Part 2:Skin-Whitening And Anti-Wrinkle Effects Of Bioactive Compounds Isolated From Peanut Shell Using Ultrasound-Assisted Extraction
Mar 25, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
2.4. Effect of Extraction Conditions on CAI
Collagen is the most abundant protein in mammals and the main structural component of the extracellular matrix with gly-pro-hyp repeating units longer than 1400 amino acids. Collagenase is an enzyme that breaks down peptide bonds of collagen that form skin, bones, tendons, and ligaments. The collagen present in the dermis is decomposed by collagenase, which causes skin wrinkles and reduces skin elasticity; therefore, it is necessary to reduce the activity of collagenase to prevent skin wrinkles [32,33]. The optimization of the UAE condition was performed to maximize CAI of peanut shell extract. A total of 17 runs were needed for optimizing the three individual variables and the experimental data of CAI obtained under experimental sets were 25.2%~92.3% (Table 2). Based on the 17 experimental runs, by applying multiple regression analysis on the experimental data, response and independent variables were related by the following quadratic regression equation in terms of the coded parameters given in Table 3. Then, ANOVA was applied to determine the regression coefficients, statistical significance, and to fit the mathematical models. The mean-square values were calculated by dividing the sum of the squares of each variation source by their degrees of freedom, and a 95% confidence level (α = 0.05) was applied to determine the statistical significance in the analysis of the quadratic model. The ANOVA results confirmed that R2 of the quadratic regression equation was 0.8862 and that the p-value was 0.0134, which is less than the significance level (p < 0.05), thus indicating a good model of fit and statistical significance for predicting CAI values. In the primary term, the X2 and X3 showed significant effects and the interaction effect terms were significant in the X1X2 and X2X3 (p < 0.05). The effect of UAE conditions on CAI production was confirmed to be in the order of: extraction temperature (p = 0.0236) > ethanol concentration (p = 0.0240) > extraction time (p = 0.8505), thus indicating that the effect of extraction temperature and ethanol concentration were significant on CAI.
Figure 1c shows a perturbation plot in which two variables are fixed and it visualized the effect of a single variable on CAI. The effects of all three variables on CAI were shown to be similar, and the three variables showed significant effects and increased and subsequently decreased CAI as each independent variable increased. In our study, 3D surface response curves were developed to visualize the interaction of two independent variables in CAI using quadratic regression equations (Figure 4). When the ethanol concentration was fixed at the center point, the effect of extraction time and temperature on CAI was evaluated in Figure 4A. As the two variables changed simultaneously, CAI increased to
33.4 min and 76.8 C and decreased again after a maximum CAI of 92.8%. As shown in Figure 4B,C, CAI had the highest value at the ethanol concentration of 64.3%, showing a gradual decreasing tendency afterward, which suggests that a binary solvent consisting of 64.3% of ethanol is more suitable as an extraction solvent. This result is consistent with previous research that reported that a binary solvent of water and ethanol showed a higher CAI than water in the extraction of bioactive compounds from Orostachys japonica, which suggests that 50% ethanol would be more advantageous in extracting skin-whitening ingredients [34]. The maximum CAI of the peanut shell extract predicted by the quadratic regression model was 94.5%, which was obtained under conditions of extraction time of 45.1 min, extraction temperature of 93.6 C, and ethanol concentration of 42.3%. The CAI obtained in our study was 94.5%, which is more than twice the effects of 39.4% and 40.3% of CAI values of green tea extracts reported by Oh et al. [35].
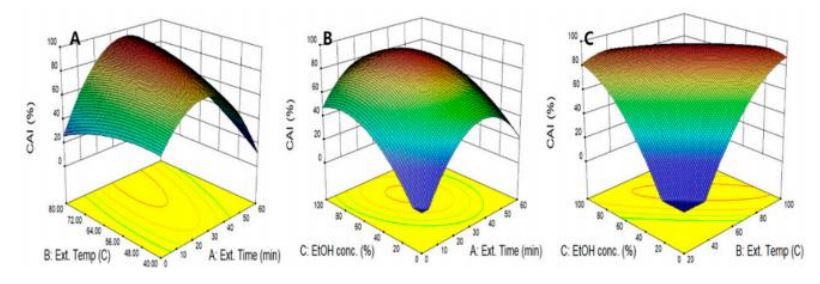
Figure 4. Response surface plots for CAI of peanut shell extracts according to extraction time, extraction temperature, and ethanol concentration. CAI as a function of extraction temperature and extraction time (A), extraction time and ethanol concentration (B), and extraction temperature and ethanol concentration (C).
