Longitudinal Neutralizing And Functional Antibody Responses To Severe Acute Respiratory Syndrome Coronavirus 2 Variants Following Messenger RNA Coronavirus Disease 2019 Vaccination
Dec 13, 2023
In this longitudinal prospective cohort of healthy adults in the United States, we found that coronavirus disease 2019 messenger RNA primary series and booster vaccinations elicited high titers of broadly cross-reactive neutralizing and antibody-dependent cell-mediated cytotoxicity antibodies, which gradually waned over 6 months, particularly against severe acute respiratory syndrome coronavirus 2 variants. These data support the indication for a subsequent booster vaccination.
Keywords.
ADCC; antibody-dependent cell-mediated cytotoxicity; durability; hybrid immunity; Omicron.

cistanche plant-increasing immune system
As of March 2023, the coronavirus 2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has caused more than 600 million cases and 6.8 million deaths worldwide [1]. The pandemic has been characterized by the sequential emergence of SARS-CoV-2 variants with increased transmissibility and the capacity to partially escape vaccine- and natural infection–induced neutralizing antibodies [2]. One such variant was the Omicron strain (B.1.1.529), which was declared a variant of concern (VOC) by the World Health Organization in November 2021 [3].

cistanche benefits for men-strengthen immune system
Click here to view Cistanche Enhance Immunity products
【Ask for more】 Email:cindy.xue@wecistanche.com / Whats App: 0086 18599088692 / Wechat: 18599088692
Since Omicron was first identified, multiple sublineages have emerged including BA.1, BA.2, BA.2.12.1, and BA.4/5 (Supplementary Figure 1), which became the predominantly circulating strain in the United States in June 2022 [4]. While Omicron has been characterized by less severe disease [5–7], it can partially evade neutralizing antibodies due to mutations in the spike protein receptor-binding domain (RBD) [8]. Less is known about the capacity of the Omicron variants to evade antibodies that mediate Fc effector functions. While the role of neutralizing antibodies in the protection against COVID-19 has been well established [9–11], the importance of Fc effector antibodies has more recently been recognized [12, 13]. Antibodies can mediate Fc effector functions, including antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cell-mediated phagocytosis, antibody-dependent neutrophil phagocytosis, and antibody-dependent complement deposition through Fab binding to target antigens and Fc engagement with either Fc receptors (FcRs; present on all innate immune cells) or components of the complement system. Because Fc effector antibodies can bind to antigenic sites distinct from neutralizing antibodies, they may retain functionality and confer protection independently from neutralization. In this study, we therefore measured the longitudinal functional ADCC and neutralizing antibody responses to currently circulating SARS-CoV-2 VOCs following COVID-19 messenger RNA (mRNA) primary and booster vaccinations in individuals with and without hybrid immunity.

cistanche tubulosa-improve immune system
METHODS
Patient Consent Statement
This study was approved by the Institutional Review Board at Emory University, and all participants provided written informed consent.
Study Cohort
Healthy ambulatory adults with or without prior history of symptomatic COVID-19 diagnosed by standard-of-care testing were prospectively enrolled into a specimen collection protocol from 27 April 2020 to 12 April 2021. Standard-of-care mRNA COVID-19 primary vaccine series and monovalent booster vaccines were administered to participants per their provider/location of choice (Table 1). Participants were followed at time points prevaccination, post–dose 2, booster, and post–dose 3 at 1 month, 3 months, and 6 months post-booster. Serum samples were collected at each time point in serum separator tubes, aliquoted, and frozen at −80°C until analysis. Pseudovirus Neutralization Assays
Pseudoviruses with SARS-CoV-2 wild-type (Wuhan-Hu-1), Delta (B.1.617.2), and Omicron (subvariants BA.1, BA.2, BA.2.12.1, and BA.4/5) spike proteins were developed and assays were performed as previously described (Supplementary Methods) [14, 15]. Plates were developed using Britelite Plus luciferase substrate (PerkinElmer), and relative luminescence units (RLUs) were read on a TopCount NXT luminometer. The neutralizing antibody titer to each variant spike protein was expressed as the effective concentration at which 50% of the virus was neutralized.
ADCC Assays The development of tetracycline-inducible green fluorescent protein and luciferase dual-reporter cell lines expressing SARS-CoV-2 variant spike proteins was performed as previously described [14, 15]. Cytotoxicity was determined from a luminescence readout indicating residual live cells. ADCC was calculated according to the following formula: ADCC (%) = [RLU × (no antibody) – RLU (with antibody)] / RLU (no antibody) × 100 and expressed as an endpoint titer.

