Metabolic Homeostasis Of Amino Acids And Diabetic Kidney Disease-25% Echinacoside
Sep 18, 2023
7. Branched-Chain Amino Acids
Branched-chain amino acids (BCAAs) leucine, isoleucine, and valine are all essential AAs that can only be synthesized by bacteria, plants, and fungi, and also can be acquired from dietary chicken breast, beef, tuna, beans, and cherries. In contrast to the majority of AAs, BCAAs are converted to their branched-chain α-keto acids (BCKAs) in skeletal muscle and then released into the systemic circulation [72]. BCAAs are well known for their crucial role in the promotion of muscle-protein synthesis and modulation of energy metabolism during exercise, which are both mediated through activation of the mTOR signaling pathway [73]. Supplementary BCAAs either act as key nutrition signals or metabolic regulators for glucose homeostasis, immune response, and intestinal development. Also of note is that BCAA supplementation is especially necessary for type 2 diabetes patients with initial renal dysfunction [74]. BCAAs have been reported to counter oxidative stress in the kidneys of diabetic rats and alleviate diabetic kidney injury, such as glomerular hypertrophy, that was mainly mediated via the JNK/TGF-β1/MMP-9 pathway [75] (Figure 6).

CLICK HERE TO GET 25% Echinacoside AND 9% ACTEOSIDE(VERBASCOSIDE) CISTANCHE FOR KIDNEY
In spite of the benefits of BCAAs in metabolic health, studies have also recognized that BCAA homeostasis disruption contributes to pathological conditions of diabetes in animal models of obesity and diabetes [76]. Clinically, strong high-plasma BCAA levels are found in diabetic patients [77]. Under high-glucose circumstances, the capability of muscle protein breakdown is stronger than BCAAs oxidation; combined with serious hypoxia due to inflammation and ER stress, it results in catabolism of BCAAs suppression [73]. Meanwhile, diabetic gut microbial dysbiosis caused by insulin resistance also arrests the degradation metabolism of BCAAs [78,79]. The altered BCAA catabolism predominately results from the altered enzymatic activity of the first two enzymes: branched-chain aminotransferase (BCAT) and branched-chain α-keto acid dehydrogenase (BCKD). In DIO mice, high levels of BCAAs/BCKAs suppressed Akt2 activation and promoted Akt2 ubiquitin–proteasome-dependent degradation through the mTORC2 pathway, depending on the E3 ligase Mul1, finally leading to serious hepatic glucose and lipid metabolic disorder and severe insulin resistance in the liver [80]. As diabetes developed and DKD occurred, plasma BCAA levels began to decline. As long terms of hyperinsulinemia make insulin induce upregulation of the BCKD complex and BCKD dephosphorylation, BCAAs decreased, leading to DKD progression in a 5/6 nephrectomy rat model [81] (Figure 6). Altogether, targeting BCAA metabolism might help prevent not only the development of severe glucose and lipid metabolic disorders but also progressive renal dysfunction, especially in patients with diabetes.
