An Effective Method To Treat Renal Fibrosis
Mar 17, 2022
for more information:ali.ma@wecistanche.com
Part Ⅰ:Endoplasmic reticulum protein TXNDC5 promotes renal fibrosis by enforcing TGF-β signaling in kidney fibroblasts
Yen-Ting Chen, Pei-Yu Jhao & et al.
Introduction
Renal fibrosis, characterized by tubulointerstitial fibrosis and glomerular sclerosis, is a common pathological manifestation of most, if not all, types of chronic kidney diseases (CKD)(1). The progression of renal fibrosis in patients with CKD leads to the gradual loss of kidney function, which could ultimately result in end-stage renal disease (ESRD) that requires dialysis or kidney transplantation (2). While multiple molecular determinants and pathways have been shown to contribute to the process of kidney fibrosis, transforming growth factor-beta (TGF-β) is generally regarded as the master regulator of renal fibrogenesis (3). Following an injury to the kidney, dam-aged tubular epithelial cells (TECs) are arrested in the G2/M phase and secrete large amounts of TGF-β, triggering the activation and transformation of adjacent renal fibroblasts into myofibroblasts (4-6). Activated myofibroblasts, featured by the expression of a-smooth muscle actin (an SMA), are highly proliferative and produce extracellular matrices (ECM)proteins like fibrillar collagen, elastin, fibronectin, and CCN2(commonly known as connective tissue growth fac-tor, CTGF)(7,8). During the early stages of kidney injury, fibrosis was initiated as an adaptive and self-limited physiological response that helps repair tissue and maintain structural integrity(9). However, following chronic, repeated renal injury, the activation, and proliferation of myofibroblasts persist, resulting in excessive ECM production and accumulation, leading to the loss of functional kidney parenchyma and ultimately, kidney failure (1O,11).
Click to Cistanche para que sirve for kidney disease
In spite of the advances in modern medicine, few clinical therapies are proven effective against kidney fibrosis. Angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers(ARBs), for example, have been shown to reduce proteinuria, ameliorate renal fibrosis, and slow down the progression of CKD (12,13). Pentoxifylline, a nonspecific phosphodiesterase inhibitor, on the other hand, has been shown to inhibit TGF-β1-induced ECM and CCN2 expression, leading to attenuated tubulointerstitial fibrosis (14). The use of ACEIs and ARBs, however, is limited by their hypotensive effects and potential adverse effects like hyperkalemia. In addition, neither ACEls/ARBs nor pentoxifylline treatment could halt the progression of renal fibrosis and CKD(15,16). It is, therefore, imperative to identify novel mediators or pathways that are critical for the pathogenesis of renal fibrosis to develop effective therapeutics against CKD( chronic kidney diseases).

Recently, we identified thioredoxin domain containing 5 (TXNDC5), an ER-resident, fibroblast-enriched protein with the enzyme activity of a protein disulfide isomerase (PDI), as a critical mediator of cardiac fibrosis(17). We showed that excessive TXNDC5 promotes the development of cardiac fibrosis through redox-dependent cardiac fibroblast activation and ECM production. Genetic deletion of Txndc5 ameliorates cardiac fibrosis and contractile dysfunction induced byβ agonist in mice (17). Based on the observed essential role of TXNDC5 in myocardial fibrogenesis, we hypothesized that TXNDC5could also contribute critically to the development of tissue fibrosis in noncardiac organs, particularly kidney fibrosis.

In the present study, we showed that TXNDC5 was highly upregulated in both human and mouse fibrotic kidneys, specifically in the collagen-secreting renal fibroblasts. Global deletion of Txndc5 markedly attenuated the extent of renal fibrosis in multiple mouse models of kidney injury. Mechanistically, we demonstrated that excessive TXNDC5, induced by TGF-β1 via ATF6-dependent ER stress pathway, augments renal fibrosis by promoting-kidney fibroblast activation/proliferation and ECM production through posttranslational stabilization and upregulation of TGF-βreceptor type I(TGFBR1). Importantly, we showed that inducing fibroblast-specific deletion of Txndc5 effectively ameliorated the development and progression of kidney fibrosis induced by various types of injuries in mice, suggesting that targeting TXNDC5 could be a novel and powerful approach to treat renal fibrosis and preserve kidney function.

Figure 1. TXNDC5 was significantly upregulated in mouse fibrotic kidneys and kidney specimens from patients with CKD ( chronic kidney diseases).
(A) IHC staining (n = 3) and (B) immunoblots (n = 6) showed protein expression of TXNDC5 was upregulated in mouse fibrotic kidneys induced by UUO or uIRI, compared with contralateral kidneys (CL). Scale bar: 50 μm.