cistanche tubulosa-improve immune system
RESULTS
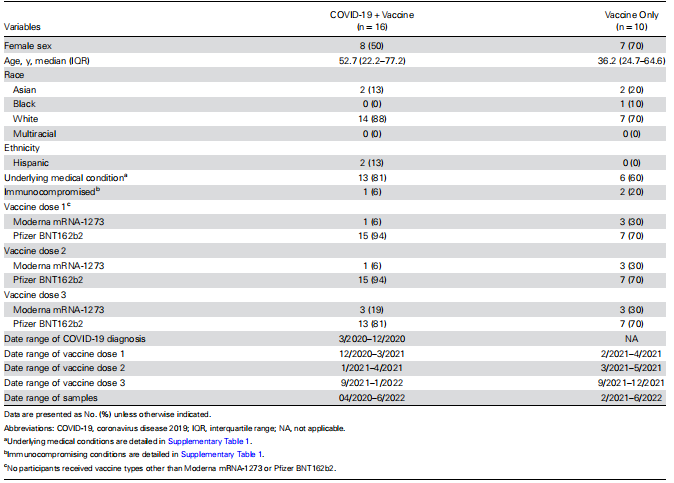
Study Cohort Sixteen participants with and 10 participants without prior history of COVID-19 who subsequently received mRNA COVID-19 vaccination were prospectively enrolled (Table 1 and Supplementary Table 1). The COVID-19 diagnoses occurred from March to December 2020, which largely preceded the emergence of VOCs in the United States. Of those with a prior history of COVID-19, 1 (7%) received mRNA-1273 and 15 (93%) received BNT162b2 for doses 1 and 2, while 3 (19%) received mRNA-1273 and 13 (81%) received BNT162b2 for dose 3. Of those without a prior history of COVID-19, 3 (30%) received mRNA-1273, and 7 (70%) received BNT162b2 for doses 1, 2, and 3.
Pseudovirus Neutralizing Antibody Responses As expected, some participants with a prior history of COVID-19 had detectable neutralizing antibodies to wild-type (9/16 [56%]) and variant strains (4/16 [25%]) at baseline, while the COVID-19–naive group had undetectable baseline neutralizing antibodies (Figure 1A). The 2-dose primary vaccination series elicited neutralizing antibodies in both groups against all 6 strains analyzed, but to lower titers against the variants, particularly among the COVID-19–naive group (Supplementary Figure 2A and Supplementary Table 2). After the primary series, neutralizing titers waned significantly over time in both groups until the administration of the third-dose booster. Following the booster vaccination, neutralizing antibodies were significantly increased in both groups against all 6 strains. The booster dose effectively narrowed the gap between COVID-19–naive and previously infected participants and provided a greater breadth of neutralizing responses to the Delta and Omicron variants. During the 6-month follow-up post booster, neutralizing antibody titers remained detectable in all groups and against all variants. However, titers gradually waned, especially in the COVID–19–naive participants.
Table 1. Baseline Characteristics of the Study Cohort
ADCC Antibodies
Similar to the neutralizing antibodies, ADCC antibodies were detectable at low levels at baseline against all 6 SARS-CoV-2 strains in participants with prior COVID-19, whereas no ADCC antibodies were detected at baseline in COVID-19–naive individuals (Figure 1B). The 2-dose primary vaccination series elicited ADCC antibodies in both groups against all 6 strains analyzed, but to lower levels among the COVID-19–naive group, especially against the Omicron variants (Supplementary Figure 2B and Supplementary Table 3). ADCC antibody titers then waned over time in both groups against all variants prior to administration of a third-dose booster. Following the third dose, ADCC antibodies were significantly increased in both groups against all 6 SARS-CoV-2 variants. During the 6-month follow-up post booster, ADCC antibodies persisted in both groups and against all variants. However, significant declines in antibody titers were observed against the Omicron variants, especially among COVID–19–naive participants. Correlation Between Neutralizing and ADCC Antibodies
Neutralizing and ADCC antibody titers were significantly correlated in both groups of participants post–dose 2, which corresponded to the time of peak antibody titers, with the exception of BA.2.12.1 in the COVID-19–naive group (Supplementary Figure 3). The greatest dissociation between neutralization and ADCC antibodies was observed prior to the booster dose in the COVID-19–naive individuals, which corresponded to the post–vaccine nadir of neutralizing antibodies to the SARS-CoV-2 variants. At that time, multiple participants in the COVID–19–naive vaccinated group had persistent ADCC antibodies in the setting of undetectable neutralizing antibodies.