Cistanche has skin whitening effect
2.5. Optimum Extraction Conditions
Antioxidant, skin-whitening and anti-wrinkle effects are all important functions for cosmetics and it is necessary to derive conditions that can maximize these three functions simultaneously in optimizing UAE conditions. Figure 5shows an optimization procedure that can simultaneously maximize RSA (Y1), TAI (Y2), and CAI (Y3) by overlapping each optimal condition of a contour graph derived through a quadratic regression equation. The ranges of independent variables for the optimization of three variables were limited to the extraction time of 5.0~55.0 min, extraction temperature of 26.0~94.0 。C, and ethanol concentration of 0.0%~99.5% (Table 5). According to individual optimal extraction conditions, optimum UAE conditions were 31.2 min of extraction time, 36.6 。C of extraction temperature, 93.2% of ethanol concentration and, under the above conditions, RSA of 74.9%, TAI of 50.6%, and CAI of 86.8% were predicted. When the predicted RSA, TAI, and CAI values were compared to those obtained from the experiment for validation, the values from the validation test were similar to those of the predicted values, where the values were 78.2%, 52.3%, and 87.7%, respectively.
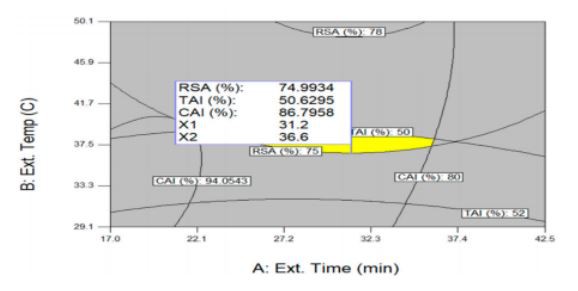
Figure 5. Superimposing contour map for simultaneous optimization of three variables for the maximization of RSA (%), TAI (%), and CAI (%). Ethanol concentration was fixed at the optimum level of 93.2%.
Table 5. Comparison of RSA, TAI, and CAI of peanut shell extract obtained by Soxhlet extraction (SE) and ultrasound-assisted extraction (UAE) at different extraction conditions.

1 Ultrasound-assisted extraction of peanut shell under center point of CCD (Run No. 15, 16, 17); 2 ultrasound-assisted extractions of peanut shell under optimal extraction condition.
2.6. Comparison of SE and UAE
To confirm the extraction effectiveness of UAE, we compare RSA, TAI, and CAI of peanut shell extract produced using UAE and Soxhlet extraction (SE) techniques. When the SE was conducted under general SE conditions using 99.5% ethanol at 70 。C for 4 h of extraction time, RSA, TAI, and CAI were found to be 75.5%, 60.2%, and 74.4%, which were not much different from the results obtained under optimal UAE condition. However, when the SE conditions were set equal to the UAE optimal conditions of 31.2 min and 93.2% ethanol, RSA, TAI, and CAI decreased by 62.0, 28.3, and 45.6%, respectively, compared to UAE under optimal conditions. The advantage of ultrasound in producing useful materials from peanut shells was evaluated as a process suitable for high productivity and industrialization due to low solvent consumption and short extraction time.
2.7. mRNA Expression of MMP-3 and TRP-1
In mammalian melanocytes, melanogenesis and collagen hydrolysis are controlled by TRP and MMP genes, respectively, and TRP-1 and MMP-3 are known as the main genes for the regulation of melanogenesis and collagen hydrolysis; therefore, RT-PCR analysis on whole-cell lysates of B16-F0 cells was performed and the effect of peanut shell extract produced from UAE under optimal conditions (31.2 min, 36.6 C, 93.2%) on mRNA expression of MMP-3 and TRP-1 was studied. As Figure 6 shows, peanut shell extract significantly downregulated the expression of MMP-3 and TRP-1 in B16-F0 cells when the gene expression experiments were performed with a peanut shell extract concentration range of 0~1 mg/mL. The peanut shell extract significantly reduced MMP-3 and TRP-1 expression by 6.1-fold and 8.7-fold, respectively, at 1.0 mg/mL. These results suggest that peanut shell extract inhibits collagen degradation in B16F0 cells by inactivation of MMP-3 to inactivation of MMP-1 and interfere with the cooperation of MMP-9 [36]. Existing studies have shown that treatment with plant extracts inhibited the expression of microphthalmia-associated transcription factor (MITF) by phosphorylating extracellular signal-regulated protein kinase (ERK). Thus, the inhibitory effect of melanin production by peanut shell extract is attributed to the inhibition of tyrosinase activity through expression inhibition of ERK and MITF [37]. Thus, peanut shell extracts reduced mRNA expression levels of TRP-1 and MMP-3, which indicates that peanut shell extract possesses strong inhibitory activities on collagenolysis and melanogenesis making it an excellent cosmetic material with skin-whitening and anti-wrinkle effects.
