Part Ⅱ Identification And Validation Of An Immune-related Gene Pairs Signature For Three Urologic Cancers
May 06, 2023
Results
1. Establishing and evaluating the IRGPI
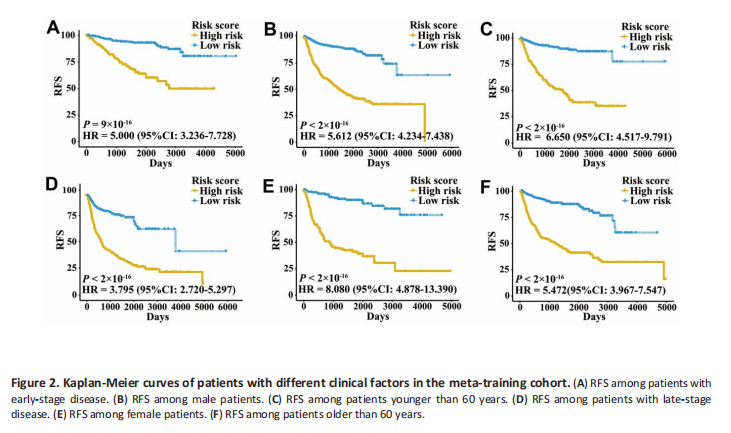
A total of 2,700 patients with three urologic cancers, including 835 bladder cancer, 888 prostate cancer, and 977 kidney cancer patients, were included in this study. As shown in Supplementary Figure 1, 1,622 patients in TCGA cohorts were used as the meta-training dataset. Another 1,078 patients from seven GEO datasets and two ICGC datasets constituted the meta-validation dataset. Among 2,483 IRGs from the ImmPort database, 606 IRGs were measured on all platforms and met the criteria (MAD > 0.5) on all independent training sets. Based on 606 IRGs, 183,315 IRGPs were constructed. After removing those not shared among all datasets or with constant ordering in any data set, 18,041 IRGPs were left and selected for further analysis. First, 10,943 IRGPs were filtered out by using the univariate Cox regression model in the meta-training data set. To further screen candidate IRGPs, LASSO was conducted 30 times to select those which appeared more than 15 times out of 30 analyses. As a result, 25 IRGPs and 41 unique IRGs were selected. The multivariate Cox regression model was then used to obtain the IRGPsbased prediction model and generate IRGPI scores for all samples. The patients in the meta-training cohort were classified into low and high-immune-risk groups by using the median IRGPI (-1.216973). There were significantly different prognoses in terms of RFS between low- and high-risk groups in the meta-training cohort (Figure 1A, hazard ratio [HR] 6.078, 95% confidence interval [CI] 4.754-7.769; P < 2×10-16). The IRGPI could also divide patients into subgroups with significantly different RFS in the training cohort of bladder cancer (Figure 1B, HR 2.308, 95% CI 1.660- 3.210; P = 3×10-7 ), prostate cancer (Figure 1C, HR 3.054, 95% CI 1.778-5.245; P = 2×10-5 ) and kidney cancer (Figure 1D, HR 5.582, 95% CI 3.899-7.992; P < 2×10-16). The time-dependent ROC curves were used to evaluate the ability of the IRGPI to predict prognosis. The area under the curve (AUC) values in predicting 1-, 3-, 5- and 10-year RFS of patients were 0.816, 0.818, 0.828, and 0.763, respectively, in the meta-training cohort (Supplementary Figure 2A). When patients were stratified by different tumor stages, genders, and age groups, low and high IRGPI groups remained significantly different for RFS, and a higher IRGPI score was associated with a significantly worse prognosis (Figure 2). The patients with high IRGPI and advanced tumor stage had the highest RFS among all patients in the meta-training cohort (Supplementary Figure 2B). Multivariate analyses suggested that the IRGPI was an independent prognostic factor (HR 4.22, 95% CI 3.23- 5.52; P < 2×10-16) after adjusting for age and stage.

