HPLC Fingerprinting And Spectrum-antitumor Effect Relationship For Discrimination Between Mylabris Phalerata Pallas And Mylabris Cichorii Linnaeus
Mar 09, 2022
Contact: emily.li@wecistanche.com
Jian-Yong Zhang, Qi-Hong Chen, Xian Pei1, Rong Yan, Can-Can Duan, Yun Liu4, Xiao-Fei Li
1 School of Pharmacy, Zunyi Medical University, Zunyi, China,
2 Key Laboratory of Basic Pharmacology of Ministry of Education and Joint International Research Laboratory of Ethnomedicine of Ministry of Education, Zunyi Medical University, Zunyi, Guizhou, China.
3 School of Basic Medical Sciences, Zunyi Medical University, Zunyi, China, 4Guizhou Provincial College-based Key Lab for Tumor Prevention and Treatment with Distinctive Medicines, Zunyi Medical University, Zunyi, China.

Click here to get more information about Cistanche anti-tumor
Highlights
This study provides an applicable, credible, and more efficient HPLC method for discriminating two Mylabris species. And, the HPLC fingerprinting and spectrum-antitumor effects were integrated and the three important differential markers were found for a new marker of Mylabris.
Abstract
Objective: Evaluation of discrimination between two Mylabris Species based on HPLC fingerprinting and spectrum-antitumor effect relationship. Methods: In this study, a simple and efficient high-performance liquid chromatography (HPLC) method integrating with chemometric analysis and spectrum-antitumor effect relationship was developed for discrimination between two species of Mylabris: Mylabris phalerata Pallas (MP) and Mylabris cichorii Linnaeus (MC). Results: In the fingerprint analysis, 14 characteristic peaks were selected to assess the differences between MP and MC using the similarity and pattern recognition analysis using PCA and OPLS-DA. The HPLC chromatograms of samples from 10 regions of China showed differences between MP and MC, and 7 characteristic chemical markers were found. In the spectrum-antitumor effect relationship analysis, 4 activity markers played a vital role in decreasing the IC50 and might be the antitumor components of Mylabris by grey relational analysis and multivariate linear regression analysis. The chemometric analysis in combination with spectrum-effect relationship results indicated that peaks 2 (cytosine), 4 (unknown), and 14 (unknown) were important differential markers for distinguishing the two species of Mylabris.
Conclusion: The method is applicable, credible, and more efficient to discriminate MP and MC, and will offer a new way for facilitating quality control of insect medicines.
Keywords: HPLC, Fingerprinting, Spectrum-antitumor effect, Mylabris, Discrimination
Background
Mylabris, called ban mao in China, has been used as traditional Chinese medicine (TCM) for treating furuncle, deep-rooted ulcers, an abdominal mass, and is an important antitumor agent. In addition, it is widely used in Europe as folk medicine [1]. The major active constituent of Mylabris is cantharidin, which is an effective anti-tumor compound [2]. Modern pharmacological studies have demonstrated that Mylabris possesses multiple activities, and is highly valued for the treatment of tumors because of its dual anti-cancer properties and the ability to increase the number of leucocytes [3-4]. At present, Mylabris has been widely used in some clinical anti-carcinoma TCM prescriptions in China, such as Aidi injection and Compound ban mao capsule.
According to the Pharmacopoeia of the People’s Republic of China (2015 version), Mylabris is defined as a dried body of Mylabris phalerata Pallas (MP) and Mylabris cichorii Linnaeus (MC), which differ in their botanical characteristics, population dynamics, and ecology [5-6]. Mylabris is also a highly toxic drug. Since the drugs available in China are usually a mixture of MP and MC, it is necessary to identify the chemical and activity differences between the two species.
Since TCM's contain numerous known and unknown ingredients, quantitative profile analysis of their chemical constituents poses a significant challenge. High-performance liquid chromatography (HPLC) fingerprinting plays an important role in the quality control (QC) of TCM because of its overall profile characteristics of most compounds in a complex system. Furthermore, fingerprinting analysis of TCMs has good reproducibility and stability for species authentication and quality evaluation, and is accepted by the World Health Organization, the State Food and Drug Administration of China (SFDA), and the European Medicines Evaluation Agency (EMEA). Recently, a chemometric analysis combining HPLC fingerprinting has been developed for QC and discriminating different complex natural sources [7]. However, it is ineffective to discriminate the differences in TCMs since it cannot reflect the active constituent differences, which is more important for clinical use. So far, spectrum-antitumor effect relationship approaches have been used to successfully identify the bioactive constituents of TCMs [8]. HPLC fingerprinting combined with chemometric analysis and spectrum-antitumor effect relationship will better elucidate the difference of multiple sources of TCMs.
