Are Natural Plant Compounds Effective In Treating Chronic Kidney Disease?
Mar 24, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
Part Ⅱ: Potential Therapeutic Effects of Natural Plant Compounds in Kidney Disease
Lorena Avila-Carrasco, Elda Araceli García-Mayorga & et al.
Abstract
Background: The blockade of the progression or onset of pathological events is essential for the homeostasis of an organism. Some common pathological mechanisms involving a wide range of diseases are the uncontrolled inflammatory reactions that promote fibrosis, oxidative reactions, and other alterations. Natural plant compounds(NPCs) are bioactive elements obtained from natural sources that can regulate physiological processes. Inflammation is recognized as an important factor in the development and evolution of chronic renal damage. Consequently, any compound able to modulate inflammation or inflammation-related processes can be thought of as a renal protective agent and/or a potential treatment tool for controlling renal damage. The objective of this research was to review the beneficial effects of bioactive natural compounds on kidney damage to reveal their efficacy as demonstrated in clinical studies. Methods: This systematic review is based on relevant studies focused on the impact of NPCs with therapeutic potential for kidney disease treatment in humans. Results: Clinical studies have evaluated NPCs as a different way to treat or prevent renal damage and appear to show some benefits in improving OS, inflammation, and antioxidant capacity, therefore making them promising therapeutic tools to reduce or prevent the onset and progression of KD pathogenesis. Conclusions: This review shows the promising clinical properties of NPC in KD therapy. However, more robust clinical trials are needed to establish their safety and therapeutic effects in the area of renal damage.
Keywords: natural plant compounds; kidney disease; renoprotective effects; bioactive compounds; clinical studies
kidney disease treatment:desertliving cistanche extract
Introduction
Historically, natural plant compounds (NPCs) have been used as treatments and their chemical structures could give rise to new therapeutic options [1]. NPCs are active substances isolated from plants that can modulate several pathways, including the epithelial-to-mesenchymal transition (EMT)via antioxidant, anti-inflammatory, or anti-fibrotic mechanisms [1]. Chronic kidney disease(CKD) is a recurrent public health problem worldwide [2]. Several diseases(e.g., hypertension, diabetes, cancers, infection, drug-induced nephrotoxicity) may disturb kidney function, eventually leading to CKD(Chronic kidney disease) development. Around the world, the prevalence of CKD(Chronic kidney disease) is 13.4%(11.7-15.1%) and it has been estimated that patients with end-stage renal disease(ESRD), which requires kidney substitution therapy, a number between 4.902 and 7.083 million, making it a leading cause of death around the world [2,3]. The higher number of CKD(Chronic kidney disease) patients over recent years has spurred great interest in searching for efficient approaches to avoid or decrease the development of CKD(Chronic kidney disease) and its progression to ESRD. Traditional treatments provide limited kidney protection, so new therapeutic compounds are necessary. Currently, inflammation is recognized as a key unfavorable pathway in the progression and development of CKD(Chronic kidney disease); consequently, new anti-inflammatory compounds targeting or directed towards specific molecular signatures may be promising therapeutic approaches for CKD(Chronic kidney disease). NPCs derived from herbs or medicinal plants have become important in preclinical and clinical research for the development of such targets. Several NPCs have been shown to have a renoprotective effect and improve the outcomes of disorders present in various categories of CKD(Chronic kidney disease) in clinical studies, mainly through activating antioxidant defense systems and decreasing pro-inflammatory signaling pathways(Table 1; Figure 1)[3]. This review collected comprehensive evidence of the kidney-protective potential of bioactive plant compounds.

