Modeling The Prediction On The Efficacy Of A Homologous Third Dose Of CoronaVac Against SARS-CoV-2 Omicron BA.1, BA.2, BA.2.12.1, And BA.4/5 — China, 2020–2021
Dec 14, 2023
Summary
What is already known about this topic?
Previous studies have reported vaccine efficacy or effectiveness against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Omicron subvariants for several vaccine platforms. However, there is currently little data on estimates of inactivated platform coronavirus disease 2019 (COVID-19) vaccines, especially against the globally dominant subvariant - Omicron BA.5.

cistanche tubulosa-improve immune system
Click here to view Cistanche Enhance Immunity products
【Ask for more】 Email:cindy.xue@wecistanche.com / Whats App: 0086 18599088692 / Wechat: 18599088692
What is added by this report?
The study predicts vaccine efficacy against four Omicron subvariants - Omicron BA.1, BA.2, BA.2.12.1, and BA.4/5 - after vaccination with a homologous third dose of CoronaVac across clinical endpoints and age groups.
What are the implications for public health practice?
The results suggest that CoronaVac-elicited immunity may not provide adequate protection against Omicron subvariants after the homologous third dose, and a heterologous booster and Omicron-specific vaccination may be alternative strategies.
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Omicron variant, a designated variant of concern (VOC) by the World Health Organization (WHO), was first identified in November 2021 in South Africa (1). Three major subvariants, BA.1, BA.2, and BA.3, were identified nearly simultaneously. Soon after its discovery, BA.1 rapidly emerged to become the dominant subvariant worldwide. Gradually, BA.2 and its constituent subvariants, such as BA.2.12.1, overtook BA.1 as the dominant variant worldwide. More recently, two new subvariants, BA.4 and BA.5, were first discovered in South Africa. As of October 1, 2022, the Omicron BA.5 subvariant has been observed in 139 countries across all six WHO regions and has become a globally dominant subvariant due to its substantial growth advantages and faster spread compared to previous subvariants (2). Preliminary data suggest that highly divergent mutations in the spike protein of Omicron may be associated with a high level of humoral immune evasion.

cistanche tubulosa-improve immune system
There are limited efficacy or effectiveness data on the Omicron subvariants for the CoronaVac inactivated vaccine, and the duration of protection after a homologous inactivated vaccine booster dose has not been fully explored. Given the extensive resources and time required to identify and distinguish variants in vaccine trials, statistical models were used to predict CoronaVac-specific efficacy against Omicron BA.1, BA.2, BA.2.12.1, and BA.4/5 across three clinical endpoints - infection, symptomatic coronavirus disease 2019 (COVID-19), and severe COVID-19 - 28 days and 6 months after a homologous third dose.

cistanche tubulosa-improve immune system
Age-specific neutralizing data was extracted from a randomized, double-blind, placebo-controlled, phase 1/2 clinical trial of CoronaVac among healthy adults aged 18 years and older (3) (Supplementary Table S1, available in https://weekly.chinacdc.cn/). Briefly, in the clinical trial, blood samples were obtained from a group of predefined participants who were vaccinated with a homologous third dose of 3 μg of CoronaVac 28 days or 6 months after two primary series doses of CoronaVac. Fold change data on neutralizing antibodies against SARS-CoV-2 Omicron subvariants compared to the prototype strain were extracted from a published study, which separately estimated the reduction fold of geometric mean antibody titers (GMTs) through a live virus neutralization assay (4) (Supplementary Table S2, available in https://weekly. china dc. cn/). Following the models by Khoury et al. (5), the vaccine protection of CoronaVac, for 28 days and 6 months after the homologous third dose with the relationship between neutralizing antibody levels, and vaccine efficacy was predicted. Model details and parameters are summarized in Supplementary Table S3 (available at https://weekly.chinacdc.cn/). All statistical analyses were performed using R software (version 4.0.1, R Core Team, Vienna, Austria). For vaccine-induced protection against infection caused by four Omicron subvariants, the predicted efficacies of CoronaVac were very low even with homologous booster doses, with less than 30% and 10% of the predicted efficacy against Omicron subvariants 28 days and 6 months after a homologous third dose, respectively. Age did not significantly affect the predicted efficacy against virus infection over time in the model results (Figure 1).