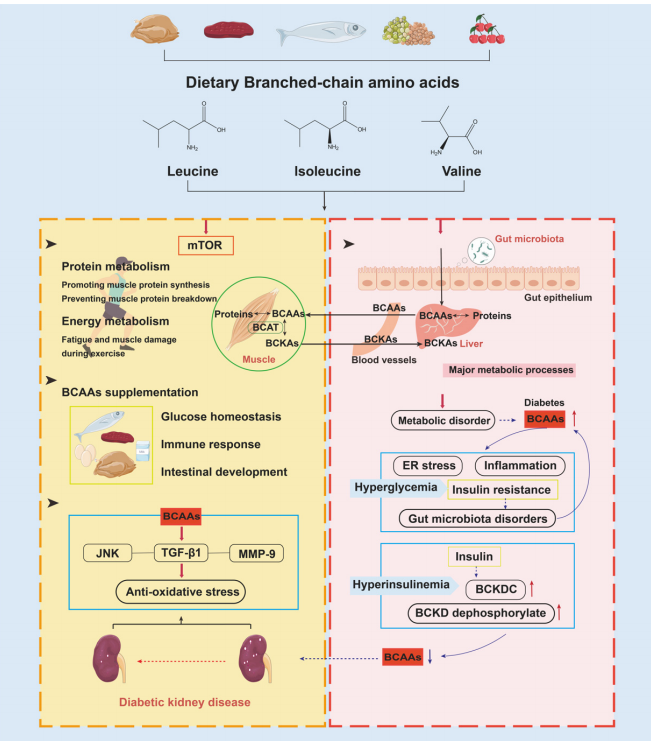
Figure 6. The role of BCAAs and their degradation metabolism in health and disease. The reversible transamination reaction of BCAA catabolism mostly occurs in skeletal muscle. After BCKAs are released back into circulation, most of them are oxidatively decarboxylated in the liver. In health states, BCAA supplementation maintains protein synthesis, energy metabolism, glucose homeostasis, immune response, intestinal development, and renal protection. In diabetic states, excessive BCAA levels, combined with gut microbial dysbiosis, will contribute to hyperglycemia and hyperinsulinemia. Conversely, low BCAA levels will generate renal-function decline. Abbreviations: BCKDC, BCKDC complex; JNK, c-Jun N-terminal kinase; MMP-9, matrix metalloproteinase-9.

8. Discussion and Expected Future Prospects
As is known for essential nutrients, AAs are involved in the maintenance and regulation of metabolic homeostasis. Under normal circumstances, intake of proper amounts of AAs plays a beneficial role in sustaining functions of the body, such as the synthesis of proteins and polypeptide hormones, energy balance, regulation of glucose, and lipid metabolism. This review summarized the benefits of several AAs. In humans, histidine can play an anti-inflammatory, antioxidant, and renal-protective role; tryptophan can regulate glucose metabolism; glutamine can sustain blood glycogen levels; and BCAAs can resist oxidative stress and alleviate renal damage in diabetes. Furthermore, tryptophan showed protective effects on intestinal-barrier injury in vitro, methionine was useful to resistance of oxidative stress in vitro and restoration of fetal kidney dysplasia caused by malnutrition in pregnant rats, and glutamine was reported to inhibit oxidonitrosative stress and protect renal function in STZ-induced rats (Figure 7)

Figure 7. Action mechanisms of altered AA metabolic homeostasis on DKD progression, AAs their metabolites (i.e., IMP, IS, PS, and Hcy) activate renal oxidative stress, ER stress, inflammation fibrosis, and the apoptosis relative signaling pathway, which can promote progressive renal damage under diabetic conditions. Abbreviations: IGR, impaired glucose regulation; IGT, impaired glucose tolerance; IR, insulin resistance.
However, the function of AAs also depends on metabolic homeostasis. Factors such as inappropriate intake, insulin resistance, and intestinal bacteria imbalance will destroy the original metabolic balance of AAs, making them lose their beneficial effects and even causing damage to the body via becoming a pathogenic factor of DKD progression mainlining the basic experimental study stage. For instance, due to diabetic intestinal microbial dysbiosis, abnormal accumulation of intermediate product IMP, derived from histidine not only damages the intestinal barrier and induces inflammation but also causes insulin resistance, destroys glucose tolerance, and may even be a main factor that leads to DKD.

The microbially produced IS derived from tryptophan binds to AHR as a specific ligand and stimulates the NF-κB p65/TGF-β1 signaling pathway to induce renal inflammation and fibrosis. Methionine metabolic disruption not only causes DNA methylation disorder but also leads to abnormal increases of metabolic intermediates Cys and Hcy, causes insulin resistance, induces apoptosis of renal tubular cells and podocytes, and aggravates renal function damage. Excessive glutamine intake will cause renal inflammation and fibrosis via activation of GFPT. Due to intestinal disorder, tyrosine is abnormally converted to PS through the liver metabolism, which can cause thickening of the glomerular basement membrane, destruction of podocytes, vascular inflammation, and fibrosis, leading to proteinuria. Disrupted catabolism of BCAAs blocks the insulin-signaling pathway and causes glucose and lipid metabolism disorders (Figure 7). This is revisable for either positive or adverse effects of AAs metabolic homeostasis upon occurrence and development of DKD. An imbalance in AA metabolism often results in an increase in harmful circulating metabolites, which leads to alterations in cellular signaling pathways and then directly or indirectly induces DKD progression. Although studies should focus on target clarification, consideration of the connections of metabolic homeostasis of different AAs on the spectrum to explain DKD pathogenesis is more conducive to recovery of AAs metabolism and provides more effective nutrition strategies for clinical treatment of DKD.