desertliving cistanche
Molecular Mechanisms Involved in Kidney Damage
Kidney damage is a global public health problem that has been growing worldwide. Renal diseases involve acute kidney injury(AKI) and CKD(Chronic kidney disease), which can lead to ESRD. Consequently, patients needing renal replacement therapy are estimated to number between 4.902 and 7.083 million [2,3]. The worldwide projected prevalence of CKD(Chronic kidney disease) is 13.4%(11.7-15.1%)[2]; likewise, in 2017, the global prevalence of CKD(Chronic kidney disease) was 9.1%, ie., approximately 700 million cases [29]. CKD(Chronic kidney disease) is frequently asymptomatic with fast progression and is commonly associated with hypertension [30] and Type 2 diabetes mellitus (T2DM). These diseases are considered the leading causes of the development of CKD(Chronic kidney disease) and ESRD[31]. Nevertheless, CKD(Chronic kidney disease) is a chronic disorder characterized by albuminuria(>30 mg) for 24 h, a reduced glomerular filtration rate(GFR)(<60 mL/min/1.73 m-)for more than 3 months, and progressive glomerular, tubular, and interstitial damage [32-34]. The prognosis of CKD(Chronic kidney disease) is classified according to the GFR and albuminuria categories, GF grades(G1 to G5), and albuminuria grades(A1 to A3)(Figure 2), according to the Kidney Disease: Improving Global Outcomes guidelines (KDIGO 2012), [35].
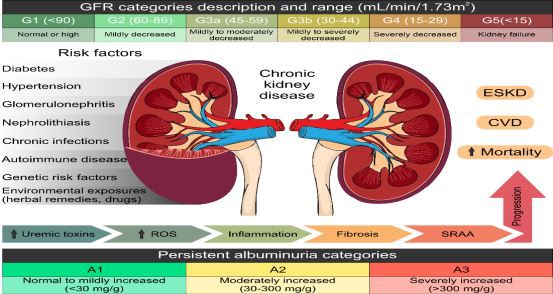
Figure 2. Causes, definition and prognosis of chronic kidney disease classed by glomerular filtration rate and albuminuria.
Categories are grouped by the risk of progression, defined by a decline in the GFR category. Adapted from the Kidney Disease: Improving Global Outcomes(KDIGO2012)categories of the National Kidney Foundation (NFK).ESKD,(Chronic kidney disease);CVD, cardiovascular disease; CKD(Chronic kidney disease), chronic kidney disease; GFR, glomerular filtration rate. Some elements of this figure were taken from the Mind the Graph platform, available at www.mindthegraph.com.(Last date of access;30 September 2021).
Numerous molecular signaling pathways are implicated in the onset and progression of renal damage, including the renin-angiotensin system (RAS), which is the central regulator of kidney fibrosis. Together, tissue and circulatory RAS are believed to be overstimulated in the progress of kidney fibrosis stimulation. RAS increases levels of angiotensin II (Ang I), which plays a dominant role in kidney fibrosis by mediating the liberation of transforming growth factor-β (TGF-β) and starting inflammatory processes [36-39]. Likewise, Ang Ⅱ and inflammation may be the key factors in the glomerulotubular pathogenic response and have been related to microalbuminuria and the development of kidney damage [40]. In patients with diabetes and severe hyperglycemia, many induced metabolic and hemodynamic disorders occur, including the increased development of advanced glycation end products (AGEs), augmented reactive oxygen species (ROS) generation, and initiation of the protein kinase C(PKC) and polyol pathway, which are believed to induce the development and evolution of diabetic nephropathy [39]. Another molecular pathway contributing to kidney damage is the formation of AGEs, which promote intracellular nitric oxide (NO) and ROS, and the mitogen-activated protein kinase (MAPK) cascade. The creation of AGEs could induce kidney damage by altering the role of proteins, oxidative stress(OS), pro-inflammatory cytokines, and growth factors [41-43]. In addition, the AGE-RAGE interaction induces activation of the nuclear factor kappa-light-chain-enhancer of triggered B cells (NF-kB) and activation of MEK and MAP kinases, increasing intracellular OS by stimulating NADPH-oxidase, an important regulator in superoxide radical generation [44,45]. Likewise, ROS and free radicals may respond to membrane lipids, nucleic acids, and proteins, and start to produce cellular injury. When ROS are created in excess quantities, OS, antioxidant defense mechanisms and cell damage can occur, which accelerates the progression of CKD(Chronic kidney disease). Other pathways associated with CKD(Chronic kidney disease) pathogenesis are stress stimuli and the activation of profibrotic growth factors such as TGF-β1, connective tissue growth factor (CTGF), and hydrogen peroxide (H2O2). In addition, TGF-β has been