For protection from symptomatic illness from Omicron infection, the predicted efficacies against BA.4/5 were 24.2% [95% confidence interval (CI): 22.3%–25.2%] and 26.2% (95% CI: 24.2%–27.3%) for younger adults (18–64 years old) and older adults (≥65 years old) 28 days after the homologous third dose, respectively. Predicted efficacies against BA.4/5 were 7.3% (95% CI: 6.6%–7.7%) for younger adults and 10.4% (95% CI: 9.4%–10.9%) for older adults 6 months after the homologous third dose (Figure 1). Compared with other Omicron subvariants, the predicted efficacies against BA.4/5 were similar to those against BA.2.12.1 but lower than those against BA.1 and BA.2 after the homologous third dose of CoronaVac. For severe COVID-19, the predicted efficacies against Omicron BA.4/5 were 71.6% (95% CI: 69.4%–72.7%) and 36.5% (95% CI: 34.0%–37.7%) 28 days and 6 months after the homologous third dose for young adults, respectively. For older adults, the predicted efficacies were 73.9% (95% CI: 71.8%–74.9%) and 45.9% (95% CI: 43.3%–47.2%) 28 days and 6 months after the homologous third dose, respectively, with no significant difference compared with younger adults (Figure 1).
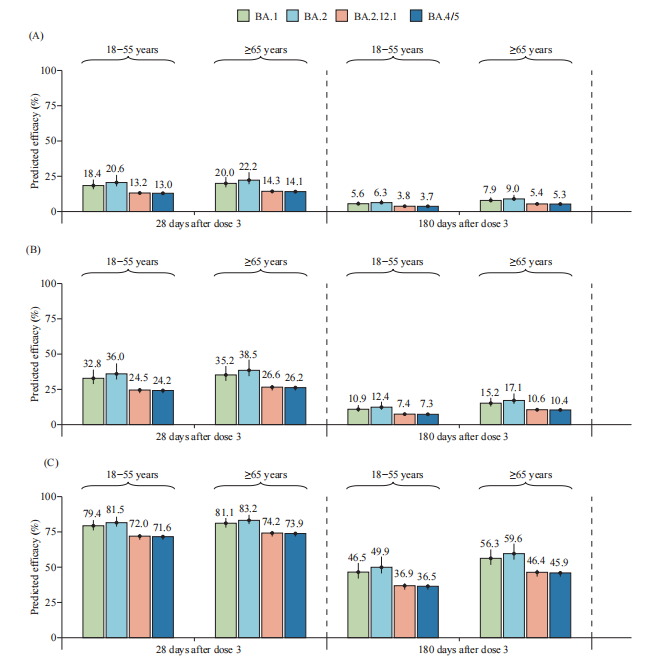
FIGURE 1. Predicted efficacy of CoronaVac against SARS-CoV-2 Omicron subvariants across three clinical endpoints. (A) SARS-CoV-2 infection; (B) Symptomatic COVID-19; (C) Severe COVID-19. Note: The number on the top of the bar represents the predicted efficacy, and the vertical line represents the 95% confidence interval. Abbreviation: COVID-19=coronavirus disease 2019; SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.
DISCUSSION
The study predicted the efficacy against the Omicron BA.1, BA.2, BA.2.12.1, and BA.4/5 subvariants after a homologous third dose of CoronaVac across three clinical endpoints. Vaccine protection against infection and symptomatic illness caused by the Omicron subvariants was found to be not adequate, even after a homologous third dose, and that protection was not maintained for 6 months. Although a homologous booster dose of CoronaVac would increase its efficacy to more than 70% for protection from severe illness within one month after a booster, the predicted efficacy from homologous boosting will wane, gradually declining to less than 50% after 6 months.
Compared to the Wuhan-Hu-1 reference genome, the Omicron variant has more than 30 mutations in the spike protein, nearly half of which are in the receptor-binding domain (6). This degree of genetic change in such an important part of the virus raised serious concerns about strong immune evasion and significant reductions in vaccine efficacy (7). Regarding Omicron subvariants, BA.2.12.1 and BA.4/5 increased evasion of neutralizing antibodies compared with BA.2 and BA.1 (8). In plasma from individuals who received an inactivated vaccine (CoronaVac) or receptor binding domain (RBD) protein (ZF2001) booster six months after two doses of CoronaVac, BA.1 and BA.2 showed no significant difference in resistance to neutralization by plasma. However, BA.2.12.1 showed increased immune evasion capability over BA.2, and BA.4/BA.5 exhibited even greater evasion, with the major contributions made from L452R and F486V mutations (8–9). Such a large degree of immune escape for BA.4/5 may partly verify our predicted results of a lower efficacy, compared to ancestral strains, across all three clinical endpoints.

