Focus On Clinical Management Points Of CKD Combined With T2DM
Jul 07, 2023
Focus on the early management of CKD combined with T2DM, and the new non-steroidal MRA provides a new strategy
CKD can lead to increased medical and economic burdens in patients with T2DM, and with the progression of CKD, medical expenditures increase substantially. However, there is limited information on medical use and medical expenditure in Chinese patients with CKD and T2DM. In the ERA 2023 CKD Oral Presentation session, Peking University First Hospital published a retrospective, regional EHR series cross-sectional study on Ningbo Yinzhou District Health Database (YRHCD), describing the medical expenses of patients with CKD and T2DM in the Chinese regional database and medical use 1.

Click to cistanche deserticola vs tubulosa for kidney disease
The study analyzed 16,521 adult patients (aged ≥18 years) with CKD and T2DM included in the database from January 1, 2017, to December 31, 2020, excluding serum creatinine (SCr) values or not Patients receiving renal replacement therapy, and CKD staging statistics are performed on eligible patients every year. Demographics, comorbidities, and drug prescription data for all included patients, as well as annual medical costs and medical usage by CKD stage, were provided at baseline and between 2017 and 2020.
The results of the study showed that among the 16,521 CKD patients with T2DM included in the study, more than 70% of the patients were 65 years old and above, about 55% were female, and most of the patients were in the CKD G1-G2 stage. More than 90% of patients had hypertension, followed by peripheral vascular disease; renin-angiotensin system inhibitors (RASI) were the most prescribed drugs, followed by metformin. From 2017 to 2020, the age, comorbidity ratio, and drug prescription ratio of patients showed an upward trend, but the proportion of G1-G4 stage patients decreased from 58.2% in 2017 to 48.7% in 2020, compared with 64.4% at baseline.
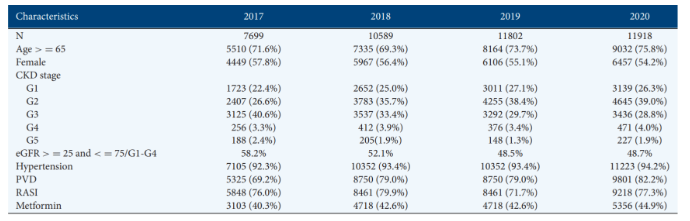
Figure 1: Summary statistics of patients by different CKD stages
From 2017 to 2020, the medical expenses of patients with different CKD stages showed an upward trend; in terms of medical use, the average number of outpatient visits per person per year for each CKD stage exceeded 10.0. Medical costs and medical use increase significantly in patients with advanced CKD. In 2020, the average annual medical expenses per capita of G1-G5 patients were 6,084, 8,675, 14,291, 24,971, and 42,580 yuan (908, 1,294, 2,132, 3,726, and 6,353 U.S. dollars), and the average hospitalization days were 2.2, 3.3, 6.2, 12.6, 16.1 days. Compared with hospitalization, patients with G1 and G2 had more outpatient visits, patients with G4-G5 had significantly higher hospitalization expenses, and hospitalization expenses were the main contributors to the total medical expenses.

Figure 2: Average medical expenses and length of stay for patients with different CKD stages from 2017 to 2020
Findings showed that medical costs and resource use increased modestly over time in the early stages of CKD, but increased significantly from stage G4 onwards. This demonstrates the correlation of the CKD stage with medical costs and medical use. This study further confirmed that early recognition of CKD and timely treatment can reduce the overall medical and economic burden of CKD patients with T2DM.
Professor Cheng Hong pointed out that based on the above research results, early identification and timely treatment of CKD can reduce the overall medical and economic burden of CKD patients with T2DM, which emphasizes the importance of early intervention from the economic perspective. The advancement of the CKD management threshold is inseparable from the management of proteinuria, which requires an effective starting point for early management - UACR, a marker of renal injury, whose increase is closely related to the increased risk of renal injury and CV events 2-4, UACR can also effectively predict the risk of CKD progression, the risk of CV events and death, and the risk of all-cause mortality. The selection of effective drugs for the management of proteinuria in the clinic is conducive to early intervention of the disease and helps patients achieve "early treatment and early benefits".
