Part 2:Why Cytokinin Plant Hormones Have Neuroprotective Activity in In Vitro Models Of Parkinson’s Disease?
Mar 22, 2022
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791
Pls click here to Part 1
Cistanche herb has a very good neuroprotective effect
2.6. Anti-Apoptotic Effects of Cytokinins Determined by Caspase-3/7 Activity Measurements
As shown by the PI staining assays described above, SAL induced increases in the SH-SY5Y cells’ death rates. As SAL is associated with both apoptosis and necrosis [51] we also investigated the activation of caspase-3 and 7 (casp-3/7) as a specific marker of apoptosis (the execution phase) [52] after exposing the cells to CKs. Caspase-3/7 activities recorded following treatment with each of the test compounds were normalized with respect to those recorded following treatment with 500 uM SAL (set as 100%). As shown in Figure4, the well-known caspase inhibitor Ac-DEVD-CHO (included as a specific, apoptosis-related control) strongly inhibited caspase-3/7 activity at sub-micromolar concentrations (to 36.3 ± 2.66 and 25.2 ± 2.69% of levels in cells treated with SAL alone at 0.05 and 0.5 uM, respectively). Similar levels of inhibition have been previously ob- served [53] in different in vitro SH-SY5Y cell-based models of neurodegeneration. The positive control NAC also significantly reduced caspase-3/7 activity, to 88.7 ± 1.87% and 78.5 ± 2.56% of SAL-induced levels at 100 uM and 1000 uM, respectively. The tested CKs had a wide array of effects on caspase-3/7 activities. Interestingly from the CK ribosides, only cZR had significant effects on the activities at 0.1 uM (reducing them to 83.3 ± 4.33% of SAL-induced levels, respectively), while iPR at 1 uM did not significant reduction of casp-3,7. Finally, K3G decreased caspase-3/7 activity with maximum effect at 10 uM (to 81.7 ± 4.31% of the SAL-induced level). Overall, SAL-induced a 1.6-fold increase in caspase-3/7 activity compared to healthy control cells (CTR, Figure 4), in accordance with results of an earlier study, in which a similar SAL concentration (400 uM) and the same cell line were used [54]. NAC is known to influence caspase-3/7 activity in several models of neuronal death [55–58]. However, the results presented here provide the first demonstration of their caspase-3/7 in an SH-SY5Y cell-based SAL-induced model of neuronal death. The positive control reduced the caspase-3/7 activity induced by 500 uM SAL in a similar manner to CKs, but cZR and K3G were effective even at sub-micromolar or micromolar concentrations. Other studies with stress models associated with PD (proteasome inhibitor MG 132- or H2O2-induced toxicity in SH-SY5Y cells [17] and fibroblasts [15]) and Hunt- ington’s disease (serum starvation model in PC12 cells [19]) have also shown that the CKs K and tZR can have anti-apoptotic (caspase-3) activities [17], and that both K and tZR can have anti-senescence activities [17,19]. Moreover, associations between decreasing casp-3,7 activation and neuroprotective activity were reported for SAL and SAL-related models [54,59,60].

Figure 4. Caspase-3/7 activity in the SAL-induced model of PD. Triplicates in at least four independent days. * P compared with the vehicle with 500 uM SAL, # P compared with the vehicle without 500 uM SAL.