Figure 6. Effect of peanut shell extract on the expression of TRP-1 and MMP-3 mRNA. B16-F0 cells were treated with various concentrations of peanut shell extract for 24 h. The mRNA levels of MMP-9 and TRP-1 were measured using RT-PCR (a). Band intensities were estimated using Quantity One software and normalized to β-actin (* p < 0.05). All data are expressed as mean o SD of three separate experiments performed in triplicate. N.T = not treated sample, Quantification of TRP-1 mRNA expression (a), quantification of MMP-3 mRNA expression (b).

cistanche phelypaes benefit skin
3. Materials and Methods
3.1. Materials and Reagents
Peanut shells were purchased from Nonghyup mart (Gochang, Jeonbuk, Korea) in March 2019 and the shells were dried at 60 。C using dry oven (FC 49, Lab House, Seoul, Korea) for 24 h until the dry weight remained constant. Dried peanut shells were pulverized using a food processor (Hanil HMF-3800, Seoul, Korea) and then passed through a 600 µm sieve. Ethanol was purchased from Samchun chemical (95.0% v/v, Seoul, Korea). Folin– Ciocalteu reagent, gallic acid (97%), and quercetin were purchased from Merck (Kenilworth, NJ, USA). 2,2-Diphenyl-1-picrylhydrazyl (DPPH), ascorbic acid, and 3,4-dihydroxy-L-phenylalanine (L-DOPA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). All other chemicals used in this experiment were of analytical grade and purchased from Sigma-Aldrich. All the stock solutions were prepared by purified deionized water using a Milli-Q purification system (Millipore, Burlington, VT, USA).
3.2. Ultrasound-Assisted Extraction and Soxhlet Extraction
Powdered peanut shell (1 g) was placed into an extraction vessel, each with 10 mL of solvent and mixed using vortex mixer (VM-10, Daihan Scientific Co., Ltd., Wonju, Korea) for 1 min. Extraction was carried out by circulating water in the ultrasonic extractor (250 W, SD-D250H, Daihan Scientific Co., Ltd., Wonju, Korea) using an external refrigerated bath circulator (CDRC8, Daihan Scientific Co., Ltd., Wonju, Korea) with a digital timer and a temperature controller. The extraction was performed with the ultrasonic device equipped with a digital timer and a temperature controller. Sample were sonicated for various experimental durations and temperatures at working frequency of 40 kHz. Then, the extract was centrifuged at 10,000 rpm for 10 min (236R, Labogene, Seoul, Korea). After centrifugation, the sample volumes were made up to 5 mL and filtered through a 0.2 µm membrane filter prior to analysis. For the Soxhlet extraction, the powdered peanut shell (5 g) were continuously extracted with 100 mL using 99.5% ethanol for 4 h (8 cycles) at a maximum temperature of 70 。C in a Soxhlet apparatus. The ultrasound-assisted extraction technique was shown to be very efficient in the extraction of oil from grape seeds the advantage of the ultrasound, compared to the conventional extraction methods both for oil and polyphenols, was similar since oil/polyphenols yield obtained with a lower solvent consumption and a shorter extraction time.