Recently some methods have been reported for determining the content of cantharidin, which can be used to determine the quality of Mylabris [9]. Some chromatographic finger printings were also applied for QC [10-11]. Only one study utilized a method involving a gradient elution of the water-soluble compounds of Mylabris based on HPLC. Meanwhile, only ten samples were used, and no MC samples [12]. To the best of our knowledge, discriminating MP and MC based on chemical composition and activity has not been studied before.
This study aimed to develop a simple and efficient method to discriminate two species of Mylabris using HPLC fingerprinting combined with chemometric analysis and spectrum-antitumor effect relationship, which will provide a basis for the QC of Mylabris. This method could also be applied for identifying the QC marker of other TCMs.

Materials and methods
In total, 20 batches of Mylabris phalerata Pallas (MP) and Mylabris cichorii Linnaeus (MC) were collected from different provinces of China (Table 1) and identified by our prof Xiao-Fei Li. The voucher specimens were deposited in our labs.

The reference standard cytosine, uridine, guanosine, and adenosine were purchased from ChengduPush Biotechnology Co., Ltd. (Chengdu, China). HPLC grade acetonitrile and methanol were purchased from Thermofisher Scientific (Fairlawn, NJ, USA). Watson’s pure water was used throughout the study. Other chemicals and solvents were of analytical grade and obtained from Aladdin Reagent (Shanghai, China), Chengdu Kelong Reagent (Chengdu, China) and Sinopharm Chemical Reagent (Beijing, China)
Apparatus and chromatographic conditions
The HPLC analysis was conducted using an Agilent 1260 Infinity HPLC system at 30 °C on a Phenomenex Synergy Polar-RP80 (4.6 mm ×250 mm, 5 μm) for fingerprinting. Linear gradient elution with eluents A (water/glacial acetic acid, 100:1, v/v) and B (methanol) was used for separation. The gradient program was developed as follows: 0-1 min, 3.0-4.6% B; 1-9 min, 4.6-6.8% B; 9-25 min, 6.8-51.0% B; 25-30 min, 51.0-100%. The flow rate of the mobile phase was 1.0 mL/min. The chromatogram was monitored at 254 nm. After a 15 min equilibration period, 10 μL of samples were used for injection.
Sample preparation
The dried samples were crushed into powder, and 2.5 g of each powdered sample was extracted twice with 50 mL of 75% ethanol by reflux for 1.5 hours each time. The extracted sample was mixed and concentrated under reduced pressure to 20 mL. The mixture was then precipitated with 80 mL water for 24 hours under 4 °C. Subsequently, the extract was centrifuged at 3000 g min-1 for 10 min to separate the supernatant. The supernatant was concentrated under reduced pressure to 25 mL. Finally, the supernatant was filtered through a 0.22 μm Millipore film before HPLC analysis.
Methodology validation
The HPLC method was validated for precision, reproducibility, and stability (0 h, 2 h, 4 h, 8 h, 12 h, 24 h) . The validation was estimated based on the retention time (RT) and peak area (PA). Finally, chemical fingerprints of 20 batches of Mylabris were established to identify the integrated chemical characteristics of multiple compounds. Some chemometric techniques were introduced on the chemical fingerprint for analysis.
Pattern recognition analysis
Differences between two species of the peak areas were evaluated for statistical significance using unpaired Student’s t-test. A p-value less than 0.05 was considered statistically significant. As a classic unsupervised method, principal component analysis (PCA) is widely applied for statistical data analysis. Instead of using many variables, PCA takes a small number of PCs without losing much information, and score plots are then visualized for free separation of observations. In this study, PCA was conducted on the normalized peak areas of each component in the HPLC fingerprints using the SIMCA-P+14.0 software (Umetrics, Umea, Sweden) to find the discrimination between the two species of Mylabris.