In the interaction between AA metabolic homeostasis and DKD, it can be seen that the production of many harmful metabolites is greatly affected by disruption of the intestinal barrier and disturbance of the intestinal microbiota [82,83]. Under diabetic conditions, the abundance of intestinal bacteria is significantly reduced, the structure of the intestinal bacteria changes, and the proportion of Bacteroides/Firmicutes phyla is altered [84]. At the same time, a series of omics analyses of intestinal microbial metabolites also showed that many uremic toxins are produced in these conditions. Due to the destruction of intestinal microbiota, the immune response increases, and thus the barrier function of the intestinal tract is gradually damaged, resulting in continuous leakage of toxins from the intestinal tract into blood circulation. Nitrogenous groups of AAs are ultimately metabolized and cleared by the kidneys, which means that if the intestinal barrier and the intestinal microbiota are not effectively recovered in the diabetic stage, renal burden will eventually be increased, causing progressive renal dysfunction. Consequently, this paper reviewed the effects of microbially produced toxins, derived from AAs, on DKD development, and also demonstrated potential cellular signaling pathways that they mediate to cause renal injury. Collectively, it is essential for diabetes patients to pay attention to protecting their intestinal barriers and modulating their intestinal microbiota to help reduce intestinal leakage of harmful metabolites at an early stage and better prevent complications of diabetes and DKD from the perspective of nutrition.
The deficiencies of existing diagnostic biomarkers, such as common blood creatinine, urea nitrogen, serum cystatin C, etc., have become increasingly prominent. Clinically, blood creatinine levels have been found to be highly susceptible to internal and external factors such as protein intake and drug and metabolic differences of patients [85]. Blood urea nitrogen is highly susceptible to nephritis [86]. Current shortages have an impact on diagnosis accuracy and lack of evidence of nutritional status. According to links between AA homeostasis and DKD progress, some potential biomarkers, such as IMP, IS, Hcy, and PS, mentioned above, appear to be used for clinical diagnosis of DKD. The imbalance in plasma concentration or AA proportion in the body can also be used for disease prediction and diagnosis. For example, valine, Cys, N-acetyl aspartate, isoleucine, asparagine, betaine, and L-methionine may be the main factors of type 2 diabetes patient progression to DKD, while decreased plasma histidine and valine levels may be used to distinguish DKD patients from type 2 diabetes patients and healthy controls [8,87]. Particularly, it has been clarified that BCAAs and related metabolites are recognized as potential biomarkers of obesity, insulin resistance, type 2 diabetes, and cardiovascular disease in human cohorts [88,89]. Moreover, it has also been confirmed that modulation of BCAA catabolism is helpful in the improvement of diabetes progression [90], which is further presented by sodium/glucose cotransporter 2 inhibition [91,92]. Changes in AA metabolic homeostasis in diabetes progression are expected to be applied for the prediction and diagnosis of DKD. In fact, integrative analysis of metabolite biomarkers, including valine, leucine, isoleucine, proline, tyrosine, lysine, glutamate, glycine, alanine, palmitic acid, 2-aminoadipic acid serine, and citrulline, has been established for clinical diagnosis and treatment of prediabetes and type 2 diabetes [93,94]. Nowadays, taking advantage of machine learning to analyze large data from genomics, epigenetics, transcriptomics, proteomics, and metabolomics in real-world clinical applications provides a great opportunity to develop multiple biomarkers and avoid information fluctuation of single or isolated biomarkers on overall evaluation efficiency. For clinical prediction and diagnosis of DKD progression, it may be more suitable to carry out a comprehensive evaluation based on the AA homeostasis spectrum and the whole metabolism condition.