Finerenone is the world's first new type of non-steroidal MRA approved for the treatment of CKD combined with T2DM. It can precisely target and inhibit the excessive activation of MR, exert anti-inflammatory and anti-fibrosis effects, and thus effectively improve the prognosis of patients with kidney and heart6. The FIDELITY study has confirmed⁷ that based on maximally tolerated RASI treatment and good blood pressure and blood sugar control, finerenone can further significantly reduce the risk of renal composite endpoints by 23%, and significantly reduce the risk of cardiovascular composite endpoints by 14%; treatment After 4 months, UACR was significantly reduced by 32%, and the effect lasted. Finerenone's unique mechanism of action and clear kidney-heart benefits have directly promoted disease management into the "Golden Triangle" era of blood pressure and blood sugar risk factor management, and direct proteinuria management. It is an indispensable standard for CKD combined with T2DM Treatment programs.
High-risk patients need to strengthen blood potassium monitoring, the new non-steroidal MRA hyperkalemia is clinically preventable and controllable
Hyperkalemia (HK) is significantly associated with premature death, adverse clinical outcomes, and a more rapid decline in underlying renal function. However, the epidemiological data for HK in established CKD patients vary greatly, and the incidence of HK in CKD patients with different CKD stages, eGFR, and UACR levels in the real world is also unclear. During the ERA 2023 Congress, a study was presented to assess the incidence and relative frequency of hyperkalemia in patients with chronic kidney disease8.

The study used Optum electronic health record data, including at least two met estimated glomerular filtration rates (eGFR) between January 2009 and December 2020 with an interval of 90 to 365 days. min/1.73 ㎡ and/or UACR value (≥30mg/g), CKD patients aged ≥18 years. Patients with renal failure, hemodialysis, or renal transplantation were excluded at baseline. In this study, HK was defined as (1) an increase in serum potassium (sK⁺) ≥ 5.5 mmol/l in an inpatient or outpatient with an interval of no more than 7 days, or (2) an sK⁺ increase followed by a Combination of drug therapy (eg, intravenous calcium or insulin-glucose, nebulized albuterol, potassium binders) or HK diagnostic codes, no more than 3 days apart. The relative frequency and incidence of HK in the overall CKD enrollment population and specific subgroups were analyzed.
A total of 1,771,900 patients were included in the study, with an average age of 75 years, and women accounted for 57.7%. 83.8% were Caucasian and 9.8% were African American. The most common baseline comorbidities were hypertension (68.5%), hyperlipidemia (55.1%), and T2DM (34.2%). Antihypertensive drugs were used in 69.3% of patients, statins in 45.7%, and arrhythmia drugs in 45.6%. 99.1% of patients had at least one baseline serum potassium measurement, with a mean of 4.3 mmol/L (median, 4.3; IQR, 4.0-4.55).
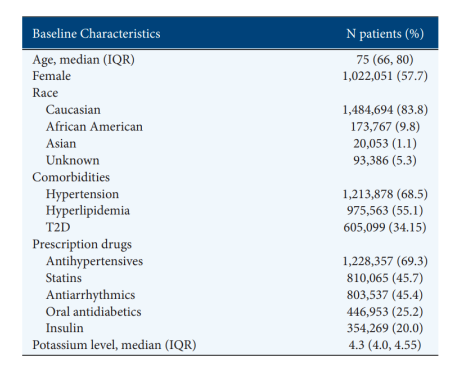
Figure 3: Baseline characteristics of patients in the study
During a mean follow-up of 3.9 years, 220,339 (12.4%) patients experienced at least one episode of hyperkalemia. Among them, 69.3%, 17.5%, and 13.2% of the patients had 1, 2, or ≥3 episodes of hyperkalemia, respectively. The average incidence of hyperkalemia in the overall CKD population was 3.37/100 persons/year (95% CI 3.36-3.38), and it was associated with decreased eGFR and increased UACR values, with a ratio of 1.32. The incidence of hyperkalemia in patients with CKD stages 1, 2, 3, and 4 was 1.32 (95% CI 1.25-1.39), 2.48 (95% CI 2.40-2.55), 3.00 (95% CI 2.99-3.01) and 8.80 (95% CI 8.71-8.88) cases/100 persons/year. The incidence of hyperkalemia was highest in patients with UACR ≥3500 mg/g (13.81; 95% CI 12.96-14.70), independent of eGFR value. In addition, in disease-related subgroups, patients with baseline heart failure (8.70, 95% CI 8.62-8.77), use of MRA (7.66, 95% CI 7.57-7.76), and T2DM (5.43, 95% CI 5.40-5.47) Patients had a significantly higher incidence of hyperkalemia. It can be seen that among CKD patients receiving routine clinical treatment in the United States, hyperkalemia is very common, and 12.4% have at least one episode of hyperkalemia, especially in patients with decreased eGFR and elevated UACR. In addition, hyperkalemia was more prominent in patients with T2DM, heart failure, or the use of sMRA, suggesting that more routine serum potassium monitoring is warranted in patients with the above risk factors.