demonstrated to increase ROS production and to decrease the antioxidant system, thus inducing OS and/or redox imbalance. Essentially, redox imbalance significantly helps TGF-β's pathophysiologic properties such as fibrosis [46,47]. TGF-β may drive towards kidney fibrosis through the accumulation of the extracellular matrix(ECM) and activation of epithelial dysfunction and pro-inflammatory reactions. TGF-β is the most powerful stimulator of EMT, and this can induce this process in the epithelial cells of various organs in vivo and in vitro [48-51]. Signaling pathways such as the MAPK, Smad, and PI3K pathways are related to the stimulation of EMT by TGF-β [52,53]. Essentially, ROS come from many sources, such as the mitochondria or NOXs, which have been exposed to TGF-β-induced EMT at the beginning of fibrosis and cancer [49,50,54-56]. Recent evidence has shown the process of linking ROS to TGF-β-activated EMT in the context of fibrosis [48,49,57]. Rhyu et al.discovered that NADPH oxidase-originated ROS are upstream signaling particles in TGF-β1-stimulated fibronectin and production of the plasminogen activator inhibitor-1(PAI-1). This step happens throughout MAPK stimulation in the tubular epithelial cells [57]. Likewise, other research has suggested that ROS plays a significant role in TGF-β1-activated EMT, mainly through MAPK stimulation [58,59]. These pathways overlap and interact with each other, thereby altering their biological activities, which promotes the evolution of kidney fibrosis and exacerbates kidney damage (Figure 3). Consequently, any pharmacologic agents that are capable of preventing these disadvantageous pathways can be thought of as renoprotective and a potential therapeutic approach in the control of kidney damage.

Figure 3. Alterations related to kidney injury, including diabetes, hypertension, and vascular damage(CVD and CAD) and the molecular mechanisms involved in inflammation, fibrosis, lipid disorders, and oxidative stress(highlighted in boxes). Some elements of this figure were taken from the Mind the Graph platform, available at www.mindthegraph.com (Last date of access; 30 September 2021).
CLICK HERE TO PART Ⅰ FOR MORE
Discussion
Renal disease usually develops over time, so it is not often diagnosed until significantly later when renal function is severely impaired. Pathophysiologically, CKD(Chronic kidney disease) is a consequence of numerous pathological lesions that abolish some of the nephrons; subsequently, the nephrons overcompensate with hyperfiltration. Over time, glomerular hypertension, albuminuria, and loss of renal activity are established. The increased glomerular capillary pressure triggers damage to the glomerular capillary endothelium, impairment of the podocytes lining the capillaries, and increased permeability of the macro-molecules. [118,119]. Additionally, increased pro-inflammatory moderators that induce fibrotic cell proliferation are involved. Furthermore, the increase in ECM molecules gives rise to the development of scarring and renal damage [120. Currently, there are therapeutic approaches to CKD(Chronic kidney disease), with all alternatives targeted at dismissing or preventing deterioration of the disorder, including medication, conservative care, dialysis, and transplantation [121].
Research has recommended that diets rich in vegetables and fruits help to control body weight and defend against chronic disorders, including metabolic and cardiovascular disease, cancer, and CKD(Chronic kidney disease) [20,122,123]. Only a limited number of trials have analyzed bioactive compound supplementation in humans. Further research must be undertaken to establish the best dosage and method of administration of bioactive compounds [114,115]. Furthermore, future investigations should be directed towards analyzing the particular signaling/cellular mechanisms modulated by bioactive compounds that contribute to the prevention or attenuation of renal damage. Further investigations are also needed to evaluate the pharmacokinetic parameters of bioactive compounds, such as the ideal route of administration, dosage and bioavailability, metabolism, tissue distribution, and clearance, and pharmacodynamic parameters, such as the molecular mechanisms of action of bioactive compounds, to demonstrate their efficacy and safety in the short and long term in clinical studies.