For more preferred discrimination between the two Mylabris species, a supervised method of orthogonal partial least squares discriminant analysis (OPLS-DA) was applied to analyze the differences in the normalized peak areas of each component in the HPLC fingerprints. The VIP value is a weighted sum of squares of the OPLS weights, reflecting the relative contribution of each X variable to the model. The variables with VIP > 1, together with S-plot were considered to be influential for the separation of samples in the score plots generated from OPLS-DA analysis. Then, the characteristic chemical markers for discrimination were obtained.

Spectrum-antitumor effect relationship analysis
To explore the anti-tumor effect peak area for Mylabris samples integrated with fingerprint data, and compare the differences between the two species, the correlation between the peak of fingerprint and anti-tumor effects were studied. Two methods combining grey relational analysis and multivariate linear regression analysis (MLRA) were applied by SPSS19.0 (IBM, USA) and GM6.0 soft (Grey Systems Theory Institution, NUAA, China). The anti-tumor test was carried out by our published methods [12], human hepatocellular carcinoma cell line HepG2 cells were maintained in RPMI1640 medium supplemented with 10% fetal bovine serum. The cells were grown in a humidified atmosphere containing 5% CO2 at 37 C. SRB method was used for assessing the proliferation for anti-tumor activities, then the IC50 of HepG2 cells was calculated, finally, the IC50 and normalized peak areas were used for chemometric analysis. Then, the characteristic activity markers of Mylabris were got.
Results and Discussion
Optimization of extract conditions
For sufficient extraction of water-solute compounds from Mylabris, the extraction system was optimized. Ultrasonic and refluxing showed that these compounds could be optimally extracted by refluxing.
Optimization of HPLC method
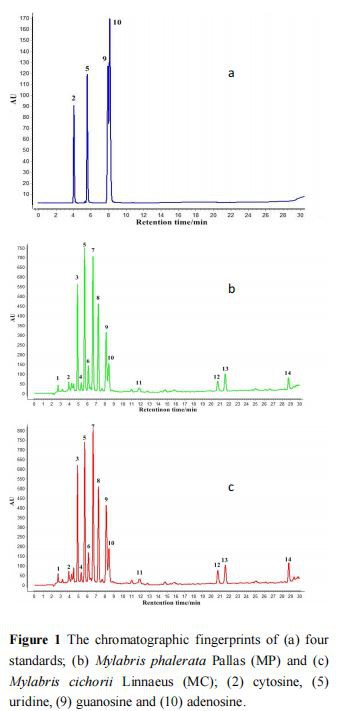
To obtain maximal chromatographic peaks to describe the overall feature of the herb, the composition of the chromatographic column, mobile phase, and detection wavelengths (200, 254, 265, and 278 nm) were investigated. The results showed that Phenimenex Synergy Polar-RP80 column, water (containing 1% glacial acetic acid) and methanol, and 254 nm were the best conditions for Mylabris HPLC analysis (Figure 1), the structure of 4 standard substances is shown in Figure 2.

Method validation
In this study, 14 peaks were well separated and used as “common peaks”. The precision, repeatability, and stability were based on the retention time (RT) and peak area (PA). The RSD values of RT and PA for precision (n=6) did not exceed 0.2 and 4%, respectively; The RSD values of RA and PA for repeatability (n=6) were below 0.2 and 4%, respectively, and the RSD values for stability (0-24 h) were less than 0.4 and 5%, respectively, which indicated that the samples were stable within 24 h. These results illustrated that the quality of the studied samples and HPLC method were stable and well-controlled.
Fingerprint similarity evaluation
The fingerprint similarity analysis was used to evaluate the similarity of HPLC peaks. The similarities of MP and MC were calculated by the reference HPLC fingerprint, respectively. As shown in Table 1, except for two MP samples (from Guizhou2 and Guizhou5) with lower similarities (0.880 and 0.920, respectively), other samples of MP were >0.930. The similarities of MC samples were >0.921 except for MC samples (from Guizhou2 and Guizhou4), whose similarities were 0.888 and 0.887, respectively. These data indicated that the quality of Mylabris within one species was stable. However, the similarities of only three MP samples compared with the reference HPLC fingerprint were > 0.930; and the similarities of only two MC samples compared with the reference HPLC fingerprint were > 0.921. These results showed that the two species of Mylabris were considerable variation in some peak areas.