Professor Cheng Hong said that the management of CKD combined with T2DM should take into account the management of blood potassium while paying attention to the benefits of the kidney and heart. Hyperkalemia was particularly evident in CKD patients with decreased eGFR and elevated UACR and was more prominent in patients with T2DM or sMRA use. Excessive albumin and other proteins can damage the glomerular filtration membrane and tubular cells in the process of glomerular filtration and renal tubular reabsorption, and promote glomerular sclerosis and tubulointerstitial fibrosis9. In patients with diabetic nephropathy, due to impaired renal function and glomerular damage, renal blood flow is greatly reduced, and the excretion capacity of blood potassium is reduced. Therefore, the blood potassium level can increase significantly, and hyperkalemia is more common in the end stage10. In addition, diabetic patients have a relative or absolute lack of insulin, a hypertonic state caused by high blood sugar, etc., which can reduce the entry of potassium ions into cells and lead to hyperkalemia10.
At present, the use of drugs with clear kidney-heart benefits is an effective means to improve the prognosis of patients with kidney-heart. Among them, finerenone has good pharmacological properties, balanced distribution in kidney-heart tissue, short half-life, and no active metabolites, compared with traditional drugs. The risk of hyperkalemia in steroidal MRA is low, and it is preventable and controllable. In the Phase III FIDELITY clinical study, compared with placebo, the maximum difference in the effect of finerenone on serum potassium was 0.19 mmol/L, and the proportion of discontinuation due to hyperkalemia was only 1.7%, and none occurred due to hyperkalemia. The incidence of death, drug withdrawal, or hospitalization caused by potassium hyperemia is low, and it is easy to manage clinically7. It should be noted that CKD, diabetes, heart failure, and hypertension are all risk factors for hyperkalemia. In clinical practice, it is necessary to strengthen the monitoring of high-risk groups and actively intervene. On the premise of actively managing blood potassium, fennel should be avoided as much as possible. Reduction and withdrawal of ketones 10-13.

At present, finerenone has entered the stage of widespread clinical application, and its usage and dosage should be selected according to the patient's eGFR level. For patients with eGFR ≥ 60ml/min/1.73㎡, the standard dose can be directly started at 20mg; for eGFR 25 -For patients with 60ml/min/1.73㎡, start with half dose of 10mg, and after 4 weeks of treatment, if the blood potassium is normal and the eGFR decline is not more than 30% compared with the baseline, adjust the dose to the standard dose of 20mg, orally once a day Medicine can be 14. Phase II ARTS-DN study showed that finerenone significantly reduced UACR in a dose-dependent manner, and did not significantly increase the risk of hyperkalemia in a wide dose range15. Potassium monitoring is still required during finerenone therapy, at 1 month of initial treatment, 4 months, and every 4 months thereafter14. If the patient’s serum potassium>5.5mmol/L, suspend the use of finerenone, wait for the blood potassium level to return to normal after 72 hours, then start finerenone at a half dose of 10mg; if the patient’s serum potassium>5.0mmol/L, it can be adjusted through diet Or additional management of potassium-lowering drugs14, reduction or withdrawal of drugs, and long-term administration of new oral potassium-lowering drugs under blood potassium monitoring as a means of clinical intervention are also double insurance for the safe application of finerenone.
epilogue
CKD combined with T2DM has a heavy disease burden, and early management can reduce the overall medical and economic burden of patients with CKD combined with T2DM. UACR is an effective starting point for early management. The new non-steroidal MRA finerenone can precisely target and inhibit the excessive activation of MR, play an anti-inflammatory and anti-fibrosis role, directly manage proteinuria, and then realize the double protection of kidney and heart, and promote the clinical management of blood pressure and blood sugar risk factors to directly attack protein The new upgrade of urine management strategy makes the clinical management of CKD combined with T2DM enter the "Golden Triangle" era! In addition, the treatment of CKD combined with T2DM should pay attention to the management of serum potassium while taking into account the benefits to the kidneys and heart. The incidence of hyperkalemia with finerenone is lower than that of steroidal MRA, and it is clinically controllable, which provides a strong foundation for safe drug use. Assure. The inclusion of finerenone in medical insurance reduces the economic burden for patients with CKD and T2DM, and has broad prospects for clinical application, which can effectively achieve a win-win situation of clinical value and cost-effectiveness!

How does Cistanche treat kidney disease?
Cistanche is a traditional Chinese herbal medicine used for centuries to treat various health conditions, including kidney disease. It is derived from the dried stems of Cistanche deserticola, a plant native to the deserts of China and Mongolia. The main active components of cistanche are phenylethanoid glycosides, echinacoside, and acteoside, which have been found to have beneficial effects on kidney health.
