Part 1:Echinacoside Protects Against MPP+-Induced Neuronal Apoptosis Via ROS/ATF3/CHOP Pathway Regulation
Mar 04, 2022
Qing Zhao1 • Xiaoyan Yang2 • Dingfang Cai3,4 • Ling Ye5,6 • Yuqing Hou1 •
Lijun Zhang1 • Jiwei Cheng1 • Yuan Shen1 • Kaizhe Wang5 • Yu Bai1
Contact: joanna.jia@wecistanche.com
Pls click here to Part 2

species (ROS) also contribute to its development [3]. Characteristics similar to PD can be induced by several environmental toxins, including 1-methyl-4-phenyl- 1,2,3,6-tetrahydropyridine (MPTP), cyanide, carbon disulfide, and toluene. MPTP is widely used to induce animal models of PD, and its active metabolite, 1-methyl-4- phenylpyridinium ion (MPP?), is used in cell models of PD [4, 5]. MPP? selectively inhibits the electron transport chain and results in excessive ROS production, thereby inducing neuronal death and a syndrome resembling PD [6]. Recent studies have shown that MPP? induces multiple pathways in an in vitro PD model, including increased GSK3B phosphorylation (pY206) and a-synuclein accumulation [5], upregulation of the heme oxygenase-1/ni- nicotinamide adenine dinucleotide phosphate oxidase/ROS axis [7, 8], increased expression of active transcription factor 3 (ATF3), and growth arrest via DNA damage-inducible 153 (GADD153)/C/EBP-homologous protein (CHOP) [9]. In clinical treatment, suppression of ROS products is one of the most important strategies for the survival of neurons and the treatment of PD [1].
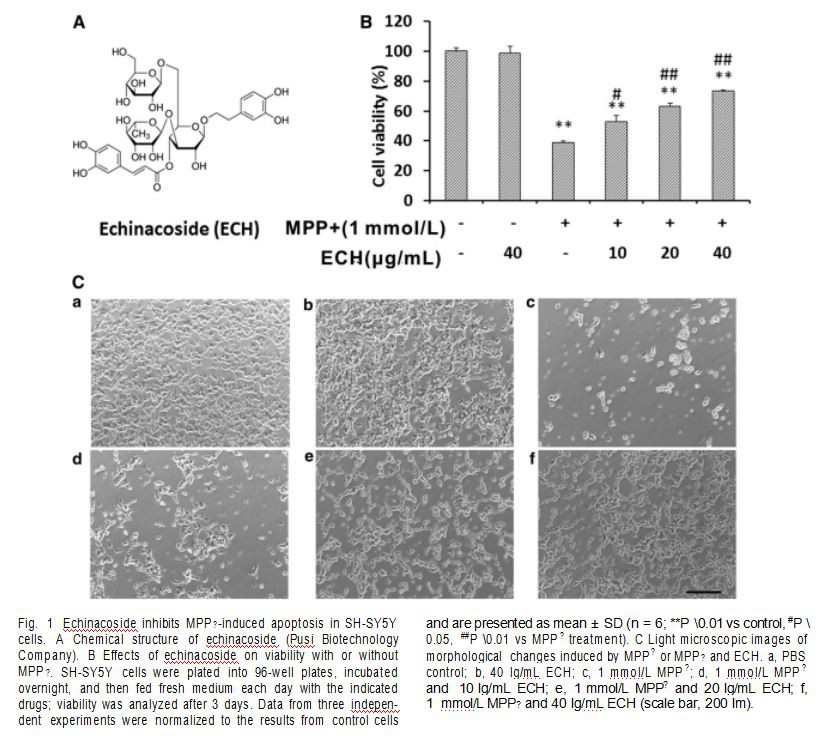
Echinacoside (ECH, Fig. 1A), a natural phenylethanoid found in many medicinal plants, is a principal constituent of the phenylethanoid glycosides isolated from the traditional Chinese herb, Cistanche salsa [10]. Interestingly, recent studies have shown that ECH has protective effects in a mouse model of PD induced by MPTP [10, 11]. ECH also attenuates neuroblastoma cell apoptosis induced by tumor necrosis factor-a or 6-hydroxydopamine in vitro [12, 13]. However, the mechanisms by which ECH promotes the survival of neurons in the MPP?-induced PD cell model remain unclear.
We showed that ECH markedly reduced the ROS products induced by MPP? in SH-SY5Y cells. ECH also inhibited the MPP?-induced expression of ATF3, CHOP, and a-synuclein, as well as suppressing activation of the pro-apoptosis proteins caspase-3 and poly (ADP-ribose) polymerase (PARP).