DISCUSSION
This prospective longitudinal cohort study found that the primary 2-dose series of COVID-19 mRNA vaccination elicited broadly cross-neutralizing and cross-ADCC antibodies to SARS-CoV-2 wild-type, Delta, and Omicron variant strains, which quickly waned. A third-dose booster vaccination effectively narrowed the gap between COVID-19–naive and previously infected participants and provided a greater breadth of neutralizing and ADCC responses to the Delta and Omicron variants. The third dose also improved the durability of serologic responses, as antibodies to all 6 strains remained detectable through 6-month follow-up. Nevertheless, both neutralizing and ADCC antibody titers gradually waned by 6 months, especially to the Omicron variant among COVID–19–naive individuals. These findings support the indication for a subsequent booster vaccination in the setting of waning immunity. Like many other viral infections [16], neutralizing antibodies to SARS-CoV-2 have been found to correlate with protection from symptomatic disease [9–11].
Nevertheless, the correlation of neutralizing antibodies with protection is incomplete. Since Fc effector antibodies have also been found to correlate with protection in preclinical models [12, 13], the combination of neutralizing and Fc effector antibodies may improve the correlation with protection. Fc effector antibodies can bind to unique antigenic sites [17] that are distinct from neutralizing epitopes, facilitating Fc engagement with the FcR on effector cells. This implies that Fc functional antibodies may have the capacity to confer protection against COVID-19 despite the waning of neutralizing antibodies postvaccination and in the setting of the emergence of VOCs [17]. Future studies are needed to determine the effectiveness of Fc functional antibodies in the prevention of COVID-19 caused by currently circulating VOCs.

Benefits of cistanche tubulosa-strengthen immune syste
Limitations of our study include the small cohort size enrolled at a single center. The 2 cohorts were not matched, and differences in baseline characteristics could have contributed to immune responses. Because vaccinations were administered as standard of care at the participants' chosen provider/ location, there was variability in the timing of vaccine doses relative to the timing of sample collection. The timing of enrollments overlapped with the emergence of the Omicron variant, and breakthrough infections may have impacted antibody titers. This study did not assess immune responses following variant-containing mRNA vaccination, which was authorized by the United States Food and Drug Administration and recommended after completion of this study. The study also did not assess antibody responses to other SARS-CoV-2 variants, durability of antibodies beyond 6 months, cellular immune responses, other functional antibody responses, or vaccine effectiveness. In conclusion, COVID-19 mRNA primary series and booster vaccination elicited high titers of broadly cross-neutralizing and cross-ADCC antibodies, which gradually waned over 6 months, particularly against SARS-CoV-2 variants and among COVID-19–naive individuals. These data support the indication for subsequent booster vaccination in the setting of waning immunity and emerging SARS-CoV-2 variants.
![figure 1. Pseudovirus neutralizing and antibody-dependent cell-mediated cytotoxicity (ADCC) activity after coronavirus disease 2019 (COVID-19) vaccination. Pseudovirus neutralizing (effective concentration at which 50% of the virus was neutralized [EC50]) (A) and functional ADCC (B) endpoint antibody titers to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Wuhan Hu-1 (Wu-1), Delta, and Omicron variants (BA.1, BA.2, BA.2.12.1, and BA.4/5) were measured longitudinally following COVID-19 messenger RNA vaccination in participants with or without prior history of COVID-19. The geometric mean titers were calculated for each variant and time point, and statistical comparisons of log-transformed titers were made using Mann-Whitney tests. The dashed lines represent the lower limit of detection for the assay. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant. Heatmaps show pseudovirus neutralizing (EC50) (C) and ADCC (D) endpoint antibody titers against SARS-CoV-2 variants over time. figure 1. Pseudovirus neutralizing and antibody-dependent cell-mediated cytotoxicity (ADCC) activity after coronavirus disease 2019 (COVID-19) vaccination. Pseudovirus neutralizing (effective concentration at which 50% of the virus was neutralized [EC50]) (A) and functional ADCC (B) endpoint antibody titers to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Wuhan Hu-1 (Wu-1), Delta, and Omicron variants (BA.1, BA.2, BA.2.12.1, and BA.4/5) were measured longitudinally following COVID-19 messenger RNA vaccination in participants with or without prior history of COVID-19. The geometric mean titers were calculated for each variant and time point, and statistical comparisons of log-transformed titers were made using Mann-Whitney tests. The dashed lines represent the lower limit of detection for the assay. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant. Heatmaps show pseudovirus neutralizing (EC50) (C) and ADCC (D) endpoint antibody titers against SARS-CoV-2 variants over time.](/Content/uploads/2023842169/202312081048246c8a03cf659548e69e15e518bfa6f062.png)
figure 1. Pseudovirus neutralizing and antibody-dependent cell-mediated cytotoxicity (ADCC) activity after coronavirus disease 2019 (COVID-19) vaccination. Pseudovirus neutralizing (effective concentration at which 50% of the virus was neutralized [EC50]) (A) and functional ADCC (B) endpoint antibody titers to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Wuhan Hu-1 (Wu-1), Delta, and Omicron variants (BA.1, BA.2, BA.2.12.1, and BA.4/5) were measured longitudinally following COVID-19 messenger RNA vaccination in participants with or without prior history of COVID-19. The geometric mean titers were calculated for each variant and time point, and statistical comparisons of log-transformed titers were made using Mann-Whitney tests. The dashed lines represent the lower limit of detection for the assay. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant. Heatmaps show pseudovirus neutralizing (EC50) (C) and ADCC (D) endpoint antibody titers against SARS-CoV-2 variants over time.