Kidney disease, also known as renal disease, refers to a condition in which the kidneys are not functioning properly. This can result in a buildup of waste products and toxins in the body, leading to various symptoms and complications. Cistanche may help treat kidney disease through several mechanisms.
Firstly, cistanche has been found to have diuretic properties, meaning it can increase urine production and help eliminate waste products from the body. This can help relieve the burden on the kidneys and prevent the buildup of toxins. By promoting diuresis, cistanche may also help reduce high blood pressure, a common complication of kidney disease.
Moreover, cistanche has been shown to have antioxidant effects. Oxidative stress, caused by an imbalance between the production of free radicals and the body's antioxidant defenses, plays a key role in the progression of kidney disease. Cistanche's antioxidant properties help neutralize free radicals and reduce oxidative stress, thereby protecting the kidneys from damage. The phenylethanoid glycosides found in cistanche have been particularly effective in scavenging free radicals and inhibiting lipid peroxidation.
Additionally, cistanche has been found to have anti-inflammatory effects. Inflammation is another key factor in the development and progression of kidney disease. Cistanche's anti-inflammatory properties help reduce the production of pro-inflammatory cytokines and inhibit the activation of inflammatory pathways, thus alleviating inflammation in the kidneys.
Furthermore, cistanche has been shown to have immunomodulatory effects. In kidney disease, the immune system can be dysregulated, leading to excessive inflammation and tissue damage. Cistanche helps regulate the immune response by modulating the production and activity of immune cells, such as T cells and macrophages. This immune regulation helps reduce inflammation and prevent further damage to the kidneys.
Moreover, cistanche has been found to improve renal function by promoting the regeneration of renal tub with cells. Renal tubular epithelial cells play a crucial role in the filtration and reabsorption of waste products and electrolytes. In kidney disease, these cells can be damaged, leading to impaired renal function. Cistanche's ability to promote the regeneration of these cells helps restore proper renal function and improve overall kidney health.
In addition to these direct effects on the kidneys, cistanche has been found to have beneficial effects on other organs and systems in the body. This holistic approach to health is particularly important in kidney disease, as the condition often affects multiple organs and systems. Cistanche has been shown to have protective effects on the liver, heart, and blood vessels, which are commonly affected by kidney disease. By promoting the health of these organs, cistanche helps improve overall kidney function and prevent further complications.
In conclusion, cistanche is a traditional Chinese herbal medicine used for centuries to treat kidney disease. Its active components have diuretic, antioxidant, anti-inflammatory, immunomodulatory, and regenerative effects, which help improve renal function and protect the kidneys from further damage. Moreover, cistanche has beneficial effects on other organs and systems, making it a holistic approach to treating kidney disease.
Reference:
1. Medical cost and healthcare utilization in adults with chronic kidney disease and type 2 diabetes mellitus in a Chinese Regional Database. ERA 2023.
2. Lambers Heerspink HJ, et al. Clin J Am Soc Nephrol. 2015;10(6):1079-88.
3. Neuen BL, et al. Am J Kidney Dis. 2021;78(3):350-360.e1.
4. Scirica BM, et al. JAMA Cardiol. 2018;3(2):155-163.
5. Hiddo J. Gansevoort CJASN June. 2015, 10 (6) 1079-1088.
6. Barrera-Chimal J, et al. Kidney Int. 2019;96(2):302-319.
7. Agarwal R, et al. Eur Heart J. 2022;43(6):474-484.
8. Incidence and frequencies of hyperkalemia in patients with chronic kidney disease: A retrospective database study from Us clinical care. ERA 2023.
9. Expert Group of Shanghai Nephrology Clinical Quality Control Center. Chinese Journal of Nephrology. 2022;38(5):453-464.
10. Expert Group of Nephrology Branch of Chinese Medical Association. Chinese Journal of Nephrology. 2020;36(10):781-792.
11. Kovesdy CP, et al. Eur Heart J. 2018;39(17):1535-1542.
12. Hougen I, et al. Kidney Int Rep. 2021;6(5):1309-1316.
13. "Chinese Journal of Internal Medicine" Editorial Committee, Expert Group for Clinical Application Consensus of Mineralocorticoid Receptor Antagonists. Chinese Journal of Internal Medicine, 2022,61(9):981-999.
14. Instructions for Keshenda® (Finerenone Tablets).
15. Bakris GL, et al. JAMA. 2015 Sep 1;314(9):884-94.