2.7. Identification of Cytokinin Neuroprotective Activity in the Glutamate-induced CellDeath Model
As SH-SY5Y cells do not express all NMDA receptor subunits [61], the most likely mechanism of glutamate (Glu) toxicity is blockage of the cystine/glutamate Xc-antiporter with massive induction of oxidative stress [62]. Previous authors have already demonstrated an association between glutamate-induced cell death and necroptosis in SH-SY5Y cells [63], and the importance of caspase-3 activation in glutamate-induced toxicity to-wards them [62,64]. Several studies have also shown the occurrence of iron-driven cell death (ferroptosis) after glutamate intoxication [62,65,66], and indicated the occurrence of three types of cell death (necroptosis, apoptosis, and ferroptosis) following blockade of the Xc-antiporter system. In order to complete a comprehensive evaluation of the neuroprotective potential of the tested cytokinins, the glutamate-induced cell death assay system was included in an analytical panel with the known neuroprotective iron chelator deferoxamine (DFO) and inhibitor of necroptosis necrostatin-1 (NEC-1) [67] as positive controls. Here, SH-SY5Y cells were differentiated for 48 h, then treated with 160 mM Glu or in co-treatment thereof with various cytokinin concentrations (0.1–10 uM) for 24 h and stained by PI. In this model, the effect of Glu on cell death was 100% PI signal, so the reduction in cell death by the test compounds was evaluated. Glu induced almost a 4-fold increase in cell death compared to healthy SH-SY5Y controls (26.1 ± 0.42%). Screening of positive controls and cytokinins revealed that both positive controls NEC-1 (50 uM,76.6 ± 2.51%) and DFO (10 uM, 84.2 ± 4.54%) decreased the cell death induced by glutamate. As shown in Figure5A, from the panel of cytokinins, only three cytokinins were able to protect neuron-like SH-SY5Y cells in a similar fashion as positive controls. The most active cytokinins were trans-zeatin (tZ) (0.1 uM, 79.5 ± 2.91%; 1 uM, 81.3 ± 2.77%) and cis-zeatin (CZ) (0.1 uM, 82.1 ± 3.29%; 1 uM, 86.2 ± 2.89%) and kinetin (K) (1 uM, 88.0 ± 3.76%; 10 uM, 79.9 ± 3.44%). To confirm their promising activity, an orthogonal assay was used to elucidate in more detail CKs’ biological effects in this model: A lactate dehydrogenase (LDH)-release assay [68], which also showed a dramatic (4-fold) increase in toxicity following Glu treatment. Interestingly, the determination of LDH-release allowed us to differentiate between the activities of CKs and the positive controls (NEC-1 and DFO). The CKs had moderate but significant protective activity (tZ reduced the death rates to 91.9 ± 1.40% at 0.1 uM; cZ reduced them to 92.3 ± 1.15% at 0.1 uM and 91.7% ± 1.56% at 1 uM; K reduced them to 92.2 ± 1.06% at 1 uM). The positive controls NEC-1 and DFO had stronger protective effects but were effective at much higher concentrations (reducing the rates to 76.9 ± 2.94% and 81.6 ± 3.74% at 50 and 10 uM, respectively) (Figure 5B). The LDH assay validated the indications obtained with PI staining and the observed effects of the positive controls were consistent with published data [69,70]. Findings regarding effects of K in the Glu-induced cell death model in HT22 cells [18] and our observations of effects of representative CKs such as tZ, cZ, and K show that CKs are promising candidates for treating neurodegenerative diseases associated with oxidative stress [71]. Finally, tZ and cZ were preferred because they were effective at lower concentrations and were selected, together with K, for further studies of their effects on levels of oxidative stress and caspase-3,7 activation.

cistanche reviews on the effects of neuroprotection
2.8. Effects of Cytokinins on Glu-induced Oxidative Stress in SH-SY5Y cells
Unlike the previous model, Glu can induce oxidative stress in SH-SY5Y cells by different pathways. One is based on blockage of the Cystine/glutamate (Xc-) antiporter, which leads to glutathione (GSH) depletion and negative effects on superoxide dismutase (SOD) activity [62]. Glu-mediated cell death also putatively involves Rac-NADPH-oxidase-driven ROS (particularly superoxide radical) formation in SH-SY5Y cells [72]. These findings indicate that the primary cause of cell death in the Glu-induced model is OS. Within the model, neuron-like cells were co-treated with Glu and tested compounds, stained by DHE, and observed by fluorescence microscopy. As can be seen in Figure 6A, a dramatic rise in red DHE fluorescence was observed in Glu-treated cells, which was ameliorated by the positive control substances and tested CKs. Based on the microscopy observation, the quantification of superoxide levels was performed in Glu-induced cells in a similar manner, by the DHE assay, to those in SAL-induced cells. The results were normalized with respect to levels induced by Glu alone (set as 100%), which caused a dramatic, almost 3-fold increase in superoxide levels within just 4 h in neuron-like SH-SY5Y cells relative to levels in healthy control cells (CTR: 33.79 ± 1.45%). As shown in Figure 6B, CKs such as tZ, cZ, and K significantly reduced the superoxide level in Glu-intoxicated cells: 0.1 uM tZ, 0.1 uM cZ, 1 uM, and 10 uM K reduced it to 81.0 ± 4.03, 80.6 ± 3.89, 81.8 ± 3.39 and 83.8 ± 2.32% of levels in cells treated with Glu alone. These levels are comparable to those obtained with the necroptosis inhibitor NEC-1 (80.5 ± 3.19% at 5 uM and 80.4 ± 2.70% at 50 uM) and iron-chelator DFO (80.2 ± 3.80% at 10 uM). The results obtained with the Glu-induced cell death model, particularly the effects of positive controls, are consistent with previous reports [70,73]. Previous evidence that CKs have OS-reducing effects in Glu-induced or similar models is limited to a demonstration that K is a potent OS-reducing agent (at approximately 23 uM) in the HT22 cell-based Glu-induced cell death model [18]. We found that K had an OS-reducing effect at lower concentrations (1–10 uM) in our SH- SY5Y cells. In addition, beneficial long-term effects of tZ on human fibroblasts, including hydrogen peroxide-decomposing activity, have been observed [15], suggesting that tZ and cZ may have indirect OS-reducing effects in Glu-induced cell death models. Additionally, other reports pointed out the associations between CKs-mediated OS-reducing effect and protective activity [22,74–76]. Taken together, tZ, cZ, and K showed surprisingly strong OS-reducing effects, comparable to those of positive controls, despite differing strengths in overall neuroprotective effect.
