Dental Mesenchymal Stem Cell Secretome: An Intriguing Approach For Neuroprotection And Neuroregeneration Part 3
Aug 14, 2024
Early pre-symptomatic DPSC-CM administration improved neuromuscular junction innervation compared to vehicle-treated SOD1G93A mice.
Neuromuscular and memory are closely related. Our nervous system and muscle system are interdependent, and they work together to complete various activities of our body. And this cooperation is also very important for our memory.
First, our nervous system is responsible for transmitting information and controlling the movement of various parts of the body. When we learn something new, our brain will produce new neurotransmitters and synapses, which can help us better remember new knowledge.
Secondly, our muscle system is also closely related to our memory. Studies have shown that our muscle memory can affect our brain memory, that is, through muscle training, we can improve our memory.
More specifically, when we do muscle training, our muscles will form muscle memory. This muscle memory can help us better control our muscles and make our movements more accurate and coordinated. At the same time, this muscle memory can also enhance our brain memory because our brain will record our muscle movements and sensations.
Therefore, we can improve our memory by doing muscle training. For example, we can do balance training, dancing, playing table tennis, and other sports, which can help us exercise our muscles, improve our coordination and reaction ability, and thus enhance our muscle memory and brain memory.
In summary, the relationship between neuromuscular and memory is very close. We need to maintain good health and perform proper muscle training to improve our memory. At the same time, we also need to pay attention to diet, rest, and mental health, and maintain a good living habit to comprehensively improve our physical and brain health. It can be seen that we need to improve memory. Cistanche can significantly improve memory because Cistanche has antioxidant, anti-inflammatory, and anti-aging effects, which can help reduce oxidation and inflammatory reactions in the brain, thereby protecting the health of the nervous system. In addition, Cistanche can also promote the growth and repair of nerve cells, thereby enhancing the connectivity and function of neural networks. These effects can help improve memory, learning ability, and thinking speed, and can also prevent the occurrence of cognitive dysfunction and neurodegenerative diseases.

Click know ways to improve brain function
The administration during late pre-symptomatic stages not only increased neuromuscular junction preservation but also motor neuron survival in the spinal cord ventral horn.
However, astrogliosis and microglia reactivity remained unaffected. Interestingly, the daily DPSC-CM treatment from symptom onset increased post-onset survival as well as overall lifespan [69].
DPSC-EXO administration in a murine model of transient middle cerebral artery occlusion (tMCAO) injury reduced brain edema, cerebral infarction, and neurological impairment. DPSC-EXOs inhibited the ischemia/reperfusion (I/R)-mediated expression of Toll-like receptor (TLR) 4, MyD88, and Nuclear Factor kappa-light-chain-enhancer of activated B cells (NF-κB).
DPSC-EXOs also reduced the protein expression of pro-inflammatory cytokines IL-6, IL-1β and TNF-α, and cytoplasmic translocation of high mobility group box protein (HMGB) 1 in vivo but also in vitro in OGD/reperfusion (OGD/R)-induced BV2 cells.
Thus, the results indicated that DPSC-EXOs may exert neuroprotection against cerebral I/R-induced neuroinflammation via the inhibition of the HMGB1/TLR4/MyD88/NFκB signaling pathway [70].
DPSC-CM ameliorated aneurysmal subarachnoid hemorrhage (aSAH)-induced vasoconstriction and improved oxygenation in an injured brain. DPSC-CM administration also ameliorated cognitive and motor impairments.
DPSC-CM administration decreased neuroinflammation as demonstrated by the reduction in the number of Iba1-positive cells. The major constituent of DPSC-CM was IGF-1.
Antibody-mediated neutralization of IGF-1 moderately deteriorated the rescuing effect of DPSC-CM on microcirculation, Iba1-positive cells in the injured brain area, and cognitive/motor impairments [71].
DPSC-CM administration ameliorated sciatic motor/sensory nerve conduction velocity, sciatic nerve blood flow, and intraepidermal nerve fiber density in the footpads of streptozotocin-induced diabetic rats.
Furthermore, the capillary density of the skeletal muscles increased while pro-inflammatory reactions in the sciatic nerves of diabetic rats were reduced [72]. Kanada et al. confirmed the positive effects of CM in sciatic nerve conduction velocity and sciatic nerve blood flow.
