either sulpiride or
normal saline and underwent the weak training protocol with
feeding responses measured as in Fig. 2 (A and B). We found that
sulpiride-injected animals performed significantly more ingestion
events in response to the weak training than saline-injected
animals (Fig. 5, E to G), and significantly more of the sulpiride-injected animals performed no egestion responses at all (Fig. 5H).
Moreover, there was a significant decrease in the transition probability between states after sulpiride injection (fig. S10, A to D).
Therefore, both intense training and sulpiride injection shift the
network state in vitro and stabilize the perception of weak training
in vivo.
We next tested whether the shift in perception induced by sulpiride was sufficient for the acquisition of LTM after weak training, as
we have shown in the case of strong training. Animals were injected
with sulpiride or saline and then underwent weak training with
LTM tested 1 day later. Consistent with the effects of strong training,
we found that animals injected with sulpiride before weak training
had a significantly greater response to GNL compared to naïve or
saline-injected trained animals (Fig. 5I). Furthermore, sulpiride injection in the absence of weak training did not increase the feeding
response to GNL when tested 1 day later (Fig. 5I). Therefore, pharmacologically manipulating the network state causes a change in the
perception of the weak training that is sufficient for the animal to
acquire and fully consolidate a memory.
Next, we examined whether the identified learning-induced change in network state after vital training was involved in the expression of the original strong memory or whether this was a parallel process that served the purpose of enhancing future learning events. To test this, we injected animals with sulpiride or saline and recorded their response to the strong training CS (AA) but in the absence of prior vital training. We hypothesized that if the learning-induced change in the network state was involved in the expression of the original memory, then feeding behavior in response to the CS (AA) used for intense training would be increased by artificially inducing the same change in the network state with sulpiride, but in the absence of prior vital training. However, we found that sulpiride injection did not cause an increase in the response to AA compared to naïve or saline-injected animals (Fig. 5J). Therefore, although the vital training causes a shift in the network state, this learning-induced change does not actively participate in the expression of the strong memory itself, suggesting that distinct mechanisms are involved. Together, these results demonstrate that strong learning causes parallel changes in neural activity: one for the expression of the memory itself and one for altering the perception of future appetitive learning.

Memory-linked shifts in perception generalize to an alternative training paradigm
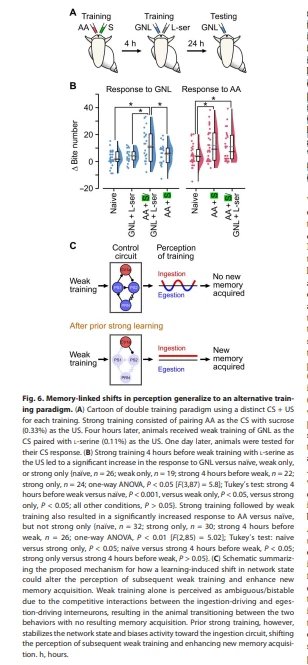
What role does the mechanism identified here serve? The ability to link strong and weak learning events that are closely temporally coupled suggests that Lymnaea could use this capability to identify “learning-rich” periods, for example, compatible with the arrival of the animal in a bountiful environment. If so, we would expect that learning should generalize rather than depend on the same US for both the strong and weak training. To test this important idea, we carried out experiments in which we substituted the sucrose US used in the weak training protocol with L-serine (Fig. 6A), a known alternative appetitive stimulus in Lymnaea (29). Thus, both the CS and US are distinct across the two different training paradigms. We found that while the GNL + L-serine pairing alone did not yield a conditioned response, a robust 1-day memory expression to GNL was seen if the strong training preceded it (Fig. 6B). Thus, memory expression is not confined to one US but can instead enable the formation of different associations, suggesting it would be highly appropriate for enabling a generalized lowering of the threshold for forming new memories.