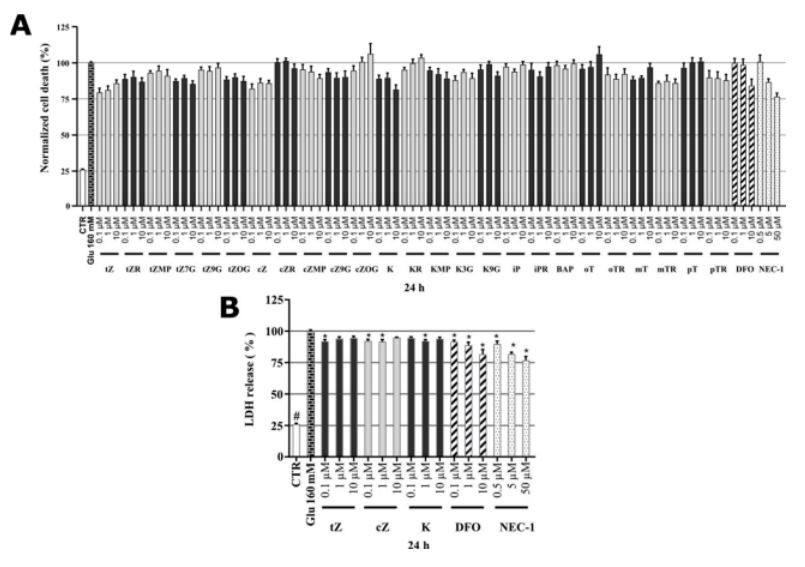
Figure 5. (A) Death rate of neuron-like SH-SY5Y cells in glutamate (Glu)-induced model of cell death; (B) Glu-induced toxicity (LDH-release) of neuron-like SH-SY5Y cells. Triplicates in at least three independent days.* P compared with the vehicle with 160 uM Glu, # P compared with the vehicle without 160 uM Glu.

Figure 6. (A) Glu-induced oxidative stress (OS) and OS-reducing activity of indicated compounds in human neuron-like SH-SY5Y cells visualized by fluorescence microscopy following dihydroethidium (DHE) labelling. Bars = 50 um. Images show neuron-like SH-SY5Y cells treated by DMSO (Control), 160 mM glutamate (Glu) alone, and together with: 10 uM deferoxamine (+DFO), 50 uM necrostatin-1 (+NEC-1), 0.1 uM tZ (+tZ) and 0.1uM cZ (+cZ) for 4 h, then stained by DHE. (B) Glu-induced superoxide radical formation in neuron-like SH-SY5Y cells after 4 h. The graph displays the quantification of DHE stained cells using the Infinite M200 Pro microplate reader (Tecan, Austria). Triplicates in five independent days.
* P compared with the vehicle with 160 uM Glu, # P compared with the vehicle without 160 uM Glu.