Moreover, the treatment also increased muscle bundle size, vascular density in the skeletal muscles, and intraepidermal nerve fiber density in diabetic rats.
However, no differences were found between the results for DPSCs and DPSC-CM. These results suggested that the efficacy of DPSC and DPSC-CM administration was probably due to the secretome.
In particular, DPSC-CM contained angiogenic factors such as VEGF-C, neurotrophic factors, such as BDNF, and immunomodulatory factors including IL-1β, IL-4, and TLR4 [73]. An overview of the studies presented in this paragraph is available in Table 2.
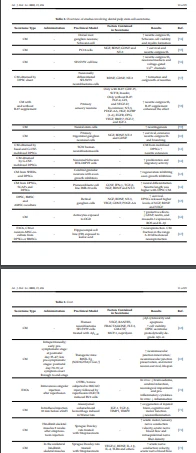
Aβ, amyloid β; aSAH, aneurysmal subarachnoid hemorrhage; AMSCs, adipose tissue derived MSCs; BDNF, brainderived neurotrophic factor; BMP, bone morphogenetic protein; BMSCs, bone marrow MSCs; CM, conditioned medium; DFSCs, dental follicle stem cells; DPSCs, dental pulp stem cells; EXOs, exosomes; FGF, fibroblast growth factor; GDNF, glial-cell-derived neurotrophic factor; GFAP, glial fibrillary acidic protein; G-CSF, granulocyte colony-stimulating factor; HGF, hepatocyte growth factor; IFN, interferon; IGF, insulin-like growth factor; IGFBP, insulin-like growth factor binding protein; IL, interleukin; MCP, Monocyte Chemoattractant Protein; MSCs, mesenchymal stem cells; NGF, nerve growth factor; NT, neurotrophin; OGD/R, oxygen-glucose deprivation– reperfusion; OPG, osteoprotegerin; PDGF, platelet-derived growth factor; ROS, reactive oxygen species; SCAPs, stem cells from apical papilla; SHEDs, stem cells from human exfoliated deciduous teeth; TGF, transforming growth factor; TIMP, tissue inhibitor of metalloproteinase; TLR, Toll like receptor; tMCAO, transient middle cerebral artery occlusion; VEGF, vascular endothelial growth factor; ↑, increase/improvements; ↓, reduction.

3.2. Stem Cells from Human Exfoliated Deciduous Teeth Secretome
The secretome of SHEDs was reported to modulate microglial cell activity. EVs derived from SHEDs inhibited lipopolysaccharide (LPS)-induced activation of NF-κB signaling pathway in human microglial cells.
Moreover, EVs induced an upregulation of phagocytic activity in unpolarized cells, a slight decrease in M1 polarized cells, and a moderate increase in M2 polarized cells. EVs induced an immediate and sustained increase of glycolytic activity in M0, M1, and M2 polarized cells. Interestingly, EVs acted in an inverse dose-dependent manner [74].
EVs also induced a rapid increase in intracellular Ca2+ and ATP release in microglial cells. EVs were also able to promote microglial motility through P2X4 receptor/milk fat globule-epidermal growth factor-factor VIII (MFG-E8)-dependent mechanisms [75].
Different studies report beneficial effects of SHED CM in both in vitro and in vivo Parkinson's disease (PD) models. Fujii et al. evidenced that dopaminergic-neuron-like cells induced from SHEDs were able to exert therapeutic benefits in a 6-hydroxy-dopamine (6-OHDA)-induced Parkinsonian rat model, improving neurological deficits and increasing dopamine (DA) levels more efficiently than undifferentiated SHEDs.
However, paracrine effects may contribute to neuroprotection against 6-OHDA-induced neurodegeneration. Indeed, the CM obtained from differentiated SHEDs was able to protect primary neurons against 6-OHDA toxicity and accelerated neurite outgrowth in vitro [76].
Different doses of SHED-CM were tested in a PD model. The dose of 10 µg/mL of SHED-CM did not restore motor ability, while 30 µg/mL of SHED-CM induced only mild improvements. Instead, 100 µg/mL of SHED-CM induced the maximal improvement of motor deficits in PD rats and a higher dose did not induce further improvement.
SHED-CM increased tyrosine hydroxylase (TH) amounts and decreased synuclein levels in both the substantia nigra and striatum. In addition, SHED-CM treatment decreased both Iba-1 positive cells and CD4 levels in the same brain areas.