DISCUSSION
The ability to learn new associations is critical for survival in an unpredictable environment. Given that memory acquisition and consolidation are energetically costly processes (1, 2, 34), there are key potential benefits in using strategies that help guide decisions about what and when to learn. Here, we identify a simple mechanism in Lymnaea by which past events are used to bias perception. Notably, we also demonstrate that this mechanism can guide future learning —facilitating memory acquisition for associations that would previously have been ignored—and we elucidate the neural circuits responsible. We propose that this serves to alert an animal to efficiently direct resources to new learning where recent experience suggests that there may be a particular advantage in forming positive associations.

A key feature of the mechanism we elucidate is that strong learning initiates parallel processes in the brain: one for the expression of the original memory and one to alter the perception of future learning events and facilitate new memory acquisition. These appear to be independent since the shift in neural network state measured after strong training is not sufficient for the expression of the original memory, as evidenced by the absence of a false memory when the network state is changed pharmacologically. Similar parallel pathways have been identified in conditioning experiments in mice. For example, in an olfactory-discrimination task, extensive training changed the excitability of pyramidal neurons in the piriform cortex coinciding with the enhanced capability to learn new tasks (rule learning), but this did not correlate with the expression of the original memory (35, 36). As such, while these learning-induced changes are not part of the memory-expressing “engram,” they nonetheless serve critical adaptive functions in animals, allowing them to use their past experiences to guide their future behavior—a process arguably as important as recall of the memory itself.
The mechanism that we have identified exhibits key timing characteristics. It permits enhanced learning capabilities from 30 min up to 4 hours after strong training, suggesting that new memory acquisition is facilitated in a critical time window. Informed by our previous work, this time frame aligns with the emergence of intermediate-term memory for strong training and lasts until the emergence of LTM (17). The expression of this memory over this time frame is protein synthesis-dependent, while the memory trace recorded at a shorter, 10-min time point is not (17). This strongly suggests that the shift in network state identified in our present study is also protein synthesis-dependent, explaining the absence of an effect at 10 min after strong training. Furthermore, the enhanced learning capability is not maintained at longer time points: Weak learning memory formation is absent from ~6 hours onward, although the original strong memory is still present and expressible. Therefore, the learning-induced shift in network state is transiently induced and does not outlast the molecular mechanisms known to be necessary for consolidation of the original memory during the first 6 hours after strong training (37). We hypothesize that a permanent shift in perception due to past learning could be detrimental to the animal, leading to energetically costly and potentially maladaptive memories. There is also a requirement for a strict temporal sequence of strong followed by weak training. This differs from a previously identified process, behavioral tagging (38, 39), which facilitates the interaction and enhancement of memories based on synaptic tags and capture (40, 41). During behavioral tagging, a tag set in motion by a weak learning event is targeted and enhanced by a strong learning event regardless of the temporal sequence of the two learning events (42, 43).
This key feature of behavioral tagging leads to an alteration of the memory of the event, not the learning event itself. By contrast, the learning-induced shift in perception we identify here guides the animal to decide which future events to learn about, rather than which recently acquired memories should be further consolidated. As such, while the two mechanisms have some parallels and both serve to increase the number of consolidated long-term memories, they function under different circumstances and use distinct circuit mechanisms. We also demonstrate that the learning-induced shift in perception generalizes to other forms of appetitive learning since a second type of weak appetitive training could also be enhanced by past strong learning. A similar generalized enhancement of learning capabilities has been found in mice, where hippocampus-dependent olfactory-discrimination learning is thought to switch the hippocampal network into a “learning mode,” enhancing other types of hippocampus-dependent education, such as spatial learning (44). The time course and nonspecific nature of the mechanism identified in Lymnaea could serve to alert the animal to a “learning-rich” period permitting positive associations to be made to cues that if encountered in isolation, would likely be disregarded.