(C) Quantitative RT-PCR showed Txndc5 transcript was upregulated in mouse fibrotic kidneys induced by UUO and uIRI (n = 4–7).
(D) Reanalyses of microarray data on human kidney specimens from patients with CKD (GSE66494) showed that TXNDC5, COL3A1, and FN1 were significantly upregulated in the kidney tissues from CKD patients (healthy control n = 8, CKD n = 51). For A–C, data are representative of 3 or more independent experimental replicates.
For all panels, data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 by 2-sided t-test (A–C) or Mann-Whitney test (D).
Results
TXNDC5was significantly upregulated in human and mouse fibrotic kidneys. To investigate the potential involvement of TXNDC5 in the pathogenesis of kidney fibrosis, we first determined its protein expression levels in the fibrotic mouse kidney tissues induced by unilateral ureteral obstruction (UUO), unilateral ischemia-reperfusion injury (uIRI), or by the administration of folic acid (FA). Immunohistochemistry studies revealed marked increases in the intensity of TXNDC5 staining in the kidney sections from all 3 mouse models of renal fibrosis (Figure 1A and Supplemental Figure 1A; supplemental material available online with this article;https://doi.org/10.1172/JCI143645DS1). Immunoblotting and quantitative reverse transcription PCR(RT-PCR) also showed significant upregulation of the protein(Figure 1B) and transcript(Figure 1C and Supplemental Figure 1B) expression levels ofTXNDC5 in fibrotic mouse kidneys. Consistent with these results, reanalysis of microarray data obtained from renal biopsy specimens of patients with CKD(GSE66494)(18)showed that the expression levels of TXNDC5(by 2.24 fold, P<0.001), as well as of markers for kidney fibrosis such as COL3A1and FN, were also significantly increased in the kidney samples from patients with CKD compared with those from healthy controls(Figure 1D and Supplemental Figure 1C). Taken together, the observed upregulation of TXNDC5/Txndc5 expression in fibrotic human/mouse kidneys and its positive correlation with fibrogenic ECM protein genes suggest a potential role of TXNDC5 in the pathogenesis of kidney fibrosis.
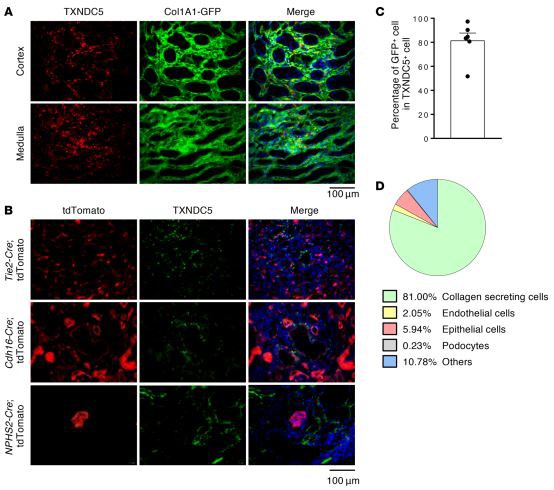
Figure 2. TXNDC5 was highly upregulated in renal fibroblasts, but not in TECs, endothelial cells, or podocytes, of the fibrotic kidneys.
(A and C) IF staining of TXNDC5 (red) on sections of fibrotic kidneys induced by UUO in Col1a1-GFPTg mice showed TXNDC5 was mainly expressed in collagen-secreting renal fibroblasts (green), both in renal cortex and medulla (n = 6). Cell nuclei were stained with DAPI (blue). Scale bar: 100 μm.
(B) IF staining of TXNDC5 (green) on the section of fibrotic kidneys induced by UUO in Cdh16-Cre, NPHS2-Cre, and Tie2-Cre/ERT2 tdTomato mice. Cell nuclei were stained with DAPI (blue). Scale bar: 100 μm.
(D) A pie chart to illustrate the proportion of TXNDC5+ cells in different types of kidney cells in the UUO-induced fibrotic kidneys.
Data are representative of 3 or more independent experimental replicates. Data in C are presented as mean ± SEM.