cistanche tubolosa extract
3.3. Experimental Design
The experimental design was carried out using CCD, a type of RSM to minimize the number of experimental runs and study the interaction between the factors. The Design-Expert® software 8.0 (State-Ease, City, MN, USA) was used for the design of experiments, data analysis, and optimization of extraction conditions for the maximization of the extraction of bioactive compounds having antioxidant, skin-whitening, and anti-wrinkle effects from peanut shell. The experiments were designed according to CCD, the range and center point values of three independent variables presented were based on the results of preliminary experiments (Table 1). The CCD was applied to predict the optimal UAE conditions for the maximization of responses including RSA, TAI, and CAI from peanut shells. As independent variables, the three variables chosen were extraction time (X1), extraction temperature (X2), and ethanol concentration (X3). A total of 17 experimental runs were generated with three replications at the central points to estimate the reproducibility. The quadratic regression model was used to fit the experimental data and applied to predict the response variables, as shown in Equation (1):
Y = β0 + β1X1 + β2X2 + β3X3 + β11X12 + β22X22 + β33X32 + β12X1X2 + β13X1X3 + β23X2X3 (1)
where Y is the predicted response; β0 is the constant (intercept); β1, β2, and β3 are the regression coefficients for the linear effect terms; β11, β22, and β33 are the quadratic effect terms; and β12, β13, and β23 are the interaction effect terms, respectively. A response surface analysis and the ANOVA were employed to determine the regression coefficients and statistical significance of the model terms and to fit the mathematical models of the experimental [38].
3.4. DPPH Radical Scavenging Activity (RSA)
RSA of the peanut shell extract was as described by Pereira-Caro et al. [39]. Solution of 0.01 mM DPPH in methanol (95%) was prepared and 1.25 mL was added to 0.25 mL of diluted extract. RSA was determined to measure absorbance at 517 nm using UV-Vis spectrophotometer (UV1650PC, Shimadzu, Kyoto, Japan) after 20 min of incubation. Blank was prepared using distilled water and the RSA was calculated according to the below.
(Equation (2)):
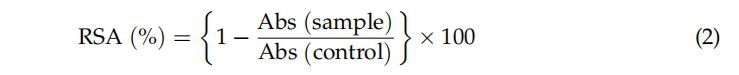
3.5. Tyrosinase Activity Inhibition (TAI)
TAI was performed according to the modified method using L-DOPA as substrate by Jo et al. [40]. Samples were mixed with 200 µL L-DOPA and 200 µL potassium phosphate buffer (pH 6.8) and 200 µL of tyrosinase (125 U/mL) was added in the test tube and incubated at 37。C for 20 min. The sample absorbance was measured at 475 nm using a UV-Vis spectrophotometer and the results were compared to the control. For each concentration, enzyme activity was calculated as a percentage compared to that of the assay using a buffer without any inhibitor, and TAI was calculated based on the following formula. (Equation (3)):

where Abs (control) is the absorbance of buffer + collagenase; Abs (sample) is the absorbance of buffer + collagenase + sample/standard.
3.6. Collagenase Activity Inhibition (CAI)
The measurement of CAI of extracts was carried out by modifying the methods of Wünsch and Heindrich [41]. The substrate, 4-phenylazobezyloxylcarbonyl-Pro-Leu-Gly- Pro-Arg (FALGPA), was dissolved in 10 mL buffer to 1.2 mg/mL and then 125 µL of solution was added and incubated for 60 min at 37 。C. Collagenase was dissolved in the buffer to 0.4 mg/mL, and 75 µL of enzyme solution was added to buffer solution. The enzyme-substrate mixture was incubated in a water bath at 37 。C for 30 min and the reaction was stopped by adding 75 µL of 20% citric acid (w/v). After adding 1.5 mL of ethyl acetate, the ethyl acetate layer was separated and absorbance was measured at 320 nm. The percent of inhibition was calculated according to the following formula.

w
where Abs (control) is the absorbance of buffer + collagenase; Abs (sample) is the absorbance of buffer + collagenase + sample/standard.
3.7. Maintenance and Culturing of Cell Lines
Melanin-producing B16-F0 melanoma cell was obtained from Korea Cell Line Bank (KCLB, Chongno, Seoul, Korea) and was cultured in Dulbecco’s modified Eagle’s medium (DMEM, Sigma-Aldrich, St. Louis, MO, USA) supplemented with fetal bovine serum (FBS, 10%, Welgene, Gyeongsan, Korea) and antibiotic solution of penicillin-streptomycin (Sigma-Aldrich, St. Louis, MO, USA). Trypsin-EDTA (Gibco, Grand Island, NY, USA) was used for the trypsinization of cells. All materials used were of cell culture grade.