What mechanism facilitates weak training after strong training? We note that naïve animals exposed to the weak training “flip-flop,” or transition, between ingestion and egestion behaviors, suggesting that the animal has a bistable perception of the CS + US stimuli during the training, which represents a period of ambiguity (45). Notably, we also find that the strong training biases and stabilizes the animal’s perception of the weak training to favor ingestion behavior. We suggest that bistable perception during weak training is an emergent property of the relative activity of the ingestion and egestion circuits since the same switching persists in vitro in the absence of any external stimulation. By characterizing the connectivity between the two circuits, we show that competitive interactions are due to a reciprocal inhibition circuit motif (32, 46), suggesting that action selection is likely generated by a winner-takes-all model. Feedforward inhibition from the egestion circuit prevents coactivation of the ingestion circuit (PRN + PS2 → PS1 → CV1a), whereas the ingestion circuit directly inhibits part of the electrically coupled egestion circuit (CV1a → PS2) (Fig. 6C and Fig. S9I).
Since both circuits are innervated by the same core feeding central pattern generator (16, 31), we propose that cycle expression is dictated by the first circuit to reach a sufficient spike-rate threshold to inhibit the other. After strong training, there is a shift in the relative activity of the two circuits and, as such, expression of ingestion cycles is dominant (Fig. 6C). A similar circuit motif is seen during fear conditioning in mice, where one of two incompatible behaviors can be generated: escape or freezing. Learning shifts the balance of two reciprocally inhibitory classes of neurons in the central amygdala, which biases expression toward one or the other behavior (47). In Lymnaea, however, the learning-induced shift in activity is not used for the expression of the memory but rather for the facilitation of new learning (Fig. 6C). We show that pharmacologically stabilizing the ingestion circuit by blocking the feedforward inhibition from the egestion circuit is sufficient to alter the animal’s perception of the CS + US during weak training in vivo. Furthermore, we show that this can substitute for the acquisition of a strong memory, shifting the network state to enable new positive associations, suggesting that memory formation can be facilitated by reducing ambiguity during a learning event. Previous studies in humans have demonstrated that prior learning can alter attentional control, which tunes perceptual sensitivity (3–5), and attention has also been shown to have a role in perceptual multistability (45, 48).
Although only the animal’s perceptual readout was measured in this study, it is possible that the mechanism identified here also includes an attentional shift that could modulate the perception of the weak training. The fact that the shift in network state is present before exposure to the weak training could suggest that there is an anticipatory attention mechanism that, in turn, tunes the perception of future learning events. However, if such an attentional shift is involved, it is only able to modulate perception and, thus, enhance new memory formation from ~30 min after the strong training.
Why does the stabilization of the network toward ingestion events facilitate weak training? One simple explanation is that, during ingestion, the animal actively draws the CS and US into the buccal cavity and then the esophagus, allowing each to be swallowed. This contrasts with egestion where contents are efficiently expelled from the buccal cavity. Earlier work has demonstrated that successful in vitro conditioning depends on the US reaching the esophagus (17) and activating esophageal neurons (49), which reinforce the CS via D1 receptor activation in follower neurons (50– 52). Therefore, favoring ingestion cycles will serve to increase the US reaching and activating these neurons thus reinforcing the CS + US.
Here, we have uncovered a so far unreported mechanism by which changes in perception can couple prior and new learning under specific conditions and time frames. We propose that this can serve as a general state-setting mechanism, allowing an animal to form associations between new combinations of stimuli, which, in isolation, would be insufficient to induce memory. Because associations between learning and perceptual changes are well established in higher animals, including humans, we suggest that a learning → perception → learning pathway may be a broadly conserved feature that deserves further attention in learning studies. Given that the formation of LTM is associated with increased energetic costs—particularly the engagement and recruitment of molecular machinery for memory consolidation—a mechanism that directs learning has potentially important value for survivability. In the case of a foraging animal such as Lymnaea, operating on a tight energy budget (53), learning different associations is highly beneficial, alerting them to potential food sources or possible dangers in their environment, but this needs to be balanced against the energetic costs of consolidating those memories. This mechanism thus allows them to tune their future learning to past learning success, by effectively lowering the threshold needed to learn new associations.