References
1. Johns Hopkins University and Medicine. Coronavirus resource center. Available at: https://coronavirus.jhu.edu/map.html. Accessed 30 December 2022.
2. Harvey WT, Carabelli AM, Jackson B, et al. SARS-CoV-2 variants, spike mutations, and immune escape. Nat Rev Microbiol 2021; 19:409–24.
3. World Health Organization. Classification of Omicron (B.1.1.529): SARS-CoV-2 variant of concern. 2021. Available at: https://www.who.int/news/item/26-11- 2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern. Accessed 30 December 2022.
4. Centers for Disease Control and Prevention. COVID data tracker. 2023. Available at: https://covid.cdc.gov/covid-data-tracker/#variant-proportions. Accessed 9 January 2023.
5. Iuliano AD, Brunkard JM, Boehmer TK, et al. Trends in disease severity and health care utilization during the early Omicron variant period compared with previous SARS-CoV-2 high transmission periods-United States, December 2020–January 2022. MMWR Morb Mortal Wkly Rep 2022; 71:146–52.
6. Havers FP, Patel K, Whitaker M, et al. Laboratory-confirmed COVID–19–associated hospitalizations among adults during SARS-CoV-2 Omicron BA.2 variant predominance- COVID-19–Associated Hospitalization Surveillance Network, 14 states, June 20, 2021–May 31, 2022. MMWR Morb Mortal Wkly Rep 2022; 71:1085–91.
7. Adjei S, Hong K, Molinari NM, et al. Mortality risk among patients hospitalized primarily for COVID-19 during the Omicron and Delta variant pandemic periods -United States, April 2020–June 2022. MMWR Morb Mortal Wkly Rep 2022; 71: 1182–9.
8. Cao Y, Wang J, Jian F, et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature 2022; 602:657–63.
9. Feng S, Phillips DJ, White T, et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat Med 2021; 27:2032–40.
10. Gilbert PB, Montefiori DC, McDermott AB, et al. Immune correlates analysis of the mRNA-1273 COVID-19 vaccine efficacy clinical trial. Science 2022; 375: 43–50.
11. Khoury DS, Cromer D, Reynaldi A, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat Med 2021; 27:1205–11.
12. Mercado NB, Zahn R, Wegmann F, et al. Single-shot Ad26 vaccine protects against SARS-CoV-2 in rhesus macaques. Nature 2020; 586:583–8.
13. Gorman MJ, Patel N, Guebre-Xabier M, et al. Fab and Fc contribute to maximal protection against SARS-CoV-2 following the NVX-CoV2373 subunit vaccine with Matrix-M vaccination. Cell Rep Med 2021; 2:100405.
14. Chen X, Rostad CA, Anderson LJ, et al. The development and kinetics of functional antibody-dependent cell-mediated cytotoxicity (ADCC) to SARS-CoV-2 spike protein. Virology 2021; 559:1–9.
15. Rostad CA, Chen X, Sun HY, et al. Functional antibody responses to SARS-CoV-2 variants in children with COVID-19, MIS-C, and after two doses of BNT162b2 vaccination. J Infect Dis 2022; 226:1237–42.
16. Plotkin SA. Correlates of protection induced by vaccination. Clin Vaccine Immunol 2010; 17:1055–65.
17. Kaplonek P, Fischinger S, Cizmeci D, et al. mRNA-1273 vaccine-induced antibodies maintain Fc effector functions across SARS-CoV-2 variants of concern. Immunity 2022; 55:355–365.e4.