The major constituents of SHED-CM included insulin-like growth factor binding protein-6 (IGFBP-6), tissue inhibitor of metalloproteinase (TIMP)-2, TIMP-1, and TGF-1. Moreover, bioinformatics analysis indicated that SHEDCM was able to promote neural regeneration.
Indeed, RNA sequencing evidenced that SHED-CM administration shifted the gene expression profile to a pattern similar to that of control rats, upregulating genes that were involved in neurodevelopment and nerve regeneration.
The major constituents of SHED-CM may participate in the molecular networks involved in cholinergic and serotoninergic synapses, calcium signaling pathways, and axon guidance [77].
EXOs and MVs derived from SHEDs have also been evaluated for their neuroprotective effects in PD models. EXOs, but not MVs, derived from SHEDs grown on laminin-coated three-dimensional alginate micro-carriers suppressed 6-OHDA-induced apoptosis in dopaminergic neurons. On the contrary, no protective effects were exerted by MVs or EXOs derived from SHEDs grown in standard culture conditions [78].
Instead, intranasal administration of EVs derived from SHEDs was shown to be effective in a rat model of PD, improving motor function in association with the normalization of TH expression in the striatum and substantia nigra [79].
The intranasal administration was also tested in an AD model, showing that SHEDCM improved cognitive function. SHED-CM reduced oxidative stress, shifted the M1-type pro-inflammatory microenvironment toward the M2-type anti-inflammatory and neuroprotective one, and increased neurotrophic factor levels. BMSCs-CM was less efficacious.
It reduced oxidative stress and inflammation, but could not upregulate the expression of the anti-inflammatory M2 markers. Treatment with SHED-CM also suppressed glutamate-induced neuronal death in vitro [80]. SHED-CM was also able to improve disease scores and reduce demyelination, axonal injury, inflammatory cell infiltration, and proinflammatory cytokine expression in the spinal cord of experimental autoimmune encephalomyelitis (EAE) mice.
These changes were associated with a change in the microglia/macrophage phenotype from M1 to M2. The treatment of EAE mice with the secreted ectodomain of sialic-acid-binding Ig-like lectin-9 (ED-Siglec-9), a major component of SHED-CM, resulted in similar effects compared to SHED-CM treatment, while ED-Siglec-9 depletion abolished the protective effects of SHEDCM.
On the contrary, HGF depletion did not cause an inhibition of SHED-CM-mediated protection, indicating that HGF had little effect on the efficacy of SHED-CM. SHED-CM inhibited the proliferation of myelin oligodendrocyte glycoprotein-specific CD4+ T cells, as well as their production of proinflammatory cytokines in vitro [81].
Matsubara et al. showed that SHED and SHED-CM administered into rats' injured spinal cords during the acute postinjury period induced functional recovery. SHED-CM showed anti-inflammatory activity, reducing the levels of pro-inflammatory cytokines, and immunoregulatory action, inducing M2 anti-inflammatory macrophages.
To identify factors responsible for the therapeutic effects of CMs, soluble factors present in SHEDCM were characterized. A total of 79 proteins were identified, some of them known to be involved in neurodegenerative processes, with anti-apoptotic, anti-inflammatory, and axonal elongation properties. In particular, MCP-1 and ED-Siglec-9 may be involved in M2-like macrophage differentiation.
Indeed, depleting these factors from the SHED-CM reduced CM's ability to induce M2-like macrophages and promote functional recovery after SCI. Interestingly, the administration of BMSC-CM induced no or only slight M2-like cell differentiation and did not induce recovery such as SHED-CM [82].
In agreement with the previous study, the treatment with SHED-CM loaded on a collagen hydrogel, used as a delivery system, induced functional recovery in SCI rats, as demonstrated by improvement in scores evaluated through Basso, Beattie, and Bresnahan scoring, inclined plane, cold allodynia, and beam walk tests [83].
The treatment with SHED-CM loaded on a collagen hydrogel also increased the volume of preserved white and gray matter and the total number of neurons and oligodendrocytes in a rat SCI model. On the contrary, lesion volume and lesion length decreased. However, in this study, SHED-CM alone exerted no protection.