3.8. Reverse Transcription Polymerase Chain Reaction (RT-PCR)
RT-PCR was performed to measure changes in MMP-3 and TRP-1 gene expression levels associated with whitening and anti-wrinkle effects, B16-F0 cells were cultured in a 24-well plate treated with the different concentrations of peanut shell extract in serum-free DMEM, and incubated for 24 h. The untreated cell control was maintained under the
same conditions as the tested group during the experiment. RNA isolation from cells was conducted using the AccuPrep® Universal RNA Extraction Kit (Bioneer, Daejeon, Korea). Complementary DNA was synthesized using AmfiRiert Platinum cDNA Synthesis Master Mix (GenDEPOT, Barker, TX, USA). RT-PCR analysis was performed using the CFX 96 touch PCR System (Bio-Rad, Hercules, CA, USA) to determine mRNA levels. The primers used were as follows: MMP-3 sense, 5/-AGTTTGGTGTCGCGGAGCAC-3/ and antisense, 5/- TACATGAGCGCTTCCGGCAC-3/; and TRP-1 sense, 5/-GCTGCAGGAGCCTTCTTTCTC- 3/ and antisense, 5/-AAGACGCTGCACTGCTGGTCT-3/. An appropriate set of primers mentioned above were used to amplify respective genes using the following cycling conditions: 94 。C for 5 min, followed by 25 cycles at 95 C for 5 s, 60 C for 30 s (for MMP-3), and 60 C for 30 s (for TRP-1), and 72 C for 30 s extension. The PCR products were electrophoresed on a 1% agarose gel, stained with ethidium bromide, and visualized by using Gel Doc TM XR+ System and Quantity One software 2.0 (Bio-Rad, Hercules, CA, USA). A housekeeping protein, β-actin, was used as a loading control with the assumption that the expression levels of these proteins remain constant.

cistanche deserticola has whitening and anti-wrinkle effects
4. Conclusions
In this study, the complementary approach was employed for the recovery and use of bioactive substances from agricultural byproducts of peanut shells to develop added-value ingredients with multiple uses. First of all, we attempted to increase the extraction efficiency of bioactive compounds with antioxidant, skin-whitening, and anti-wrinkle effects by optimizing the UAE process. Therefore, this study employed UAE for efficient production of bioactive compounds with skin-whitening and anti-wrinkle effects from peanut shells and applied statistically-based optimization to maximize RSA, TAI, and CAI simultaneously. The UAE conditions were optimized using CCD and it was confirmed that the choice of solvent and concentration should be considered in the extraction of bioactive compounds from peanut shells. By overlapping the response surfaces, curves of three dependent variables, an extraction time of 31.2 min, extraction temperature of 36.6 C, an ethanol concentration of 93.2% were determined to be the optimal conditions of UAE. It has been confirmed that RSA of peanut shell extracts is very high and can be expected to increase in TAI and CAI, which are indicators of skin-whitening and anti-wrinkle effects, respectively. The optimization of UAE conditions confirmed an increase in the production of bioactive substances in peanut shells, and whitening and anti-wrinkle activities of peanut shell extract through tyrosinase and collagenase activity downregulations. Based on this, the effect of peanut shells on the expression levels of MMP and TRP were evaluated to assess whether they have whitening and anti-wrinkle effects at the gene expression level. Whitening and anti-wrinkle effects of peanut shell extracts were confirmed through the downregulation of mRNA expressions as well as the inhibition of protein expressions of MMP-3 and TRP-1. Therefore, peanut shell extract has been shown to be effective in whitening and wrinkle improvement at the protein expression and gene levels. Peanut shell extract, using UAE, has high antioxidant activity and excellent skin-whitening and anti-wrinkle effects, giving peanut shell great potential as a natural cosmetic and food ingredient. Furthermore, it is believed that the production of bioactive compounds using UAE can be applied to the commercialization process for the production of cosmetics, food, and pharmaceutical materials, given the higher production yield and reduced processing costs compared to conventional processes.
Author Contributions: The individual contributions of authors are specified as following: writing- original draft preparation, funding acquisition, supervision, writing, editing, J.W.K.; validation, analysis, methodology, D.H.G.; Investigation, methodology, data curation, editing, J.W.H.; analysis, methodology, review, J.H.K.; All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
Data Availability Statement: No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest: The authors declare no conflict of interest.
Sample Availability: Samples of the compounds are not available from the authors.