TXNDC5 was highly enriched in collagen-secreting renal fibroblasts in the fibrotic kidneys. Multiple types of kidney cells have been implicated in the process of kidney fibrogenesis. Following kidney injuries, resident (19) and perivascular(20)renal fibroblasts were shown to differentiate into active myofibroblasts and result in the accumulation of ECM. TECs and endothelial cells were also reported to contribute to the development of kidney fibrosis through epithelial- and endothelial-to-mesenchymal transition, respectively (10,21).To determine the cell types where TXNDC5 was expressed during the process of renal fibrosis, multiple reporter mouse lines that allow fluorescence labeling of renal fibroblasts(Colla1-GFP', GFP driven by Collal enhancer/promoter, ref.22), TECs(Cdh16-Cre ROSA26-tdTomato), endothelial cells(Tie2-Cre/ERT2 ROSA26-tdTomato), and podocytes (NPHS2-Cre ROSA26-tdTomato) were subjected to UUO, uIRI, or FA treatment to induce kidney fibrosis. Immunofluorescence (IF)staining of the kidney sections from these animals revealed that TXNDC5 was markedly upregulated and highly colocalized with GFP-positive, collagen-secreting renal fibroblasts in fibrotic mouse kidneys induced by UUO(81%)(Figure 2, A and C), uIRI (76.8%), and FA treatment (73.4%)(Supplemental Figure2, A and B).In UUO-induced fibrotic kidneys,TXNDC5was only expressed sporadically in TECs(5.94%),podocytes(0.63%),and endothelial cells(2.05%)(Figure 2,B and D).Flowcytometry analysis of fibroblasts isolated from mouse kidneys subjected to UUO revealed a marked expansion in TXNDC5* kidney fibroblasts as well as a significant upregulation ofTXNDC5 in these cells, compared with those from contralateral control kidneys(Supplemental Figure 2C). The observed strong enrichment of TXNDC5 in renal fibroblasts in fibrotic suggests that TXNDC5 could contribute to kidney fibrosis by regulating the activity/behavior of these collagen-secreting fibrogenic cells.

TXNDC5 was both essential and sufficient to induce the activation, proliferation, and ECM production of kidney fibroblasts. Next, we determined the functional role of TXNDC5 in kidney fibroblasts. Primary human kidney fibroblasts (HKFs)stimulated with TGF-β1(10 ng/mL) showed strong upregulation in the protein (Figure 3A) and transcript(Figure 3B)expression levels of TXNDC5, as well as of fibroblast activation marker periostin and ECM proteins(COL1Al, fibronectin, and CCN2). Knockdown of TXNDC5 with shRNA significantly attenuated TGF-β1-induced protein/mRNA upregulation of periostin and multiple ECM proteins in HKFs(Figure 3, A and B). In addition, TGF1 treatment enhanced the proliferative activity of HKFs, which was abrogated completely by TXNDC5 knockdown (Figure 3D). These results suggest that TXNDC5 is essential for TGF-β1-induced HKF activation, proliferation, and ECM production. Overexpression of TXNDC5, on the other hand, resulted in marked upregulation of periostin, COL1A1, and fibronectin(Figure 3C), as well as increased proliferative activity(Figure 3E) in HKFs. Collectively, these results demonstrate that TXNDC5, downstream of TGF-β1, is both essential and sufficient to promote the activation, proliferation, and ECM production of kidney fibroblasts.

Figure 3. Knockdown of TXNDC5 attenuated TGF-β1–induced HKF activation and ECM production; overexpression of TXNDC5 was sufficient to trigger HKF activation and ECM production.
(A) Protein and (B) transcript expression levels of fibroblast activation marker (periostin) and ECM proteins (COL1A1, fibronectin, and CCN2) were increased in control (Scramble) HKFs following TGF-β1 (10 ng/mL) treatment. Knockdown of TXNDC5 attenuated the upregulation of these fibrogenic markers induced by TGF-β1 in HKFs (n = 5–10).
(C) Overexpression of TXNDC5 was sufficient to induce upregulation of fibroblast activation marker (Periostin) and ECM proteins (COL1A1, fibronectin) in HKFs (n = 3–10).
(D) Treatment of TGF-β1 (10 ng/mL) increased the cellular proliferation activity of HKFs, which was abrogated by TXNDC5 knockdown.
(E) Overexpression of TXNDC5 increased the cellular proliferation activity of HKFs. In D and E, n = 10. Data are representative of 3 or more independent experimental replicates.
For all panels, data are presented as mean ± SEM. The statistical significance of differences for 2 groups was determined by a 2-sided t-test and among 3 or more groups it was determined using 1-way ANOVA, followed by Sidak’s post hoc tests. *P < 0.05, **P < 0.01, ***P < 0.001.