Altogether, for patients with nondialysis-dependent CKD, in addition to the advised dietary protein intake of 0.8 g/kg body weight/day recommended by experts [95], the role of AA metabolic homeostasis should be considered comprehensively. For example, it is appropriate for DKD patients to supplement histidine and BCAAs and limit intake of tryptophan, methionine, glutamine, and tyrosine. At the same time, we also need to pay attention to AA proportion, and constantly optimize the protein diet.
Author Contributions: L.L. and Q.C. contributed to drafting this article. Q.C. and T.W. contributed to this discussion. L.L., J.X., Z.Z., D.R., Y.W., D.W., Y.Z., S.Z., Q.C. and T.W. all have revised this manuscript critically for important intellectual content and approved the final version to be published. L.L., Q.C. and T.W. are responsible for the integrity of this work as a whole. All authors have read and agreed to the published version of the manuscript.
Funding: This work was funded by the National Natural Science Foundation of China (No. 81873100) and the Scientific Research Project of Tianjin Educational Committee (No. 2021KJ133).
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
Conflicts of Interest: The authors declare no conflict of interest.
References
1. Kidney Disease: Improving Global Outcomes (KDIGO) Diabetes Work Group. KDIGO 2022 clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int. 2022, 102, S1–S127. [CrossRef]
2. American Diabetes Association. Introduction: Standards of medical care in diabetes-2022. Diabetes Care 2022, 45, S1–S2. [CrossRef] [PubMed]
3. Tu, X.; Luo, N.; Lv, Y.; Wang, B.; Li, Y. Prognostic evaluation model of diabetic nephropathy patients. Ann. Palliat. Med. 2021, 10, 6867–6872. [CrossRef] [PubMed]
4. Vartak, T.; Godson, C.; Brennan, E. Therapeutic potential of pro-resolving mediators in diabetic kidney disease. Adv. Drug Deliv. Rev. 2021, 178, 113965. [CrossRef] [PubMed]
5. Cortinovis, M.; Perico, N.; Ruggenenti, P.; Remuzzi, A.; Remuzzi, G. Glomerular hyperfiltration. Nat. Rev. Nephrol. 2022, 18, 435–451. [CrossRef] [PubMed]
6. Md Dom, Z.; Satake, E.; Skupien, J.; Krolewski, B.; O’Neil, K.; Willency, J.; Dillon, S.; Wilson, J.; Kobayashi, H.; Ihara, K.; et al. Circulating proteins protect against renal decline and progression to end-stage renal disease in patients with diabetes. Sci. Transl. Med. 2021, 13, eabd2699. [CrossRef]
7. Soultoukis, G.; Partridge, L. Dietary protein, metabolism, and aging. Annu. Rev. Biochem. 2016, 85, 5–34. [CrossRef] 8. Zhu, H.; Bai, M.; Xie, X.; Wang, J.; Weng, C.; Dai, H.; Chen, J.; Han, F.; Lin, W. Impaired amino acid metabolism and its correlation with diabetic kidney disease progression in type 2 diabetes mellitus. Nutrients 2022, 14, 3345. [CrossRef]
9. Hu, X.; Guo, F. Amino acid sensing in metabolic homeostasis and health. Endocr. Rev. 2021, 42, 56–76. [CrossRef]
10. Yuskaitis, C.; Modasia, J.; Schrötter, S.; Rossitto, L.; Groff, K.; Morici, C.; Mithal, D.; Chakrabarty, R.; Chandel, N.; Manning, B.; et al. DEPDC5-dependent mTORC1 signaling mechanisms are critical for the anti-seizure effects of acute fasting. Cell Rep. 2022, 40, 111278. [CrossRef]
11. Fang, H.; Stone, K.; Wanders, D.; Forney, L.; Gettys, T. The origins, evolution, and future of dietary methionine restriction. Annu. Rev. Nutr. 2022, 42, 201–226. [CrossRef] [PubMed]
12. Green, C.; Lamming, D.; Fontana, L. Molecular mechanisms of dietary restriction promoting health and longevity. Nat. Rev. Mol. Cell Biol. 2022, 23, 56–73. [CrossRef] [PubMed]
Supportive Service:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
Shop:
https://www.xjcistanche.com/cistanche-shop