Cistanche echinacoside has neuroprotective effects
Materials and Methods
Reagents
Dulbecco’s minimal essential medium (DMEM), fetal bovine serum, 0.25% trypsin, 1% penicillin/streptomycin, and TRIzol reagent were from Life Technologies (Grand Island, NY). The chromatin dye bisbenzimide (Hoechst 33342), poly-L-lysine, MPP? iodide and MPTP were from Sigma (St. Louis, MO). The lipid peroxidation malondialdehyde (MDA) assay kit and dichlorofluorescein diacetate (DCFH-DA) were from Beyotime Biotech (Haimen, China). ECH of C98% purity was from Push Biotechnology (Chengdu, China).
Animals and Treatments
Animal maintenance and treatments were as described previously [14]. We used male C57BL/6 mice (Shanghai Laboratory Animal Co., Shanghai, China) weighing 24–26 g at 10 weeks of age. The animals were maintained under standard conditions (12:12 h light: dark cycle, 21 ± 2C, and relative humidity 40%) and allowed access to food and water ad libitum. All procedures were approved by the Animal Ethics Committee of Zhongshan Hospital, Fudan University, and carried out in accordance with the National Institutes of Health Guide for the Care and Use of
Laboratory Animals.
Mice were randomly divided into four groups (10 mice/group): group A, vehicle control group, given an equal volume (500 lL) of normal saline (NS); group B, ECH control group, given 20 mg/kg ECH; group C, MPTP model group, pre-treated with an equal volume of NS; and group D, ECH-treated group, pre-treated with 20 mg/kg ECH. ECH was dissolved in NS and delivered by intra- gastric administration every 24 h for 15 consecutive days. The mouse PD model for groups C and D was generated by five consecutive intraperitoneal injections of 30 mg/kg MPTP every 24 h from day 11 to day 15. Mice were killed by cervical dislocation or perfusion being narcotized by 10% chloralhydrate (3.6 mg/kg) intraperitoneally 24 h after the final treatment.
Perfusion and tissue processing were as described pre- viously [14]. After intracardiac perfusion, brain samples were collected and fixed in 4% paraformaldehyde for 24 h at 4C, embedded in paraffin, and coronal sections encompassing the entire SNc (stereotaxic coordinates anteroposterior -3.64 to -2.92 mm relative to bregma) were cut at 5 lm.
Another 5 animals from each group were killed by cervical dislocation and the ventral midbrain was rapidly lysed in 1% SDS lysis buffer on ice. CHOP and ATF3 protein levels in the lysate were analyzed by western blotting.

Cistanche echinacoside has the effect of invigorating the kidney
Culture of Rat Ventral Midbrain (VM)
Dopaminergic Neurons
Pregnant Wistar rats were purchased from the Shanghai Laboratory Animal Co., (Shanghai, China). DA neurons in the VM were isolated on embryonic day 14. The VM was dissected and processed for establishing primary VM DA neuron cultures as previously described [15, 16]. In brief, tissue pieces were collected in ice-cold Hanks’ balanced salt solution (HBSS) and centrifuged at 1000 rpm at 4C for 5 min. The tissue pellet was incubated in 750 lL of 0.1% trypsin–HBSS for 15 min at 37C with 5% CO2. Fetal calf serum (FCS; 750 lL) was used to inactivate trypsin and tissues were dissociated by gentle trituration
using a sterilized Pasteur pipette. Cell suspensions were centrifuged at 1000 rpm for 4 min at 4C and re-suspended in differentiation medium (Dulbecco’s modified Eagle’s medium/F12, 33 mmol/L D-glucose, 1% L-glutamine, and 1% FCS, supplemented with 2% B27). Cells were seeded on poly-D-lysine (Sigma)-coated 24-well tissue culture plates at 5 9 104 cells/well in 500 lL differentiation medium at 37C with 5% CO2 for 10 days to develop into
mature DA neurons. VM DA neurons were characterized by immunofluorescence with anti-b-tubulin isotype III antibody and tyrosine hydroxylase (TH) antibody (Fig. S1). Cells were treated with 1 mmol/L MPP?, 40 LG/ mL ECH, a combination of MPP? and ECH, or PBS alone as a control. Cell viability was analyzed with CCK-8 reagent after 96 h of treatment.
and are presented as mean ± SD (n = 6; **P \0.01 vs control, #P \ 0.05, ##P \0.01 vs MPP? treatment). C Light microscopic images of morphological changes induced by MPP? or MPP? and ECH. a, PBS control; b, 40 lg/mL ECH; c, 1 mmol/L MPP?; d, 1 mmol/L MPP? and 10 lg/mL ECH; e, 1 mmol/L MPP? and 20 lg/mL ECH; f, 1 mmol/L MPP? and 40 lg/mL ECH (scale bar, 200 lm).