Global deletion of Txndc5 protected against kidney b. To determine the requirement of TXNDC5 in the development of kidney fibrosis in vivo, WT and Tndc5-/mice were subjected to UUO, uIRI, and FA treatment. Picrosirius red and Masson's trichrome staining on the kidney sections showed extensive renal fibrosis in WT mice following UUO(Figure 4A),uIRI (Figure 5A), and the administration of FA(Supplemental Figure 3A). Global deletion of Txndc5 significantly reduced the extent of renal fibrosis in all 3 kidney injury models (Figure 4A, Figure 5A, and Supplemental Figure 3A).

Figure 4. Deletion of Txndc5 attenuated renal fibrosis induced by UUO.
(A) Picrosirius red staining (top 2 panels) and Masson's trichrome staining (bottom 2 panels) of kidney sections from WT and Txndc5-7mice 10 days after UUO. Bar graphs of the quantitative results of Picrosirius red and Masson's trichrome staining areas are shown on the right (n=5-10).Scale bar∶ 100 μm.
(B)SHG images of the kidney sections from WT and Txndc5-/mice 10 days after UUO. The quantitative results of SHG-positive areas showed increased accumulation of fibrillar collagen (green) in WT but not in Txndc5mice kidneys following injury.
For each of the kidney sections imaged for SHG, TPEF imaging was obtained to show the profile of the scanned tissue (red color in bottom panels)(n=3).Scale bar: 50 um. (C)Immunoblots showed protein expression levels of fibroblast activation marker(Periostin)and ECM(COL1A1) in the whole-kidney extract from WT and Txndc5-/mice 10 days after UUO (n= 3-6). Data are representative of 3or more independent experimental replicates.
For all panels, data are presented as mean± SEM. The statistical significance of differences among3 or more groups was determined using 1-way ANOVA, followed by Sidak's post hoc tests.**P<0.01,***P< 0.001.
To quantify pathogenic fibrillar collagen (type I and II)specifically and to avoid overestimation of fibrotic areas with traditional staining methods, second harmonic generation (SHG) microscopy, a novel optical tissue imaging system that allows visualization of fibrillar, but not non-fibrillar, collagen in fibrotic organs(23,24), was employed to assess the extent of fibrosis in WT and Tndc5+mouse kidneys following injury. Consistent with the aforementioned results, SHG microscopy showed marked increases in fibrillar collagen deposition in WTkidneys following UUO(Figure 4B),uIRI (Figure 5B), and FA treatment (Supplemental Figure 3B), the extent of which was significantly reduced in Txndc5-/ mice (Figure 4B, Figure 5B, and Supplemental Figure 3B).
Consistent with the attenuated postinjury fibrotic response in Txndc5mouse kidneys observed by imaging studies, deletion of Tendc5 also mitigated the upregulation of ECM protein genes, including Collar, Eln, Fnl, and Ccn2 in the mouse kidneys following injury (Supplemental Figure 3, C-E). Immunoblots of whole-kidney tissue lysates also showed reduced protein level of ECM(COLIA1)and fibroblast activation marker (periostin)in Txndc5-/mice compared with WT mice following UUO and uIRI (Figure 4C and Figure 5C).
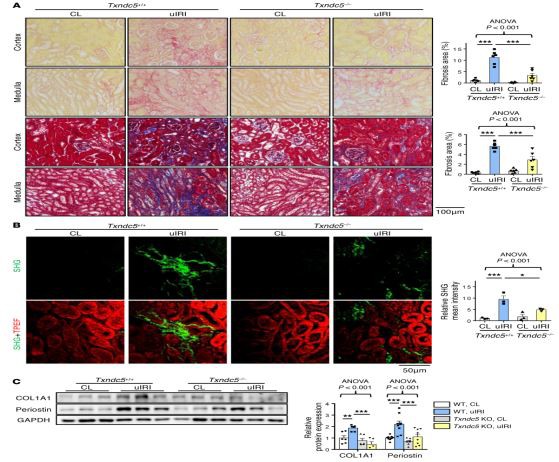
Figure 5. Deletion of Txndc5 ameliorated renal fibrosis induced by ulRI.
(A) Picrosirius red staining (top 2 panels) and Masson's trichrome staining (bottom 2 panels) of kidney sections from WT and Txndc5-7mice 28 days after ulRI.Bar graphs of the quantitative results of Picrosirius red and Masson's trichrome staining areas are shown on the right (n=6). Scale bar∶ 100 μm.
(B) SHG images of the kidney sections from WT and Txndc5-mice 28 days after ulRI. The quantitative results of SHG-positive areas showed increased accumulation of fibrillar collagen (green) in WT but not in Txndc5mice kidneys following injury. For each of the kidney sections imaged for SHG, TPEF imaging was obtained to show the profile of the scanned tissue(red color in bottom panels)(n=3).Scale bar∶50 μm.