2.9. Effects of Cytokinins on Caspase-3/7 Activity in the Glu-induced Cell Death Model
Glu has similar toxic effects on SH-SY5Y cells to previously reported effects on HT22 cells associated with Xc-antiporter blockage. Thus, non-apoptotic cell death mechanisms (ferroptosis, necroptosis, etc.) are involved in both cases [62] although expression of caspase-3 and to some degree, the NMDA subunit of NR1 may be more strongly occurred in SH-SY5Y cells [61]. Hence, final stages of cell death in the two cell lines may differ. A contributory role of caspase-3 activity has been observed in many studies in the Glu- induced model of cell death of SH-SY5Y cells [62,77–79]. In this assay, neuron-like SH-SY5Y cells were treated here with 160 mM Glu, which leads to elevation of caspase-3/7 activity. In efforts to elucidate the role of apoptosis in Glu-induced death of SH-SY5Y cells, a specific caspase-3/7 inhibitor, Ac-DEVD-CHO, was applied and found to have a strong inhibitory effect: At 50 nM it reduced caspase-3,7 activity to 23.70 ± 1.01% of the level in cells treated
with Glu alone and at 0.5 uM it almost completely blocked the activity (reducing it to a normalized level of just 7.22 ± 1.05%; comparable to the level observed in healthy cells:
7.8 ± 0.39%), as shown in Figure7. Application of positive controls resulted in a gradual dose-dependent decrease in caspase-3,7 activity. Interestingly, NEC-1 had a slight effect with maximum effect at 50 uM and DFO had more robust inhibitory activity at 10 uM (reducing the activity to 80.5 ± 1.85 and 75.8 ± 3.00%, respectively). On the other hand, only slight, but significant effects on casp-3,7 were achieved by tZ at 1 uM (89.5 ± 2.50%), cZ at 0.1 uM (87.4 ± 1.62%) and K at 1 uM (91.0 ± 2.06%). Taken together, the results show that both positive controls had stronger caspase-3/7 activity-decreasing activity than the tested CKs, but the CKs had effects at much lower concentrations. Overall, the results indicate that modulation of caspase-3/7 activity plays a key role in the potent neuroprotective activity of agents such as DFO [58] in the Glu-induced model of cell death [79,80]. However, as shown in this section, NEC-1 also had neuroprotective activity, suggesting that other types of cell death might be involved in Glu-induced death of neuron-like SH-SY5Y cells [63,67,81]. Indications that caspase-independent mechanisms are involved in Glu-induced cell death have also been obtained in analyses of effects of NEC-1 [70], DFO [69], and K [18] on HT22 cells.

Figure 7. Caspase-3/7 activity in Glu-model of oxidative damage of neuron-like SH-SY5Y cells. Triplicates in at least four independent days. * P compared with the vehicle with 160 uM Glu, # P compared with the vehicle without 160 uM Glu.
3. Conclusions
In summary, our study revealed that CKs and their metabolites have neuroprotective activities in the SAL-induced model of Parkinson’s disease and Glu-induced oxidative damage in human differentiated SH-SY5Y neuronal cells. K3G, cZR, and iPR were found to have biologically significant neuroprotective activities. Moreover, the active CKs were effective at lower (sub-micromolar and micromolar) concentrations than the positive control substance NAC. K3G, cZR, and iPR also had positive effects on viability, cytotoxicity, oxidative stress, and caspase-3,7 activity (except iPR). The orthogonal demonstrations strongly indicate that anti-oxidative stress activity plays a key role in CKs’ protective effects in the SAL-induced cell death model. Only three metabolites in the tested panel of CKs (tZ, cZ, and K) protected neuron-like SH-SY5Y cells in the Glu-induced model of oxidative damage in a similar fashion to the iron chelator NEC1 and necroptosis inhibitor DFO. To confirm their promising activity, we tested their effects in orthogonal lactate dehydrogenase (LDH)-release assay. The CKs had moderate but significant protective activity. This was weaker than the corresponding activities of the control substances, but despite the differences in the modulation of neuronal health, all three tested CKs stimulated potent reductions in superoxide radical formation, similarly to the positive control substances. The CKs also had comparable effects on Glu-induced oxidative stress in SH-SY5Y cells to those of NEC1 and DFO, but these compounds had stronger inhibitory effects on caspase-3/7 activity than the CKs. As Glu-induced cell death does not rely on a caspase-dependent pathway, as shown in studies of effects of DFO and NEC-1 on HT-22 cells [70], CKs can apparently modulate both caspase-dependent and -independent cell death by reducing oxidative stress [18,82]. In ongoing studies, the detailed mechanism of action of CKs and/or their mitochondrial effects on neuronal or astrocyte cells will be investigated.