2. Validation of the IRGPI
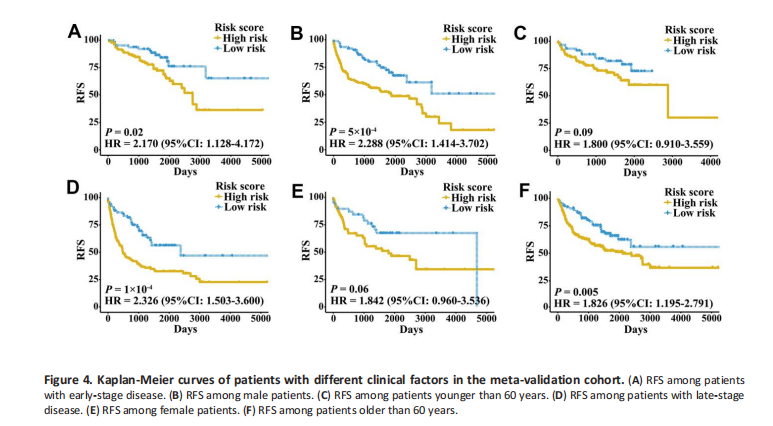
External validation cohorts were used to confirm the ability of the IRGPI to predict the RFS of patients with three urologic cancers in different populations. The same IRGPs were used to calculate the IRGPI, and the patients were also classified into low- and high-risk groups. RFS of patients in low and high IRGPI groups were significantly different in the meta-validation cohort (Figure 3A, HR 3.326, 95% CI 2.623-4.217; P < 2×10-16), validation dataset of bladder cancer (Figure 3B, HR 3.987, 95% CI 2.641-6.019; P = 1×10- 12), validation dataset of prostate cancer (Figure 3C, HR 3.277, 95% CI 2.275-4.719; P = 2×10-11), and validation dataset of kidney cancer (Figure 3D, HR 2.523, 95% CI 1.529-4.165; P = 2×10-4 ). Time-ROC curves showed stable predictive abilities, with 1-, 3-, 5-, and 10-year AUCs of 0.794, 0.764, 0.739, and 0.605, respectively (Supplementary Figure 2C). Similarly, the patients with high IRGPI and advanced tumor stage had the highest RFS among patients in the meta-validation cohort (Supplementary Figure 2D). Consistent with the training cohorts, the IRGPI was able to divide patients into significantly different groups in terms of RFS in mostly independent validation cohorts (Supplementary Figure 3). When considering patients with the early- or latest-age disease, male patients, and patients older than 60 years, the IRGPI remained highly prognostic, and a higher IRGPI score was associated with a significantly worse prognosis (Figure 4). After adjusting for age and stage in Cox regression analyses, the IRGPI remained an independent prognostic factor in the meta-validation cohort (HR 2.15, 95% CI 1.45-3.19; P = 0.000149;).

3. Functional analysis and infiltrating immune content related to the IRGPI
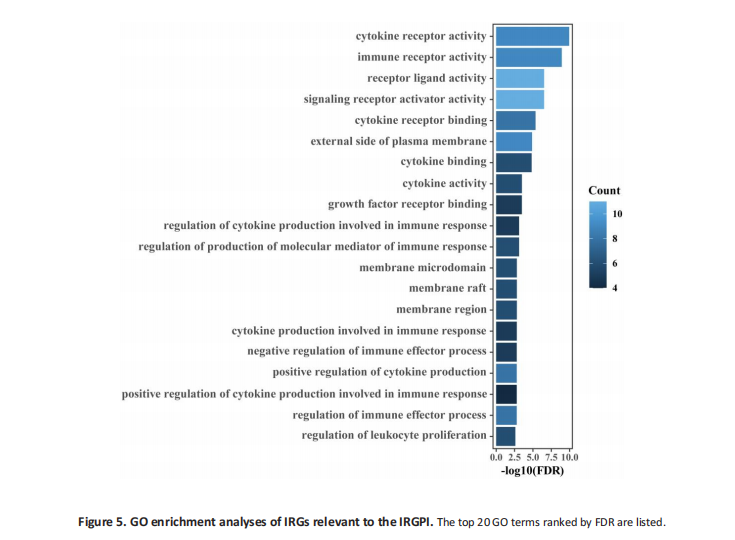
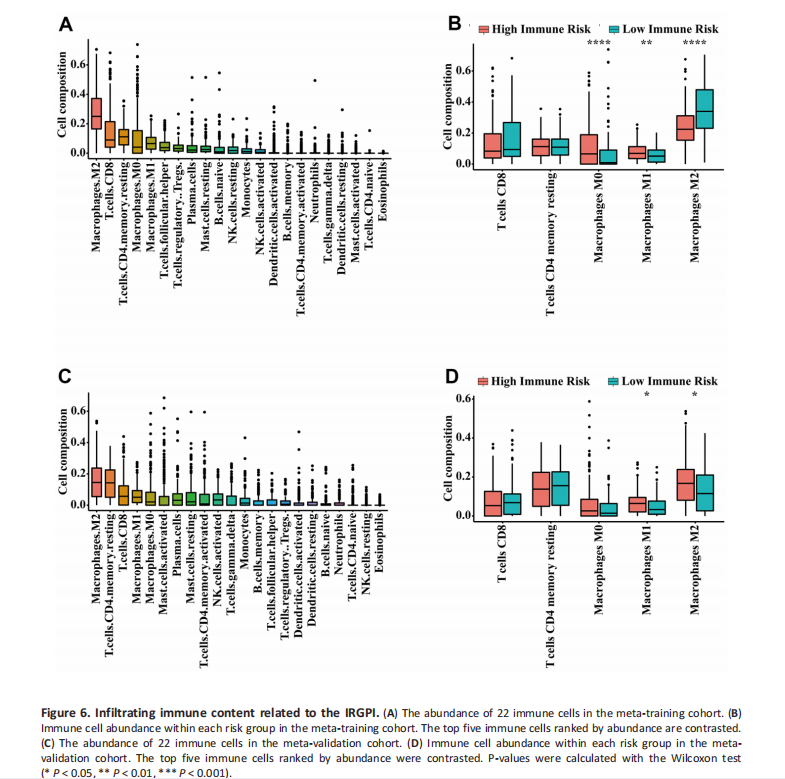