Discrimination of MP and MC by pattern recognition analysis
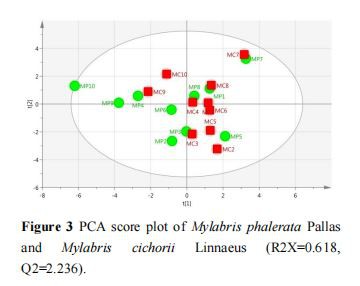
For global analysis of the difference, PCA was used to find the quality variation of the samples from the two species of Mylabris. Figure 3 shows that the two-dimensional PCA model was constructed by the first two PCs, which included approximately 61.8% of the original data. The score plot showed that the MP and MC samples could not be separated by PCA. To understand the differences between MP and MC, an OPLS-DA model was established. As shown in Figure 4, the Mylabris samples could be classified into two groups with R2X=0.502, R2Y=0.492, and Q2=0.0769 as compared to the PCA model. These results showed that the OPLS-DA model was more suitable than the PCA model for distinct separation of the test samples based on their different components. From the S-plot of OPLS-DA, peak markers including peaks 1, 2, 4, 7, 9, 12, and 14 between MP and MC could be found (Figure 5). Based on VIP>1, peaks 1, 2, 4, 7, 9, 12, and 14 might be the most significant variables in discriminating between the two species (Figure 6). The peak group (1, 2, 4, 7, 9, 12, and 14) may play a vital role in distinguishing between MP and MC as characteristic chemical markers.


Spectrum-effect relationship by grey relational analysis
To further evaluate the relationship between the variations of normalized peak area and IC50, grey relational analysis (GRA) was performed. The influence rank by normalized peak area was P13>P14>P3>P2>P12>P4>P9>P1>P10>P7>P6>P8>P5> P11 as shown in Table 2. The results indicated that the top-6 peak including peaks 13, 14, 12, 2, 3, and 4 were the main influencing factors for the antitumor effect based on the standard of Relative Grey correlative degree (RGCE) >0.75.
Spectrum-effect relationship by MLRA
MLRA model is the most common modeling method for deducing the relationship complexity between two or more variables and a response that was built by the following formula
![]()
Where Y is the estimated value and represents the response; Xn is an independent variable, b0 is the intercept, and bn is the regression coefficient for Xn. In this study, MLRA was applied to establish the fingerprint-efficacy relationship between the values of the peak areas in HPLC fingerprints and the IC50 of anti-HepG2, and then find the possible antitumor components. The collineation of data was found by a common MLRA model, which is unsuitable to explore the correlation between Y (IC50) and X (PA). A PCA MLRA model was used to study the fingerprints-effect relationship, and the first six PCs with a cumulative variance contribution rate: 91.068% were selected for analysis. Finally, the following equation was established according to the SPSS output and the PCs: IC50=1.115864 +( 1.87268PA1- 699.722PA2 + 25.7138PA3 - 24.1528PA4 + 7.22878PA5 - 40.2114PA6 + 2.95283PA7- 15.5305PA8 + 13.0297PA9 + 22.5683PA10 - 10.3462PA11 + 123.762PA12 + 31.0428PA13 - 10.702PA14) ×10-6. (R was 0.682, P < 0.05)

Integrating Analysis of Two Mylabris Species
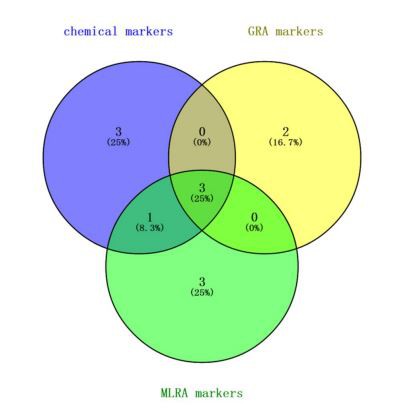
The experiment showed that the HPLC fingerprinting could be used for reflecting the chemical characteristics of Mylabris. The similarity algorithms, PCA, and OPLS-DA were applied to find the difference between MC and MP. The peak group (1, 2, 4, 7, 9, 12, and 14) was defined as characteristic chemical markers.