desertliving cistanche extract treat kidney diseases
Conclusions
This review highlights the beneficial function of bioactive medicinal plant compounds in improving renal function. All-natural compounds explained in this report appear to have some benefits in improving OS, inflammation, and antioxidant capacity. Several NPCs reduce fibrosis due to hyperglycemia-induced OS, attenuate proteinuria, decrease hematuria and systolic blood pressure, and exert anti-nephrotoxic effects, thus they are promising therapeutic tools to reduce or prevent the onset and progress of KD pathogenesis. Natural bioactive compounds are considered to exert important actions within the mechanistic pathways and to possess promising clinical properties; however, additional investigations are necessary to obtain a deeper comprehension of their specific pathophysiological functions, and the evaluation of these effects in larger trials is strongly suggested.

treatment of chronic kidney disease: Natural cistanche herb
References
1. Avila-Carrasco, L.;Majano, P.;Sanchez-Tomero, J.A.;Selgas,R.; Lopez-Cabrera, M.; Aguilera, A.; Gonzalez Mateo, G.Natural Plants Compounds as Modulators of Epithelial-to-Mesenchymal Transition. Front.Pharmacol. 2019, 10, 715. [CrossRef]
2. Ly. L.C; Zhang, LX.Prevalence and Disease Burden of Chronic Kidney Disease. Adv. Exp. Med. Biol.2019, 1165,3-15.[CrossRef]
3. Chen, T.K.; Knicely, D.H.; Grams, M.E. Chronic Kidney Disease Diagnosis and Management: A Review. IAMA 2019, 322, 1294-1304. [CrossRef]
4. Zare, E.; Alirezaei, A.; Bakhtiyari, M.; Mansouri, A.Evaluating the effect of garlic extract on serum inflammatory markers of peritoneal dialysis patients: A randomized double-blind clinical trial study, BMC Nephrol, 2019.20.26. [CrossRefl [PubMed]
5. Mashhadi, N.S.; Zakerkish, M.; Mohammadiasl, J.; Zarei, M.; Mohammadshahi, M.; Haghighizadeh, M.H. Astaxanthin improves glucose metabolism and reduces blood pressure in patients with type 2 diabetes mellitus. Asia Pac. J. Clin. Nutr. 2018, 27, 341-346. [CrossRef [PubMed]
6. Shokri-Mashhadi, N; Tahmasebi, M.; Mohammadi-Asl, J.; Zakerkish, M.; Mohammadshahi, M. The antioxidant and anti-inflammatory effects of astaxanthin supplementation on the expression of miR-146a and miR-126 in patients with type 2 diabetes mellitus: Arandomized, double-blind, placebo-controlled clinical trial. Int.J.Clin. Pract. 2021, 75, e14022.[CrossReflPubMed]
7. Dong, S.; Sun, L. Traditional Chinese drug baicalin and insulin therapy on pancreatic beta-cell function in newly diagnosed type 2 diabetes. Chin. Med.2013, 8, 348-350.