MATERIALS AND METHODS
Animal maintenance
Animals were kept in groups in large holding tanks containing Cu2+-free water at 20°C on a 12-hour light/12-hour dark regime. The animals were fed lettuce three times a week and vegetable-based fish food (Tetra-Phyll; TETRA Werke, Melle, Germany) twice a week. Animals were transferred to smaller holding tanks and food-deprived for 2 days before experiments. For all the experiments, adult (3 to 4 months old) snails (Lymnaea stagnalis) were used. Lymnaea is a lower invertebrate (molluscan) organism that does not fall under The Animals (Scientific Procedures) Act 1986 (UK). Therefore, no ethical approval or guidance was required for these experiments.
Single-trial appetitive training and testing procedures
Strong single-trial appetitive conditioning was performed by pairing AA (0.004%) as the CS with sucrose (0.33%) as the US using a previously well-described method (23, 24). Weak singletrial appetitive conditioning was performed by pairing GNL (0.004%) as the CS with sucrose (0.11%) or L-serine (0.11%) as the US. In a counterbalance control experiment, strong conditioning was performed by pairing GNL as the CS with sucrose (0.33%) as the US and weak conditioning was performed by pairing AA as the CS with sucrose (0.11%) as the US. Briefly, animals were placed individually in Petri dishes containing 90 ml of Cu2+-free water for 10 min to acclimatize to the new environment before the training procedure began. Five milliliters of the CS was added to the water, and 30 s later, 5 ml of the US was applied. Animals were left in the solution containing the CS and US for 2 min, then rinsed in Cu2+-free water, and returned to their home tanks. To test for CS responses 1 day after conditioning, animals from trained and naïve groups were transferred from their home tanks to a petri dish filled with 90 ml of Cu2+-free water and allowed to acclimatize for 10 min.
Five milliliters of water was then added to the dish and the number of feeding responses (bites) in the following 2 min was counted. Next, 5 ml of the CS was added to the dish, and the number of feeding responses was counted in the subsequent 2 min. CS responses were then assessed using a “difference score”(∆ bite number). This was obtained by subtracting the number of feeding cycles observed during 2 min after water application from the number of feeding cycles in 2 min after CS application. During dual conditioning experiments, animals received both strong and weak appetitive conditioning separated by time intervals as described in the results. To test whether preexposure to the US used during intense training (0.33% sucrose) enhanced weak training learning, we performed the intense training as described above but in the absence of presentation of the CS (AA). Animals then received the weak training of GNL and sucrose (0.11%) 4 hours later and were tested for their response to GNL 1 day later.
Measurement of perception during weak training
The effect of past learning on the animal’s perception of weak training was tested by performing vital training followed by weak training 4 hours later as above. During the weak CS + US presentation, animals’ feeding responses were videoed (33 frames/s) from below. The direction of movement of the radula and underlying odontophore structure was measured during the first 15 feeding responses as in (16). Briefly, the position of the dorsal mandible was first marked in the frame preceding the first frame in which the radula was visible during each bite using ImageJ software. The radula was then tracked for the entire bite, and the distance from the initial position of the dorsal mandible was calculated. The average difference moved between frames was then measured. A negative score, therefore, represented the radula and dorsal mandible being apart at the start of the bite and the radula moving toward the dorsal mandible during the bite, whereas a positive score represented the mandible and radula being close together at the start of the bite and the radula moving away as the bite progressed. The criteria to define whether a response was ingestion or egestion were based on whether the difference in movement was negative (ingestion) or positive (egestion). To measure how prior vital training alters the stability of the perception of the CS + US used in weak training, pairs of consecutive bites were analyzed. A stable bite pair was classified as two of the same consecutive bites (ingest-ingest or egest-egest). A switching bite pair was classified as two different consecutive bites (ingest egest or egest-ingest).