cistanche experience for the PD and memory
4. Materials and Methods
4.1. Chemicals and Reagents
Cytokinin standards were obtained from OlChemIm (Olomouc, Czech Republic). Calcein AM (1 mg/mL solution), and LDH release kit, and a Neurite outgrowth kit were purchased from ThermoFisher. Propidium iodide, dihydroethidium, components of caspase- 3/7 assay buffer, DMEM/F12 1:1 medium, fetal bovine serum, trypsin, ATRA, salsolinol hydrobromide, glutamate monosodium salt, deferoxamine, N-acetylcysteine, Ac-DEVD-CHO, and DMSO for cell cultures were purchased from Sigma Aldrich (Merck). Ac-DEVD-AMC substrate was supplied by Enzo Life Science.
4.2. ORAC Radical Scavenging Activity Assays
Compounds’ ability to scavenge free radicals in vitro was determined by the Oxygen Radical Absorbance Capacity (ORAC) assay. Briefly, fluorescein (100 uL, 500 mM), and 25 uL of the tested compound solution were added to a 96-well microplate pre-incubated at 37oC. Next, 25 uL of 250 mM 2,2<-azobis(2-amidino-propane)dihydrochloride (AAPH) was quickly added, the microplate was shaken for 5 s and red fluorescence (with 485 and 510 nm excitation and emission wavelengths, respectively) was measured every 3 min over 90 min using an Infinite 200 microplate reader (TECAN, Männedorf, Switzerland). NAUC (Net Area Under Curve) values were used to express antioxidant activities relative to that of Trolox (a synthetic hydrophilic analog of α-tocopherol, vitamin E). Substances with ORAC values greater than zero are deemed to actively trap free radicals.
4.3. SH-SY5Y Cell Culture
The human neuroblastoma cell line SH-SY5Y purchased from ATCC (American Type Culture Collection, Manassas, VA, USA) was grown in Dulbecco’s modified Ea- gle’s Medium and Ham’s F12 Nutrient Mixture (DMEM: F-12, 1:1), supplemented with 10% fetal bovine serum (FBS) and 1% penicillin and streptomycin at 37 oC in a humidified 5% CO2 atmosphere. Cells were passaged up to 20 times and media were changed twice or thrice a week. Cell density was set as appropriate for the planned assay (5000, 7000, 10,000, or 20,000 cells per well) in 96-multiwell plates in 100 uL total volumes of medium for each experiment. The day after the seeding, ATRA in 1% FBS DMEM/F12 medium was added to the cells to a final concentration of 10 uM, and incubation was continued for a further 48 h to induce differentiation, allow the formation of longer neurites, and reduce proliferation.
4.4. Microscopy
Micrographs of neuron-like SH-SY5Y cells were obtained using a DM IL LED fluorescence microscope (Leica Microsystems, Mannheim, Germany) with an appropriate excitation filter for the assay, or a brightfield setup with a DP73 high-performance digital camera (Olympus, Tokyo, Japan). Since the signal-to-noise ratio for staining was moderate, the contrast was slightly adjusted in ImageJ software (Fiji) without affecting the resulting observation. Original images are included in the Supplementary Materials.
4.5. Cell Membrane Staining (Neurite Outgrowth kit, Invitrogen™)
Neuron-like SH-SY5Y cells (5000 cells per well) obtained by the 48 h differentiation procedure were stained by a Neurite outgrowth kit (Invitrogen™) according to the manufacturer’s recommendations with minor modification. The cells were washed with PBS, labelled with a solution of the membrane staining dye supplied with the kit (following the protocol for 96 multiwell plates) in PBS for 20 min at 37 oC, washed again with PBS, and viewed under a fluorescence microscope (with 533 and 585 nm excitation and emission wavelengths, respectively).
4.6. Cell Treatment
After the differentiation procedure, the differentiation media was changed to 1% DMEM/F12 medium supplemented with test compounds at concentrations of 0.1, 1, and 10 uM (with 7000 cells per well for cytotoxicity tests), or together with the toxin SAL at 500 uM (with 7000–10,000 cells per well) or 160 mM Glu (with 20,000 cells per well) for an appropriate duration for the assay type (viability, cell death, etc.). Control cells were treated with a medium containing 。0.1% of DMSO.