The authors suggested that this may be due to the rapid diffusion of SHED-CM, and thus collagen hydrogel may act as an efficient releasing system [84]. A single intravenous injection of SHED-CM also reversed the mechanical allodynia induced by spinal nerve transection, suppressed microglia and astrocytes activation, and decreased the numbers of neurons positive for the neuronal injury marker activating transcription factor 3 (ATF3) and macrophage accumulation.
In particular, the SHED-CM fraction with a molecular weight between 30 and 50 kDa reversed the pain, suggesting that protein components with molecular mass in the range of 30–50 kDa were responsible for the reported neuroprotection [85].
The implantation of a collagen sponge enriched with the serum-free CM from SHED into the nerve gap formed by rat facial nerve transection restored neurological function. On the contrary, CM depleted of MCP-1 and ED-Siglec-9, which are anti-inflammatory M2 macrophage inducers, did not restore neurological function. Notably, MCP-1 and ED-Siglec-9 induced the polarization of M2 macrophages in vitro and in vivo. Thus, the results indicated that MCP-1/ED-Siglec-9 participated in peripheral nerve regeneration inducing M2 macrophage [86].
SHED-CM treatment increased proliferation, migration, and the expression of neuron-, ECM-, and angiogenesis-related genes in Schwann cells. Moreover, SHED-CM stimulated neurite outgrowth of dorsal root ganglia and increased cell viability.
In vivo, axon regeneration and myelination were higher in the SHED-CM group after nerve transection surgery. Motor function improved while muscle atrophy was reduced in the SHED-CM group. Thus, SHEDs may secrete various trophic factors that enhance peripheral nerve regeneration through multiple mechanisms. Specifically, SHED-CM contained NGF, BDNF, NT-3, GDNF, ciliary neurotrophic factor (CNTF), VEGF, and HGF [87].

The administration of SHED-EXOs improved rat motor functional recovery and reduced cortical lesions in rats with traumatic brain injury. SHED-EXOs can exert these effects, reducing neuroinflammation and shifting microglia polarization [88].
SHED-CM induced an improvement in motor disability and reduced infarct volume after permanent MCAO. The SHED-CM treated group showed increased levels of doublecortin, neurofilament H, neuronal nuclei, and rat endothelial cell antigen in the peri-infarct area.
Interestingly, SHED-CM induced the migration and differentiation of endogenous neuronal progenitor cells (NPC), vasculogenesis, and improved ischemic brain injury [89].
Intracerebral administration of SHED-CM in hypoxia-ischemia-injured mice improved neurological function, survival rate, and neuropathological score [90].
CM obtained from SHEDs, and specifically only the fraction of <6 kDa, promoted neurite outgrowth of DRG neurons. Moreover, SHED-CM prevented the decline in sensory nerve conduction velocities in diabetic mice and ameliorated the capillary number-to-muscle fiber ratio and capillary blood flow [91].
In an animal model of superior laryngeal nerve injury, the systemic administration of SHED-CM induced functional recovery, increasing the degree of myelination and promoting axonal regeneration shifting macrophages toward the M2 phenotype [92].
An overview of the studies presented in this paragraph is available in Table 3.

6-OHDA, 6-hydroxy-dopamine; Aβ, amyloid β; BDNF, brain-derived neurotrophic factor; BMP, bone morphogenetic protein; CM, conditioned medium; CNTF, ciliary neurotrophic factor; EAE, experimental autoimmune encephalomyelitis; ECM, extracellular matrix; ED-Siglec-9, ectodomain of sialic-acid-binding Ig-like lectin-9; EXOs, exosomes; EVs, extracellular vesicles; FGF, fibroblast growth factor; GDNF, glial-cell-derived neurotrophic factor; HGF, hepatocyte growth factor; i.c.v., intracerebroventricular; IGFBP, insulin-like growth factor binding protein; LPS, lipopolysaccharide; MCP, Monocyte Chemoattractant Protein; MVs, microvesicles; NF-κB, Nuclear Factor kappa-light-chain-enhancer of activated B cells; NGF, nerve growth factor; NT, neurotrophin; OGD, oxygenglucose deprivation; SHEDs, stem cells from human exfoliated deciduous teeth; MCAO, middle cerebral artery occlusion; SCI, spinal cord injury; TBI, traumatic brain injury; TGF, transforming growth factor; TIMP, tissue inhibitor of metalloproteinase; VEGF, vascular endothelial growth factor; ↑, increase/improvements; ↓, reduction.

For more information:1950477648nn@gmail.com