Transition probabilities were then calculated by counting the number of switching bites expressed as a fraction of the total number of bite pairs. To test whether preexposure to the US used during intense training (0.33% sucrose) altered the perception of weak training, we performed the intense training as described above but in the absence of presentation of the CS (AA) and measured ingestion/egestion behaviors as above. To test whether prior weak training altered the perception of later weak training, animals first received AA paired with 0.11% sucrose, and then their ingestion/egestion responses to GNL and 0.11% sucrose were measured 4 hours later. To test for the effects of strong training on the responsiveness of the animal to either the CS or US used during weak training, animals received the intense training as above. Four hours later, animals were placed in a petri dish of 90 ml of Cu2+-free water and allowed to acclimatize for 10 min. They then received 5 ml of water, and their feeding responses were counted. They subsequently received 5 ml of either GNL or 0.11% sucrose, and feeding responses were counted so that the ∆ bite number could be calculated as above.
Preparations and electrophysiological methods
Following procedures previously described in (16), we carried out in vitro experiments using an isolated CNS preparation. A small region of the anterior esophagus was kept attached to the CNS by the dorsal buccal nerves. Preparations were perfused with normal saline containing 50 mM NaCl, 1.6 mM KCl, 2 mM MgCl2, 3.5 mM CaCl2, and 10 mM Hepes buffer in water. Monosynaptic connections were tested by bathing the preparation in a high divalent (HiDi) saline, which increases the action potential threshold, reducing polysynaptic connections. HiDi saline was composed of 35.0 mM NaCl, 2 mM KCl, 8.0 mM MgCl2, 14.0 mM CaCl2, and 10 mM Hepes buffer in water. Intracellular recordings were made using sharp electrodes (10 to 40 megohms) filled with 3 M KAc and 0.5 mM KCl. Signals were collected using NL 102 (Digitimer Ltd.) and Axoclamp 2B (Axon Instrument, Molecular Device) amplifiers, and data were acquired using a micro 1401 Mk II interface and analyzed using Spike2 software (Cambridge Electronic Design, Cambridge, UK).
Neuron identification
The phase-switching motoneuron B11 is located in the buccal ganglia and was identified based on its location, spike shape, synaptic inputs from PRN, and ability to switch its activity pattern during ingestion and egestion cycles (16). The ingestion command-like interneuron CV1a is located in the cerebral ganglia and was identified by its electrical properties, characteristic location, and its ability to drive fictive feeding cycles when artificially depolarized to fire spikes (31). The egestion command-like interneuron PRN is located in the buccal ganglia and was identified by its location and monosynaptic excitatory connection to B11. The N2v neuron is a central pattern generator interneuron located on the ventral surface of the buccal ganglia. It can be identified by its characteristic plateau during the retraction phase of a cycle, and artificial activation causes widespread retraction phase activity in many buccal neurons (25). B9 is a retraction phase motoneuron located in the buccal ganglia. The CGCs are large, serotonergic interneurons located in the cerebral ganglia, which can be identified by their size, location, and tonic-spiking activity (30). To identify previously uncharacterized candidate members of the egestion network, we backfilled the cerebrobuccal connective (CBC) with the fluorescent dye, 5(6)-carboxyfluorescein (5-CF). Projection interneurons are known to be influential in driving patterned activity in Lymnaea (30, 31), and egestion-driving neurons have been identified in the buccal ganglia (16). Backfilling the CBC revealed a population of buccal projection interneurons that we could reidentify in other preparations and test electrophysiologically. Neurons of interest were impaled and recorded with ingestion and egestion command-like neurons.