The 41 unique IRGs are relevant to the IRGPI in the meta-training cohort were mainly involved in the immune and inflammatory response, cytokine activity, and chemotaxis ( Figure 5). Various immune infiltrates were enriched in the meta-training cohort, and Macrophages M2, T cells CD8, T cells CD4 memory resting, Macrophages M0, and Macrophages M1 showed higher abundance (Figure 6A). Among those, percentages of Macrophages M0, Macrophages M1, and macrophage M2 were significantly different between IRGPI risk groups (Figure 6B). Furthermore, those results were validated in the meta-validation cohorts in which the same five immune infiltrates with higher immune cell abundance were enriched (Figure 6C). The percentages of Macrophages M1 and macrophage M2 were also significantly different between IRGPI risk groups (Figure 6D).
4. Nomogram based on the IRGPI and clinical factors
Univariate Cox regression analysis showed that age, stage, and IRGPI score were significant predictors of prognosis. To establish a quantitative approach for RFS prediction, we performed a nomogram based on prognostic factors (IRGPI, age, and stage) in the meta-training and meta-validation sets (Figure 7A, 7B). The reliability of the nomograms was evaluated by the calibration plot. As a result, the line segments in the calibration plots were close to the 45° line, indicating an excellent agreement between the prediction and observation in the meta-training cohort (Supplementary Figure 4A–4D) and meta-validation cohort (Supplementary Figure 4E–4H).


Click here to get the Cistanche supplements
Discussion
In this study, we developed a signature based on 25 IRGPs to simultaneously predict the prognosis of urinary cancer, including bladder, prostate, and kidney cancer. The signature showed a robust ability for predicting RFS of urinary cancers in training cohorts and multiple validation cohorts. Our signature could also distinguish different RFS in defined groups of patients (e.g., early-stage) in stratified analyses. The constructed nomogram based on the IRGPI score and clinical prognostic factors was able to quantitatively predict RFS rates of bladder, prostate, and kidney cancer patients.