cistanche tubulosa-improve immune system
A previous study reported that the effectiveness of booster vaccination against a documented Omicron BA.2 infection and severe/critical illness in Shanghai Municipality, China, a city with widespread usage of inactivated vaccines, was 18.0% (95% CI: 17.0%–18.9%) and 92.8% (95% CI: 90.2%–94.7%), respectively. The study also found that a homologous booster dose provided 9 months of >80% protection against more severe outcomes, which was similar to predicted efficacies against Omicron BA.2 in this study (10). Another study reported that the effectiveness of three doses of CoronaVac against mild or moderate disease caused by Omicron BA.2 was 32.4%–51.0%, with a relatively wide confidence interval of 8.3%–60.4%, which covered the range of symptomatic estimates of 32.0%–46.0% were used in this study (11). Besides, the observed effectiveness among individuals with diabetes or kidney disease against different clinical outcomes caused by BA.2 infection was comparable to the predicted results of this study (12–13). Of note, such a comparison should be made cautiously due to differences derived from the study methodology, definition of clinical outcomes, characteristics of study participants, and time points used to calculate effectiveness/efficacy. However, there is currently little data on the effectiveness of Omicron BA.4/5 of inactivated platform COVID-19 vaccines, which limits comparisons between real-world evidence and our predictions. The efficacy 28 days and 6 months after a homologous third dose of CoronaVac was predicted and it was found that the protective effect against Omicron subvariants was not retained for 6 months due to antibody waning, indicating that a homologous booster of inactivated vaccine may not be a suitable regimen for controlling potential large-scale transmission of the Omicron variant. More recent evidence has shown that heterologous booster vaccination induces strong humoral responses and augments neutralization potency against the Omicron variant. Specifically, a third dose of BNT162b2/Pfizer vaccine given to those who received two primary doses of CoronaVac could provide protective levels of antibodies against Omicron (14). A real-world study also revealed that a BNT162b2/Pfizer vaccine booster based on two doses of ChAdOx1 nCoV- 19/AstraZeneca can provide 71.4% protection against symptomatic illness caused by infection with the Omicron variant (15). In addition, replacing vaccine antigens and accelerating the development of Omicron-specific vaccines may be alternative solutions. For example, the first bivalent COVID-19 booster made by Moderna that targets both the original virus and the Omicron BA.1 variant was approved by regulators in the United Kingdom and the Food and Drug Administration (16). Besides, more and more evidence showed that bivalent booster doses are effective in preventing moderate and severe COVID-19 caused by Omicron BA.4/5 infections compared with previous monovalent mRNA vaccine doses only (17–19).
The study was subject to at least two limitations. First, only predictions of efficacy for an inactivated vaccine of CoronaVac were made due to limited time-varying neutralization data after boosting doses for other-platform vaccines. Second, predicted estimates need further verification by real-world evidence as more effective data on inactivated vaccines are revealed.

cistanche tubulosa-improve immune system
In conclusion, the study provided predictions of vaccine efficacy against the 4 SARS-CoV-2 Omicron subvariants 28 days and 6 months after a homologous third dose of CoronaVac across 3 clinical endpoints. The findings suggest that CoronaVac-elicited immunity may not provide adequate protection after a homologous third dose. Heterologous boosting and vaccination with an Omicron-specific booster may be a viable strategy to protect people from Omicron infection.
REFERENCES
1. WHO. Tracking SARS-CoV-2 variants. 2021. https://www.who.int/ activities/tracking-SARS-CoV-2-variants/. [2022-9-15].
2. Gangavarapu K, Latif AA, Mullen JL, Alkuzweny M, Hufbauer E, Tsueng G, et al. Outbreak.info genomic reports: scalable and dynamic surveillance of SARS-CoV-2 variants and mutations. medRxiv 2022. http://dx.doi.org/10.1101/2022.01.27.22269965.
3.Xin QQ, Wu QH, Chen XH, Han BH, Chu K, Song Y, et al. Six-month follow-up of a booster dose of CoronaVac in two single-center phase 2 clinical trials. Nat Commun 2022;13(1):3100. http://dx.doi. org/10.1038/s41467-022-30864-w.
4.Cheng SSM, Mok CKP, Li JKC, Ng SS, Lam BHS, Jeevan T, et al. Plaque-neutralizing antibody to BA.2.12.1, BA.4, and BA.5 in individuals with three doses of BioNTech or CoronaVac vaccines, natural infection, and breakthrough infection. J Clin Virol 2022;156:105273. http://dx.doi.org/10.1016/j.jcv.2022.105273.
5. Khoury DS, Cromer D, Reynaldi A, Schlub TE, Wheatley AK, Juno JA, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat Med 2021;27(7):1205 − 11. http://dx.doi.org/10.1038/S41591-021-01377- 8.
6.Han PC, Li LJ, Liu S, Wang QS, Zhang D, Xu ZP, et al. Receptor binding and complex structures of human ACE2 to spike RBD from omicron and delta SARS-CoV-2. Cell 2022;185(4):630 − 40.e10. http:/ /dx.doi.org/10.1016/j.cell.2022.01.001.
7.Cao YL, Wang J, Jian FC, Xiao TH, Song WL, Yisimayi A, et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature 2022;602(7898):657 − 63. http://dx.doi.org/10. 1038/s41586-021-04385-3.