8. Yang, M.; Kan, L.; Wu, L.; Zhu, Y; Wang, Q. Effect of baicalin on renal function in patients with diabetic nephropathy and its therapeutic mechanism. Exp. Ther. Med.2019,17, 2071-2076. [CrossRef][PubMed]
9. Rahimi,P:Mesbah-Namin,SA;Ostadrahimi. A; Abedimanesh, S.; Separham, A.; Asghari Jafarabadi, M.Effects of Betalains on Atherogenic Risk Factors in Patients with Atherosclerotic Cardiovascular Disease. Food Funct. 2019,10, 8286-8297.[CrossRe]
10. Kemmner, S.; Lorenz, G.; Wobst, J.; Kessler,T.; Wen, M.; Günthner, R.; Stock, K.; Heemann, U.; Burkhardt, K.; Baumann, M.; et al. Dietary nitrate load lowers blood pressure and renal resistive index in patients with chronic kidney disease: A pilot study. Nitric Oxide 2017, 64, 7-15.[CrossRef]
11. Li, Z.Y.; Liu, B.; Zhuang, X.J.; Shen, Y.D.; Tian, H.R.; Ji, Y.; Li, LX.; Liu, F.Effects of berberine on the serum cystatin C levels and urine albumin/creatine ratio in patients with type 2 diabetes mellitus. Zhonghua Yi Xue Za Zhi Chin. 2018, 98, 3756-3761. [CrossRef]
12. Sun, T;Dong, W;Jiang, G.; Yang,J.;Liu,J.;Zhao, L.;Ma, P.Cordyceps militaris mejora la enfermedad renal cronica al afectar la via de senalizacion redox TLR4/NF-k B.Oxid Med. Cell Longev.2019,7850863. [CrossRef]
13. Alvarenga, L.; Salarolli, R.; Cardozo, L.F.M.F.; Santos, R.S.; de Brito, J.S.; Kemp, J.A.; Reis, D.; de Paiva, B.R.; Stenvinkel, P.; Lindholm, B.; et al. Impact of curcumin supplementation on the expression of inflammatory transcription factors in hemodialysis patients: A pilot randomized, double-blind, controlled study. Clin. Nutr. 2020, 39, 3594-3600. [CrossRef]
14. Panahi, Y; Kianpour, P; Mohtashami, R.; Jafari, R.; Simental-Mendia, L.E.; Sahebkar, A.curcumin lowers serum lipids and uric acid in subjects with nonalcoholic fatty liver disease: A randomized controlled trial. J. Cardiovasc. Pharmacol.2016,68, 223-229. [CrossRef]
15. Jimenez-Osorio, A.S.;Garcia-Nino, W.R.;Gonzalez-Reyes, S.;Alvarez-Mejia, A.E.;Guerra-Leon, S.;Salazar-Segovia, J.;Falcon, I.;Montes de Oca-Solano, H.; Madero, M.;Pedraza-Chaverri, J. The effect of dietary supplementation with curcumin on redox status and Nrf2 activation in patients with nondiabetic or diabetic proteinuric chronic kidney disease: A Pilot Study. I.Ren.Nutr. 2016, 26,237-244.[CrossRef] [PubMed]
16. Khajehdehi, P.; Pakfetrat, M.; Javidnia, K.; Azad, F.; Malekmakan, L.; Nasab, M.H.; Dehghanzadeh, G.Oral supplementation of turmeric attenuates proteinuria, transforming growth factor-β and interleukin-8 levels in patients with overt type 2 diabetic nephropathy: A randomized, double-blind and placebo-controlled study. Scand. J.Urol. Nephrol.2011,45, 365-370. [CrossRef][PubMed]
17. Khajehdehi, P; Zanjaninejad, B.; Aflaki, E.; Nazarinia, M.; Azad, F.; Malekmakan, L.Oral supplementation of turmeric de- creases proteinuria, hematuria, and systolic blood pressure in patients suffering from relapsing or refractory lupus nephritis: A randomized and placebo-controlled study. J. Ren. Nutr. 2012,22, 50-57. [CrossRef]
18. Pakfetrat, M.; Basiri, F; Malekmakan, L.; Roozbeh, J. Effects of turmeric on uremic pruritus in end-stage renal disease patients: A double-blind randomized clinical trial. J. Nephrol.2014, 27, 203-207. [CrossRef]