Patients with bladder [36], prostate [37], and kidney cancer [38] have a substantial risk for relapse, even after surgical resection. The use of various adjuvant therapies, particularly in early-stage patients, remains disputable [39]. Reliable prognostic biomarkers are urgently needed to identify patients with a higher risk for relapse and select patients who have responses to therapies. Many signatures based on gene expression were developed to predict the prognosis of the bladder [40– 42], prostate [43–45], and kidney cancer [46–48]. However, the common drawback in those studies was technical biases caused by the normalization of expression profiles derived from different platforms using RNA-Seq or microarrays. Based on the relative ranking of gene expression, the IRGPs signature in the present study focused on pairwise comparisons within the gene expression profile of samples. The need for data normalization was eliminated to the utmost extent, and technical biases between different platforms when combining multiple gene expression profiles were avoided. A few researchers have identified IRGPs signatures related to the prognosis of urinary cancer patients; for example, Fu et al. found a novel IRGPs signature that had significant prognostic value in predicting overall survival in bladder cancer [49]. Researchers in southern China have also developed IRGPs to predict the prognosis of patients with papillary renal cell carcinoma [50]. However, these studies lacked sufficient validation due to the relatively small sample size, and could only predict the prognosis of a single tumor. In this study, we integrated large-scale datasets from multiple platforms to identify the IRGP's signature. The signature could simultaneously predict the RFS of three main urologic tumors and was robust after being verified. Therefore, our signature can be expediently promoted to clinical usage.

Standardized Cistanche
The tumor immune microenvironment is correlated with the prognosis of bladder [22, 51], prostate [23, 52], and kidney cancer [24, 53]. In the era of immunotherapy, prognostic biomarkers relevant to the tumor immune microenvironment may break a new path for identifying novel prognostic biomarkers. In this study, most of the IRGs involved in our immune signature were cytokines, antimicrobials, and cytokine receptors, which are closely related to immune response and inflammatory processes. Enrichment analysis also indicated that the IRGs relevant to the immune signature were mainly involved in the immune and inflammatory response and cytokine activity. An increased inflammatory microenvironment was found in the main tumors of the urinary system. This finding was consistent with previous studies showing that the formation and progression of tumors were related to an increased inflammatory microenvironment [54, 55]. Diverse immune cells such as neutrophils and macrophages are involved in the inflammatory response process of tumors. Macrophages are correlated with poor prognosis in many cancers [56, 57]. In the present study, we found the infiltration level of macrophage M1 in the immune high-risk group significantly increased. The dysregulated immune contexture may result in the survival differences observed between risk groups as defined by the IRGPI. Noteworthily, the infiltration level of M1 macrophages in the high-risk and low-risk groups is inconsistent in research [49, 50, 58–61]. Of these studies, two of them are consistent with our result [50, 58], three of them report that there is no significant difference [59–61], and only one research is contrary to ours [49]. The inconsistencies in the infiltration level of M1 macrophages may be related to study population differences, that is, the samples that generate risk scores are different.
To note, there were limitations in this study. Firstly, the 25 IRGPs' prognostic signature was based on a retrospective study, although nine datasets were used for rigorous validation. Our results should be further validated in prospective cohorts with different sample attributes. Secondly, our prognostic signature also needs to be validated by quantitative real-time polymerase chain reaction before it can be applied clinically. Thirdly, we removed IRGPs with constant values to reduce the influence of batch effects between different platforms, but batch effects cannot be eliminated.

Cistanche powder
In conclusion, our 25 IRGPs signature is a promising and robust prognostic biomarker for predicting the RFS of bladder, prostate, and kidney cancer, including early-stage cancers. Moreover, this signature was associated with the infiltration of immune cell subsets and immune response, indicating the associations between the immune microenvironment and those cancers, and hence could help to formulate personalized immunotherapy strategy. Although the clinical utility of our signature needs to be validated in prospective studies, our study has nonetheless provided a panel of promising prognostic markers by integrating large-scale datasets.
The effect of Cistanche extract on improving the urinary system
Cistanche extract is derived from the plant Cistanche Deserticola, which has been traditionally used in Chinese medicine for its health benefits. Its effectiveness in improving the urinary system is one of its well-known properties.
Studies have shown that Cistanche extract contains a range of bioactive compounds that have antioxidant and anti-inflammatory effects. These compounds can help protect against oxidative stress, inflammation, and cell damage in the urinary system. This leads to improved function and reduced risks of urological diseases.
One area where Cistanche extract has shown promising results is in improving symptoms associated with benign prostatic hyperplasia (BPH). BPH is a condition where the prostate gland enlarges and compresses the urethra, causing urinary symptoms such as increased urgency, frequency, weak flow, and incomplete bladder emptying.