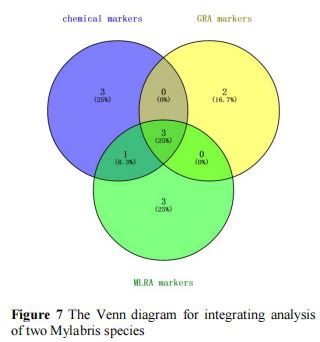
In addition, both GRA and MLRA proved to be a satisfactory methods for further discriminating. Integrating the GRA and MLRA analysis results, peaks 2, 4, and 14 as activity markers of Mylabris should be responsible for the anti-tumor effect, which may be the pharmacodynamic material basis of Mylabris.
The important differential markers were defined for the peak with characteristic chemical and activity marker properties. So, between MC and MP on HPLC fingerprints after spectrum-effect relationship analysis, the peaks 2 (cytosine), 4 (unknown), and 14 (unknown) were important differential markers for the differences between MP and MC as shown in Figure 7.
Conclusion
Conclusively, in the present research, an HPLC method was proposed for the chemical fingerprinting of MP and MC. By combining HPLC fingerprinting, PCA, and spectrum-effect relationship analysis such as GAS and MLRA, the chemical and pharmacological properties of two close species of Mylabris could be discriminated. This indicated that compounds from the peaks 2(cytosine), 4 (unknown), and 14 (unknown) as important differential markers played dominant roles in distinguishing between MP and MC. The structure of peaks 4 and 14 should be identified by other technology. The method of HPLC fingerprinting combined with statistical, chemometric analysis, and spectrum-effect relationship analysis was demonstrated to be efficient in discovering marker components or for promotion of QC of herbal medicines.

References
1 Wang GS. Medical uses of Mylabris in ancient China and recent studies. J Ethnopharmacol 1989, 26(2): 147-162.
2. Lin YS, Lia YH, Peng ZP, et al. Cantharidin content determination and its inhibitory effect on human hepatoma HepG2 cells. Biomed Res-India 2016, 27(2): 533-536.
3. Xu MZ, Lee WS, Kim MJ, et al. Acyl-CoA: cholesterol acyltransferase inhibitory activities of fatty acid amides isolated from Mylabris phalerate Pallas. Bioorg Med Chem Lett 2004, 14(16): 4277-4280.
4. Day RM, Harbord M, Forbes A, et al. Cantharidin blisters: a technique for investigating leukocyte trafficking and cytokine production at sites of inflammation in humans. J Immunol Methods 2001, 257(1-2): 213-220.
5. Mo RY, Sun NX, Peng R. Study on preferred food of adult Mylabris phalerata in different geographical populations. Zhongguo Zhong Yao Za Zhi 2014, 39(22): 4293-4296.
6. Wang XM, Cheng XS, Li XF. Biological Characteristic of Meloidae and Its Artificial Feeding. Guizhou Agricultural Sciences (Chin) 2007, 35(2): 140-142.
7. Yang Y, Zhang J, Jin H, et al. Quantitative Analysis in Combination with Fingerprint Technology and Chemometric Analysis Applied for Evaluating Six Species of Wild Paris Using UHPLC-UV-MS. J Anal Methods Chem 2016: e3182796.
8. Liu Y, Liu Z, Sun G, et al. Monitoring and evaluating the quality consistency of Compound Bismuth Aluminate tablets by a simple quantified ratio fingerprint method combined with simultaneous determination of five compounds and correlated with antioxidant activities. Plos One 2015; 10: e0118223.
9. Mehdinia A, Asiabi M, Jabbari A, et al. Analysis of cantharidin in false blister beetles (Coleoptera: Oedemeridae) by headspace solid-phase microextraction and gas chromatography-mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2011, 879(27): 2897-2901.
10. Li XM, Xiao JS. Study on the GC fingerprints of Mylabris phalerata. China Journal of Hospital Pharmacy (Chin) 2010; 30(2): 116-119.
11. Luo CX, Sun, GX, and Shi XF. Digitized fingerprints of Mylabris by HPCE. Central South Pharmacy (Chin) 2008; 6(2): 230-235.
12. Sun GX, Luo CX, and Wang, Z. Study on the Digitized HPLC fingerprints of Mylabris. Chinese Journal of Pharmaceutical Analysis (Chin) 2008;28(7): 1031-1036.
13. Guo K, Li XF, Yan R, et al. Anti-human hepatocellular carcinoma HepG 2 cells activity of cantharidin derived from Guizhou Province and surrounding regions. Journal of Modern Medicine & Health (Chin) 2016; 32(5): 648-650.