8.Cao YL, Yisimayi A, Jian FC, Song WL, Xiao TH, Wang L, et al. BA.2.12.1, BA.4, and BA.5 escape antibodies elicited by Omicron infection. Nature 2022;608(7923):593 − 602. http://dx.doi.org/10. 1038/s41586-022-04980-y.
9.Tuekprakhon A, Nutalai R, Dijokaite-Guraliuc A, Zhou DM, Ginn HM, Selvaraj M, et al. Antibody escape of SARS-CoV-2 Omicron BA.4 and BA.5 from vaccine and BA.1 serum. Cell 2022;185(14):2422 − 33.e13. http://dx.doi.org/10.1016/j.cell.2022.06.005.
10.Huang ZY, Xu SF, Liu JC, Wu LL, Qiu J, Wang N, et al. Effectiveness of inactivated and Ad5-nCoV COVID-19 vaccines against SARS-CoV- 2 Omicron BA.2 variant infection, severe illness, and death. BMC Med 2022;20(1):400. http://dx.doi.org/10.1186/s12916-022-02606-8.
11.McMenamin ME, Nealon J, Lin Y, Wong JY, Cheung JK, Lau EHY, et al. Vaccine effectiveness of one, two, and three doses of BNT162b2 and CoronaVac against COVID-19 in Hong Kong: a population-based observational study. Lancet Infect Dis 2022;22(10):1435 − 43. http:// dx.doi.org/10.1016/S1473-3099(22)00345-0.
12.Cheng FWT, Fan M, Wong CKH, Chui CSL, Lai FTT, Li X, et al. The effectiveness and safety of mRNA (BNT162b2) and inactivated (CoronaVac) COVID-19 vaccines among individuals with chronic kidney diseases. Kidney Int 2022;102(4):922 − 5. http://dx.doi.org/10. 1016/j.kint.2022.07.018.
13. Wan EYF, Mok AHY, Yan VKC, Wang BY, Zhang R, Hong SN, et al.Vaccine effectiveness of BNT162b2 and CoronaVac against SARS CoV-2 Omicron BA.2 infection, hospitalization, severe complications, cardiovascular disease and mortality in patients with diabetes mellitus: a case-control study. J Infect 2022;85(5):e140 − 4. http://dx.doi.org/10. 1016/j.jinf.2022.08.008.
14. HKU MED. HKUMed-CU Medicine joint study finds that a third dose of Comirnaty has better protection from the COVID-19 variant Omicron. 2021. https://www.med.hku.hk/en/news/press/20211223-hku-cuhk third-dose-vaccine-omicron. [2022-9-15].
15. Andrews N, Stowe J, Kirsebom F, Toffa S, Rickeard T, Gallagher E, et al. COVID-19 vaccine effectiveness against the omicron (B.1.1.529) variant. N Engl J Med 2022; 386:1532-1546. http://dx.doi.org/10. 1056/NEJMoa2119451.
16. U.S. Food & Drug Administration. Moderna COVID-19 vaccines spikevax, moderna COVID-19 vaccine and moderna COVID-19 vaccine, bivalent. 2022. https://www.fda.gov/emergency-preparedness and-response/coronavirus-disease-2019-covid-19/moderna-covid-19- vaccines. [2022-9-15].
17.Surie D, DeCuir J, Zhu YW, Gaglani M, Ginde AA, Douin DJ, et al. Early estimates of bivalent mRNA vaccine effectiveness in preventing COVID-19-associated hospitalization among immunocompetent adults aged ≥65 Years - IVY network, 18 States, September 8-November 30, 2022. MMWR Morb Mortal Wkly Rep 2022;71(5152):1625 − 30. http://dx.doi.org/10.15585/mmwr.mm715152e2.
18.Tenforde MW, Weber ZA, Natarajan K, Klein NP, Kharbanda AB, Stenehjem E, et al. Early estimates of bivalent mRNA vaccine effectiveness in preventing COVID-19-associated emergency department or urgent care encounters and hospitalizations among immunocompetent adults - VISION network, Nine States, September November 2022. MMWR Morb Mortal Wkly Rep 2022;71 (5152):1616 − 24. http://dx.doi.org/10.15585/mmwr.mm715152e1.
19. Link-Gelles R, Ciesla AA, Fleming-Dutra KE, Smith ZR, Britton A, Wiegand RE, et al. Effectiveness of bivalent mRNA vaccines in preventing symptomatic SARS-CoV-2 infection - increasing community access to testing program, United States, September November 2022. MMWR Morb Mortal Wkly Rep 2022;71(48):1526 − 30. http://dx.doi.org/10.15585/mmwr.mm7148e1.