Research suggests that Cistanche extract can relieve these symptoms by decreasing the size of the prostate gland. It does this by inhibiting the activity of an enzyme called 5-alpha-reductase, which converts testosterone to dihydrotestosterone (DHT), a hormone that stimulates prostate growth. By reducing DHT levels, Cistanche extract prevents further prostate enlargement and improves urinary symptoms.
Cistanche extract also has anti-inflammatory effects that can benefit other urinary tract conditions, such as urinary tract infections (UTIs) and interstitial cystitis (IC). Both conditions cause inflammation of the bladder wall, leading to urinary frequency, urgency, pain, and discomfort. By reducing inflammation, Cistanche extract may provide relief from these symptoms.
In conclusion, Cistanche extract's ability to improve the urinary system is derived from its anti-inflammatory and antioxidant properties. Although research is still in its early stages, the evidence so far suggests that Cistanche extract may be an effective treatment option for managing urinary tract conditions like BPH, UTIs, and IC. Nonetheless, it's important to note that individuals should always discuss any potential supplement additions to their diet with a qualified healthcare provider to ensure that it is safe and appropriate for their individual needs.

Cistanche's benefits
References
36. Wang X, Ding Y, Wang J, Wu Y. Identification of the Key Factors Related to Bladder Cancer by lncRNA-miRNAmRNA Three-Layer Network. Front Genet. 2020; 10:1398.
37. Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, Mason M, Matveev V, Wiegel T, Zattoni F, Mottet N, and European Association of Urology. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur Urol. 2014; 65:467–79.
38. Kelsey R. Kidney cancer: ccrcc1-4 classification for prediction of relapse. Nat Rev Urol. 2018; 15:205.
39. Roberts ME, Aynardi JT, Chu CS. Uterine leiomyosarcoma: A review of the literature and update on management options. Gynecol Oncol. 2018; 151:562–72.
40. Shariat SF, Karakiewicz PI, Godoy G, Karam JA, Ashfaq R, Fradet Y, Isbarn H, Montorsi F, Jeldres C, Bastian PJ, Nielsen ME, Müller SC, Sagalowsky AI, Lotan Y. Survivin as a prognostic marker for urothelial carcinoma of the bladder: a multicenter external validation study. Clin Cancer Res. 2009; 15:7012–9.
41. Mo Q, Nikolos F, Chen F, Tramel Z, Lee YC, Hayashi K, Xiao J, Shen J, Chan KS. Prognostic Power of a Tumor Differentiation Gene Signature for Bladder Urothelial Carcinomas. J Natl Cancer Inst. 2018; 110:448–59.
42. Sikic D, Eckstein M, Wirtz RM, Jarczyk J, Worst TS, Porubsky S, Keck B, Kunath F, Weyerer V, Breyer J, Otto W, Rinaldetti S, Bolenz C, et al. FOXA1 Gene Expression for Defining Molecular Subtypes of Muscle-Invasive Bladder Cancer after Radical Cystectomy. J Clin Med. 2020; 9:994.
43. Gerke JS, Orth MF, Tolkach Y, Romero-Pérez L, Wehweck FS, Stein S, Musa J, Knott MM, Hölting TL, Li J, Sannino G, Marchetto A, Ohmura S, et al. Integrative clinical transcriptome analysis reveals TMPRSS2-ERG dependency of prognostic biomarkers in prostate adenocarcinoma. Int J Cancer. 2020; 146:2036–46.
44. Wu CL, Schroeder BE, Ma XJ, Cutie CJ, Wu S, Salunga R, Zhang Y, Kattan MW, Schnabel CA, Erlander MG, McDougal WS. Development and validation of a 32-gene prognostic index for prostate cancer progression. Proc Natl Acad Sci USA. 2013; 110:6121–6.
45. Zhao SG, Evans JR, Kothari V, Sun G, Larm A, Mondine V, Schaeffer EM, Ross AE, Klein EA, Den RB, Dicker AP, Karnes RJ, Erho N, et al. The Landscape of Prognostic Outlier Genes in High-Risk Prostate Cancer. Clin Cancer Res. 2016; 22:1777–86.
46. Liang T, Sang S, Shao Q, Chen C, Deng Z, Wang T, Kang Q. Abnormal expression and prognostic significance of EPB41L1 in kidney renal clear cell carcinoma based on data mining. Cancer Cell Int. 2020; 20:356.