19. Shoskes, D.; Lapierre, C.; Cruz-Correa, M.; Murase, N.; Rosario, R.; Fromkin, B.; Braun, M; Copley, J.Beneficial effects of the bioflavonoids curcumin and quercetin on early function in cadaveric renal transplantation: A randomized placebo-controlled trial. Transplantation 2005, 80, 1556-1559. [CrossRef] [PubMed]
20. Borges, C.M.; Papadimitriou, A.; Duarte, D.A.; Lopes de Faria, J.M; Lopes de Faria, J.B. The use of green tea polyphenols for treating residual albuminuria in diabetic nephropathy: A double-blind randomized clinical trial. Sci. Rep. 2016, 6, 28282. [CrossRef] [PubMed]
21. Chen, T.-S.; Liou, S.-Y.; Wu, H.-C.;Tsai, F.-J.;Tsai, C.-H;Huang, C.-Y;Chang,Y.-L.Efficacy of Epigallocatechin-3-Gallate and Amla (Emblica officinalis)Extract for the Treatment of Diabetic-Uremic Patients. J. Med. Food 2011,14,718-723. [CrossRef]
22. Ushida, Y.; Suganuma, H.; Yanaka, A.Low-dose sulforaphane precursor glucoraphanin as a dietary supplement induces chemoprotective enzymes in humans. Food Nutr. Sci. 2015, 6, 1603-1612. [CrossRef]
23. Tracy, C.R.; Henning, J.R.; Newton, M.R.; Aviram, M.; Zimmerman, M.B. Oxidative stress and nephrolithiasis: A comparative pilot study evaluating the effect of pomegranate extract on stone risk factors and elevated oxidative stress levels of recurrent stone formers and controls. Urolithiasis 2014, 42, 401-408. [CrossRef]
24. Sattarinezhad, A.; Roozbeh, J.; Shirazi Yeganeh, B.; Omrani, G.R.; Shams, M.Resveratrol reduces albuminuria in diabetic nephropathy: A randomized double-blind placebo-controlled clinical trial. Diabetes Metab.2019, 45,53-59.[CrossRef] [PubMed]
25.Brasnvo, P.; Molnar, G.A.; Mohas, M; Marko, L.;Laczy, B.;Cseh, J.; Mikolas,E.; Szijarto, I.A.; Merei, A.; Halmai, R; et al. Resveratrol improves insulin sensitivity, reduces oxidative stress, and activates the Akt pathway in type 2 diabetic patients. Br. I. Nutr. 2011, 106, 383-389. [CrossRef [PubMed]
26. Lin, C.-T; Sun, X.-Y; Lin, A.-X. Supplementation with high-dose trans-resveratrol improves ultrafiltration in peritoneal dialysis patients: A prospective, randomized, double-blind study. Ren. Fail.2016, 38,214-221. [CrossRefl
27. Marques, B.C.A.A.; Trindade, M; Aquino, J.C.E; Cunha, A.R.; Gismondi, R.O; Neves, M.E: Oigman, W.Beneficial effects of acute trans-resveratrol supplementation in treated hypertensive patients with endothelial dysfunction. Clin. Exp.Hvpertens. 2018, 40, 218-223. [CrossRef [PubMed]
28. Mirmiran, P.; Bahadoran, Z.; Hosseinpanah, F.; Keyzadc, A.; Azizi, F. Effects of broccoli sprout with high sulforaphane concentration on inflammatory markers in type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. J. Funct. Foods 2012,4,837-841.[CrossRef]
29. Cockwell, P.; Fisher, L.-A. The global burden of chronic kidney disease.Lancet 2020, 395, 662-664. [CrossRef]30. Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P.Chronic kidney disease. Lancet 2017, 389,1238-1252. [CrossRef]
31. Kovacs, N.; Nagy, A.; Lombardi, V.; Biro, K.Inequalities in the Global Burden of Chronic Kidney Disease Due to Type 2 Diabetes Mellitus: An Analysis of Trends from 1990 to 2019.Int.J. Environ. Res. Public Health 2021,18,4723. [CrossRef][PubMed]