Cistanche echinacoside has the effect of inhibiting apoptosis
SH-SY5Y Cell Culture
SH-SY5Y cells were from the Cell Bank of Shanghai Institute of Cell Biology (Chinese Academy of Sciences, Shanghai, China) and cultured in DMEM supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 U/mL streptomycins. Cells were maintained in a humidified 37C incubator supplied with 5% CO2. MPP? was dis- solved in sterile water and used immediately. ECH was dissolved in PBS. To explore ROS-related gene expression and protein levels, cells were seeded into 6-well plates and cultured for 24 h. The cells were then fed with fresh medium containing MPP?, ECH, a combination of MPP? and ECH, or PBS alone as a control. After 24 h of treatment, the cells were lysed with 1% SDS lysis buffer (1% SDS,25 mmol/L EDTA, 45 mmol/L Tris–HCl, pH 6.5) for western blot analysis, and total RNA was isolated directly with TRIzol reagent.
To analyze the ATF3 and CHOP distribution after treatment, nuclear and cytoplasmic extraction reagent kits (Thermo Fisher, Waltham, MA) were used to extract cytoplasmic and nuclear fractions from drug-treated and control cells.
Cell Viability Assay
SH-SY5Y cells were seeded into 96-well plates and incubated overnight. The cells were fed every 24 h with a fresh complete medium containing different drugs and maintained for 3 days. Then viability was assessed using the CCK8 cell counter kit (Dojindo, Japan). Data were normalized to results from the PBS control and are presented as mean ± SD. To analyze viability in ATF3-knockdown SH-SY5Y (ATF3 shRNA) and control (GFP shRNA) cells, the cells were added to 96-well plates and cultured for 14–16 h. ATF3 shRNA or GFP shRNA plasmids were transfected into SH-SY5Y cells using Lipofectamine 2000 (Life Technologies, Carlsbad, CA). The cells were treated with 1 mmol/L MPP? or an equal volume of PBS after 24 h of transfection and were then incubated for 3 days. Cell numbers were counted every day. Data are presented as mean ± SD.
shRNA Constructs and Oligo Primers
pGV298-ATF3 shRNA (ATF3shRNA) and GFP control shRNA constructs were from GeneChem Co. (Shanghai, China). The ATF3 target sequence was 50-GCAAAGT G!CCGAAACAAGA-30 from previously-described methods [17, 18]. To detect gene expression induced by MPP?, ROS stress-related and PD-associated genes were selected for qRT-PCR, and GAPDH was used as the control. All primers were synthesized by Sangon Biotech Co. (Shanghai, China), and the primer sequences are listed in Table 1.
Real-Time PCR
Total RNA was extracted with TRIzol reagent according to the manufacturer’s instructions. Reverse-transcription and quantitative real-time PCR (qRT-PCR) were performed as previously described. The reverse transcription reaction was performed using the RevertAid First Strand cDNA Synthesis kit (Thermo Scientific, Waltham, MA) according to the manufacturer’s instructions. The SYB Premix Ex Taq II kit (TaKaRa, Dalian, China) was used for qRT-PCR: 20-lL reaction systems were used with ABI (Life Technologies, Carlsbad, CA). The PCR parameters comprised the following steps: 95C for 3 min, 1 cycle; 95C for 5 s and 60C for 40 s, 40 cycles. The final data were nor-
moralized to GAPDH and are presented as a ratio to control. The primers used for amplification are listed in Table 1.
Western Blotting
Western blot analysis was performed as previously described [19]. The primary antibodies were anti-cleaved caspase 3 and p53 (Cell Signaling Technology, Danvers, MA), anti-GAPDH (Proteintech Group, Chicago, IL), anti- GADD153/CHOP (Wanlei Bio, Shenyang, China), and anti-ATF3, anti-PARP, anti-PUMA, and anti-histone 3 (Santa Cruz Biotechnology, Santa Cruz, CA). Horseradish peroxidase-labeled secondary antibodies were from Jack- son Immune Research Laboratories (Western Grove, PA). The PVDF membranes were from EMD Millipore (Billerica, MA) and the ECL substrate was from CWBio (Suzhou, China). Relative protein levels based on gray levels from the image were analyzed with Tanon GIS software (Tanon Biotech, Shanghai, China), and data are presented as the ratio of the target protein to GAPDH.

Cistanche echinacoside has the effect of protecting the liver