(C) Immunoblots showed protein expression levels of fibroblast activation marker(Periostin) and ECM(COL1A1) in the whole-kidney extract from WT and Txndc5/mice 28 days after ulRI (n=5-9). Data are representative of 3 or more independent experimental replicates.
For all panels, data are presented as mean ± SEM. The statistical significance of differences among 3or more groups was determined using1-way ANOVA, followed by Sidak's post hoc tests.*P< 0.05,**P<0.01,***P<0.001.
To exclude the possibility that TXNDC5 expressed in other renal cell types such as TECs, podocytes, and endothelial cells might still contribute to the development of renal fibrosis, we generated Txndc5 conditional knockout mice specific to TECs (Cdh16-cre*Txcndc5i, or Txndc5Epeko), podocytes (NPHS2-cre*Txndc5/n, or Tendc5Podxko), and endothelial cells(Tie2-cre/ERT2*Tendc5/, or Txndc5Endo.eko).In experiments using Txndc5Pieko and Txndc5 Poio-cko mice, Txndc5Wmicewereused as controls; in experiments on Txndc5EMio-eko mice, Txndc5 deletion was induced using tamoxifen as described in Figure 9A, where tamoxifen-treated Tie2-Cre/ERT2 mice were used as controls. Targeted deletion of Txndc5 in TECs, podocytes, or endothelial cells, however, did not impact the extent of kidney fibrosis induced by UUO or FA (Supplemental Figure 6, B-E). These results suggest that TXNDC5 expressed in TECs, podocytes, and endothelial cells contribute little, if any, to the pathogenesis of kidney fibrosis.
Deletion of Txndc5 in kidney fibroblasts protects against TEC apoptosis in response to kidney injury. In the process of renal fibrosis, injured TECs lose mitochondrial function and apicobasal polarity, followed by apoptosis and cell death(30).To investigate if deletion of Txndc5 impacts apoptosis of TECs in response to injury, we conducted TUNEL staining on WT and Txndc5-mouse kidney sections 10 days following UUO. The number of apoptotic TECs increased significantly in the UUO-induced fibrotic kidneys of WT mice, whereas deletion of Txndc5 attenuated the extent of TECapoptosis. It has been reported that activated kidney fibroblasts could induce TEC apoptosis through paracrine effects (31). Because Tendc5 is barely expressed in TECs, it is likely that the reduced TEC apoptosis observed in Txndc5-7mice following UUO was not a cell-autonomous effect but rather secondary to reduced interstitial fibrosis and hence few-er proapoptotic signals from surrounding kidney fibroblasts. Consistent with this hypothesis, fibroblast-specific deletion of Txndc5 (Tendc5kO) attenuated TEC apoptosis to an extent similar to that observed in Txndc5-/mice 10 days following UUO(Supplemental).In contrast to the observed reduction in TECapoptosis, the number ofF4/80-positive macrophages and CD31-positive endothelial cells were comparable in WT and Txndc5-mouse kidneys, with or without UUO, suggesting that Txndc5 deletion does not impact macrophage infiltration or peritubular capillary density in the mouse kidneys. Taken together, these data suggest that the reno-protective effects of Txndc5 deletion result from reduced tubular interstitial fibrosis and TEC apoptosis, without affecting renal capillary density or macrophages.
Induced deletion of Tendc5 in renal fibroblasts lessened the progression of established kidney fibrosis. To determine if targeting Txndc5 in renal could be beneficial in established renal fibrosis, Txndc5 deletion in renal fibroblasts was induced in Txndc5eko mice 10 days after UUO, a time point when extensive renal fibrosis can be observed. In these experiments, tamoxifen-treated Colla2-Cre mice were used as controls. As shown, Colla2-Cre and Tcndc5eko mice had a similar extent of renal fibrosis 10 days following UUO prior to tamoxifen treatment. Eleven days after tamoxifen injection, however, the extent of renal fibrosis continued to progress in control mice (fibrotic area increased from 5.1%to 10.6%), whereas the progression of kidney fibrosis was nearly halted (fibrotic area changed from 5.3% to 6.9%) in Txndc5eko mice. Immunoblotting also showed markedly attenuated upregulation of CCN2, periostin, and TGFBR1 in Txndc5eko mice compared with control mice between D10 and D21 after UUO. These results demonstrate that interventional deletion of Txndc5 in renal fibroblasts significantly lessens the progression of kidney fibrosis, suggesting that in vivo targeting of TXNDC5 could be a novel and powerful therapeutic approach to ameliorate kidney fibrosis and to slow down the progression of CKD.