4.7. Cell Viability and Cell Death
The viability of neuron-like SH-SY5Y cells growing in 96-well microplates (with ca. 7000 cells per well) after 24 h of treatments was evaluated by the Calcein AM assay with minor modification [31]. The Calcein AM solution in PBS used had 0.75 uM concentration and the incubation time was set to 50 min. The number of living cells in each well was determined using an Infinite M200 Pro microplate reader (Tecan, Austria), with 495 and 517 nm excitation and emission wavelengths, respectively.
Cell death of neuron-like cells (SAL model: 10,000 cells per well; Glu-model: 20,000 cells per well) was determined by PI assay as reported by Stone et al. 2003 with small modifi- cation [83]. Briefly, the medium of the SAL-induced model was aspirated and changed to a 1 ug/mL solution of PI in PBS, but cells subjected to Glu-induction have weaker adherence so a 1 mg/mL solution of PI in PBS was added to give a final concentration of 1 ug/mL. In both cases, cells were incubated for a further 15–25 min at room temperature then PI fluorescence was measured by an Infinite M200 Pro reader (Tecan, Grödig, Austria) with 535 and 617 excitation and emission wavelengths, respectively. The resulting PI fluorescence obtained with toxins alone was set as 100% cell death.
4.8. Measurement of Oxidative Stress by the Dihydroethidium (DHE) assay
Cells were differentiated and treated as described in the previous section for SAL- induction (10,000 cells per well, 24 h or Glu-induction (20,000 cells per well, 4 h). After the treatments, cells were centrifuged at 500 x g for 330 s, then the culture media were replaced (following aspiration) with 10 uM DHE solution in PBS. 96-multiwell plates with cells were incubated in the dark for 30 min at room temperature, then DHE fluorescence was measured by an Infinite M200 Pro microplate reader (Tecan, Grödig, Austria) with 500 and 580 nm excitation and emission wavelengths, respectively. The resulting DHE fluorescence obtained with SAL or Glu alone was set as 100% superoxide radical formation. Illustrative fluorescence microphotographs of DHE-stained cells were obtained after DHE measurement with the microplate reader.

Cistanche plant has a very good neuroprotective effect
4.9. Measurement of Caspase-3/7 Activity
One-step caspase-3/7 assays were performed according to a previously published procedure [84]. For cultures subjected to SAL-induction and Glu-induction, the reaction mixtures (caspase-3,7 buffer and components with cells in 96-multiwell plates) were incubated for 2 h and 3 h at 37 oC, respectively. Caspase-3/7 activity was then measured by an Infinite M200 Pro microplate reader (Tecan, Austria) with 346 and 438 nm excitation and emission wavelengths, respectively.
4.10. Statistical Analysis
Experiments were performed in triplicates and repeated in three to five (n = 3–5) independent days. All data are expressed as mean ± SEM. Values for all measured variables are expressed as means ± SEM, which were calculated using Prism 8.4.3 (GraphPad Software, La Jolla, CA, USA), which was also used to generate the figures. The statistical analysis was carried out by the PAST (ver. 1.97) software package [85]. For the differentiation experiment, the Student t-test was used. The rest of the experiments were evaluated by non-parametric Kruskal–Wallis test with Mann–Whitney post hoc test with sequential Bonferroni correction. A value of p < 0.05 was considered significant.
Supplementary Materials: Original images are available online.
Author Contributions: Contributions of the authors were as follows: Investigation, methodology, validation, data analysis, G.G., C.W.D., and J.G.; writing—original draft preparation G.G., C.W.D., and M.S.; data curation, funding acquisition, supervision, writing—review and editing, C.W.D., M.S., and P.K. All authors have read and agreed to the published version of the manuscript.
Funding: This work was supported financially in part by grants from the Internal Grant Agency of Palacky University in Olomouc, Czech Republic (IGA_PrF_2020_021), the Czech Grant Agency (20- 15621S), and the European Regional Development Fund—Project ENOCH (No. CZ.02.1.01/0.0/0.0/ 16_019/0000868) and a student grant from Palacky University’s Endowment Fund.
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
Data Availability Statement: Data is contained within the article or supplementary material.
Acknowledgments: Authors thank Dita Jordovd, Jana Hrubešovd, and Lucie Koplikovd for excellent technical assistance. Additionally, the authors thank Sees-editing Ltd., the U.K. for the English editing of the manuscript.
Conflicts of Interest: The authors declare no conflict of interest.
Sample Availability: Samples of the compounds are not available from the authors.