Analysis and classification of in vitro cycles
Activity on B11 was measured concerning the onset of the retraction phase, as determined by the N2v plateau or large excitation on retraction phase interneuron B9. To analyze the relative activity of B11 in a cycle, it was measured 4 s before and 4 s after retraction phase onset. The number of B11 spikes after the retraction phase onset was subtracted from the number of B11 spikes before the retraction phase onset and then divided by the total number of spikes in the 8-s period to gain a normalized difference score. Using this score, a positive value represents more activity occurring before the retraction phase onset and therefore is classified as an egestion cycle. A negative score represents more activity occurring after the retraction phase onset and is therefore classified as an ingestion cycle. To compare the effects of strong training on fictive feeding cycles in vitro, the first 10 spontaneous cycles were analyzed from 19 naïve and 19 trained preparations. To measure how training alters the stability of cycle expression, pairs of consecutive cycles were analyzed. A stable cycle pair was classified as two of the same consecutive cycles (ingest-ingest or egest-egest).
A switching cycle pair was classified as two different consecutive cycles (ingest-egest or egest ingest). Transition probabilities were then calculated by counting the number of switching cycles expressed as a fraction of the total number of cycle pairs. To test whether vital training alters the responsiveness of the preparation to appetitive cues, we stimulated the main chemosensory pathway, the MLN (28), which can drive vital fictive feeding (29). The MLN was stimulated using a glass suction electrode with biphasic pulses of 4 V with 0.5-ms duration at 1 Hz for 120 s. The ∆ fictive feeding cycle number was calculated by recording activity in feeding motoneurons, such as B9, counting the number of cycles that occurred in the 120-s period preceding MLN stimulation, and subtracting this from the number of cycles in response to MLN stimulation. CGC activity was measured for the 120 s before and 120 s during the MLN stimulation. To elicit sensory-driven egestion in vitro, a 1-s tactile stimulus was applied to the esophagus, which activates mechanosensory neurons that signal aversive cues to the feeding network in response to overextension of the gut due to an inedible object lodged in the esophagus (16). The tactile stimulus was applied using a mechanical probe controlled by a transistor–transistor logic pulse from the micro 1401 Mk II (CED).
Iontophoretic dye filling of neurons
Following procedures previously described in (16), we filled target neurons with a fluorescent dye (5-CF) using a microelectrode. This was achieved iontophoretically using a pulse generator to apply regular interval negative square current pulses into the neuron for >30 min. Preparations were then left overnight at 4°C. Images of the neurons were taken using a digital camera (Andor Ixon electron-multiplying charge-coupled device) mounted on a Leica stereomicroscope.
D2 receptor blocker application in vitro and in vivo
Sulpiride is an effective dopamine antagonist in Lymnaea, blocking the effects of dopaminergic interneurons on follower neurons as well as the focal application of dopamine (16, 54). To test for the effect of sulpiride (±) (Sigma-Aldrich) on the PRN → PS1 connection, preparations were first bathed in HiDi saline (see above). Baseline EPSP amplitudes were recorded before 10−4 M sulpiride in HiDi saline was perfused into the bath for 10 min and then EPSP amplitudes were recorded again. To test the effects of sulpiride on in vitro cycle generation in naïve preparations, the first 10 spontaneous cycles generated were recorded and then 10−4 M sulpiride in normal saline was perfused onto the preparation. The first 10 spontaneous cycles generated after 10 min of perfusion were analyzed. To test the effects of sulpiride on the perception of weak training and memory acquisition/recall, animals were injected with 100 μl of 10−3 M sulpiride in normal saline. It has previously been shown that the injected concentration of the drug is diluted ~10-fold by the body fluids of the animal (55). Control animals were injected with 100 μl of normal saline alone. Animals were left for 2 hours before behavioral tests were carried out.

Data analysis
Data were analyzed using paleontological statistics (PAST version 4.1) (56) and expressed as raincloud plots (57). In all cases, individual points are plotted as dots, and the shaded region (cloud) indicates the overall shape of the distribution extending from minimum to maximum values. Internal boxplots show median (black line) interquartile range (first and third quartile) and mean (gray line). Each “n” represents an individual animal/preparation. Normality was tested using the Shapiro-Wilk test. Two-group statistical comparisons were performed using two-tailed t-test statistics (either paired or unpaired as stated in the text) or a Mann-Whitney test or Wilcoxon signed-rank test for nonparametric data. Data with more than two groups were first analyzed using a one-way analysis of variance (ANOVA) or the Kruskal-Wallis test. Subsequent comparisons were performed using Tukey’s or Dunn’s post hoc tests with Bonferroni sequential correction. The comparisons between the percentage of bites/cycles classified as ingestion or egestion and the number of animals/preparations performing zero or more than zero egestion cycles were made using a Fisher’s exact test. The significance level was set at P < 0.05.