47. Yang CA, Huang HY, Yen JC, Chang JG. Prognostic Value of RNASEH2A-, CDK1-, and CD151-Related Pathway Gene Profiling for Kidney Cancers. Int J Mol Sci. 2018; 19:1586.
48. Kim KM, Hussein UK, Bae JS, Park SH, Kwon KS, Ha SH, Park HS, Lee H, Chung MJ, Moon WS, Kang MJ, Jang KY. The Expression Patterns of FAM83H and PANX2 Are Associated With Shorter Survival of Clear Cell Renal Cell Carcinoma Patients. Front Oncol. 2019; 9:14.
49. Fu Y, Sun S, Bi J, Kong C, Yin L. A novel immune-related gene pair prognostic signature for predicting overall survival in bladder cancer. BMC Cancer. 2021; 21:810.
50. Zhou X, Qiu S, Jin D, Jin K, Zheng X, Yang L, Wei Q. Development and Validation of an Individualized Immune-Related Gene Pairs Prognostic Signature in Papillary Renal Cell Carcinoma. Front Genet. 2020; 11:569884.
51. Wong YN, Joshi K, Khetrapal P, Ismail M, Reading JL, Sunderland MW, Georgiou A, Furness AJ, Ben Aissa A, Ghorani E, Oakes T, Uddin I, Tan WS, et al. Urinederived lymphocytes as a non-invasive measure of the bladder tumor immune microenvironment. J Exp Med. 2018; 215:2748–59.
52. Luo C, Chen J, Chen L. Exploration of gene expression profiles and immune microenvironment between high and low tumor mutation burden groups in prostate cancer. Int Immunopharmacol. 2020; 86:106709.
53. Şenbabaoğlu Y, Gejman RS, Winer AG, Liu M, Van Allen EM, de Velasco G, Miao D, Ostrovnaya I, Drill E, Luna A, Weinhold N, Lee W, Manley BJ, et al. Erratum to: Tumor immune microenvironment characterization in clear cell renal cell carcinoma identifies prognostic and immunotherapeutic ally relevant messenger RNA signatures. Genome Biol. 2017; 18:46.
54. Greten FR, Grivennikov SI. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity. 2019; 51:27–41.
55. Crusz SM, Balkwill FR. Inflammation and cancer: advances and new agents. Nat Rev Clin Oncol. 2015; 12:584–96.
56. Cassetta L, Pollard JW. Targeting macrophages: therapeutic approaches in cancer. Nat Rev Drug Discov. 2018; 17:887–904.
57. Ngambenjawong C, Gustafson HH, Pun SH. Progress in tumor-associated macrophage (TAM)-targeted therapeutics. Adv Drug Deliv Rev. 2017; 114:206–21.
58. Shi YR, Xiong K, Ye X, Yang P, Wu Z, Zu XB. Development of a prognostic signature for bladder cancer based on immune-related genes. Ann Transl Med. 2020; 8:1380.
59. Zheng X, Zhou X, Xu H, Jin D, Yang L, Shen B, Qiu S, Ai J, Wei Q. A Novel Immune-Gene Pair Signature Revealing the Tumor Microenvironment Features and Immunotherapy Prognosis of Muscle-Invasive Bladder Cancer. Front Genet. 2021; 12:764184.
60. Zhang LH, Li LQ, Zhan YH, Zhu ZW, Zhang XP. Identification of an IRGP Signature to Predict Prognosis and Immunotherapeutic Efficiency in Bladder Cancer. Front Mol Biosci. 2021; 8:607090.
61. Tao Z, Zhang E, Li L, Zheng J, Zhao Y, Chen X. A united risk model of 11 immune-related gene pairs and clinical stage for prediction of overall survival in clear cell renal cell carcinoma patients. Bioengineered. 2021; 12:4259–77.
Biao Xie 1, Kangjie Li 1, Hong Zhang 1, Guichuan Lai 1, Dapeng Li 2, Xiaoni Zhong1.
1. Department of Biostatistics, School of Public Health and Management, Chongqing Medical University, Chongqing, China
2. Institute of Hepatology, National Clinical Research Center for Infectious Disease, Shenzhen Third People’s Hospital, Shenzhen, China