REFERENCES AND NOTES
1. F. Mery, T. J. Kawecki, A cost of long-term memory in Drosophila. Science 308, 1148 (2005).
2. P.-Y. Plaçais, T. Preat, To favor survival under food shortage, the brain disables costly memory. Science 339, 440–442 (2013).
3. A. C. Nobre, M. G. Stokes, Premembering experience: A hierarchy of time-scales for proactive attention. Neuron 104, 132–146 (2019).
4. M. L. Rosen, C. E. Stern, S. W. Michalka, K. J. Devaney, D. C. Somers, Cognitive control network contributions to memory-guided visual attention. Cereb. Cortex 26, 2059–2073 (2016).
5. M. G. Stokes, K. Atherton, E. Z. Patai, A. C. Nobre, Long-term memory prepares neural activity for perception. Proc. Natl. Acad. Sci. U.S.A. 109, E360–E367 (2011).
6. Y. L. Chew, Y. Tanizawa, Y. Cho, B. Zhao, A. J. Yu, E. L. Ardiel, I. Rabinowitch, J. Bai, C. H. Rankin, H. Lu, I. Beets, W. R. Schafer, An afferent neuropeptide system transmits mechanosensory signals triggering sensitization and arousal in C. elegans. Neuron 99, 1233–1246.e6 (2018).
7. J. Felsenberg, P. F. Jacob, T. Walker, O. Barnstedt, A. J. Edmondson-Stait, M. W. Pleijzier, N. Otto, P. Schlegel, N. Sharifi, E. Perisse, C. S. Smith, J. S. Lauritzen, M. Costa, G. Jefferis, D. D. Bock, S. Waddell, Integration of parallel opposing memories underlies memory extinction. Cell 175, 709–722.e15 (2018).
8. J. Huang, Z. Zhang, W. Feng, Y. Zhao, A. Aldanondo, M. G. de Brito Sanchez, M. Paoli, A. Rolland, Z. Li, H. Nie, Y. Lin, S. Zhang, M. Giurfa, S. Su, Food wanting is mediated by transient activation of dopaminergic signaling in the honey bee brain. Science 376, 508–512 (2022).
9. P. F. Jacob, S. Waddell, Spaced training forms complementary long-term memories of opposite valence in Drosophila. Neuron 106, 977–991.e4 (2020).
10. S. Sayin, J.-F. De Backer, K. P. Siju, M. E. Wosniack, L. P. Lewis, L.-M. Frisch, B. Gansen, P. Schlegel, A. Edmondson-Stait, N. Sharifi, C. B. Fisher, S. A. Calle-Schuler, J. S. Lauritzen, D. D. Bock, M. Costa, G. S. X. E. Jefferis, J. Gjorgjieva, I. C. Grunwald Kadow, A neural circuit arbitrates between persistence and withdrawal in hungry Drosophila. Neuron 104, 544–558.e6 (2019).
11. B. Senapati, C.-H. Tsao, Y.-A. Juan, T.-H. Chiu, C.-L. Wu, S. Waddell, S. Lin, A neural mechanism for deprivation state-specific expression of relevant memories in Drosophila. Nat. Neurosci. 22, 2029–2039 (2019).
12. K. Steck, S. J. Walker, P. M. Itskov, C. Baltazar, J. M. Moreira, C. Ribeiro, Internal amino acid state modulates yeast taste neurons to support protein homeostasis in Drosophila. eLife 7, e31625 (2018)
For more information:1950477648nn@gmail.com






