Part Two A Lifestyle Intervention To Delay Early Chronic Kidney Disease in African Americans With Diabetic Kidney Disease: Pre-Post Pilot Study
Jun 02, 2023
Results
1. Study Profile
Between January 9, 2017, and April 28, 2017, 77 patients were screened, and 30 eligible patients were enrolled in the study (Figure 1). All 30 (100%) patients completed the baseline assessment, and 26 (87%) completed assessments at 2 months. Four (13%) participants were lost to follow-up; hence, the analytical sample included 26 participants.

2. Baseline Demographic Profile
Table 1 shows the baseline characteristics of the study participants. The mean age of the study participants was 57 years, and the mean duration of diabetes was 14 years. The majority were female (21/30, 70%), unmarried (17/30, 57%), unemployed (20/30, 67%), and insured (30/30, 100%). Over half of the participants reported having a “good” health status (16/30, 53%) and not using any special equipment (16/30, 53%).

3. Feasibility Findings
Overall, 33 out of 77 (43%) participants contacted were eligible for the study, and 30 participants were successfully recruited for this study. Among the 30 participants, 21 (70%) completed all 6 sessions, 26 (87%) completed assessments at 2 months, and 1 (3%) dropped out of the study. The reasons for incomplete sessions were death in the family, illness, or hospitalization. One participant dropped out after enrollment because of hospitalization for pneumonia.

Click here to buy Cistanche supplements
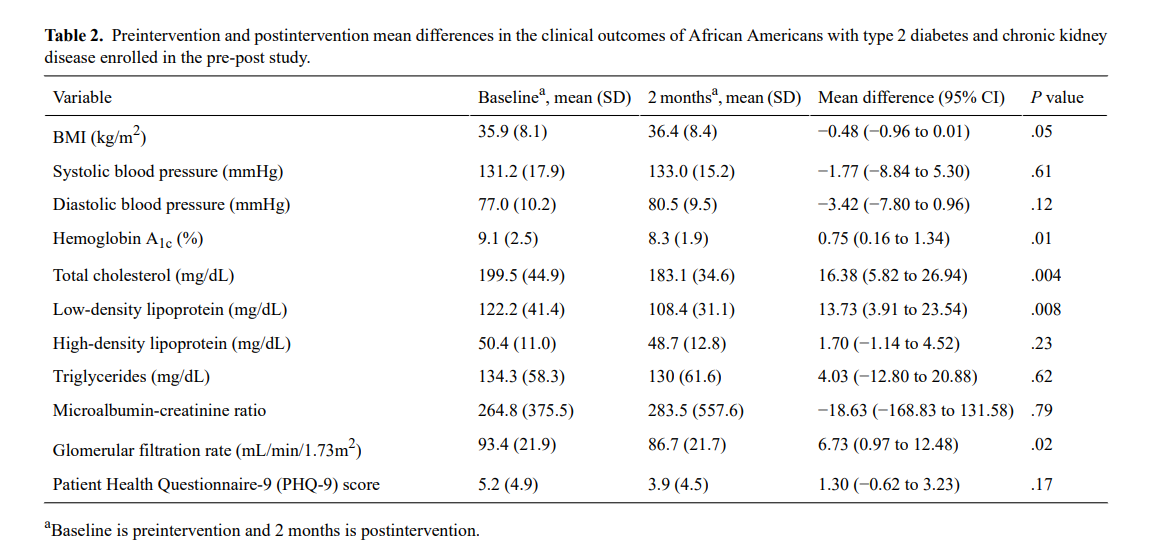
4. Preintervention and Postintervention Differences in Clinical Outcomes
Table 2 presents preintervention (baseline) and postintervention (2 months) differences in clinical outcomes. Significant preintervention and postintervention mean differences and decreases were observed for HbA1c (mean 0.75; P=.01), total cholesterol (mean 16.38; P=.004), low-density lipoprotein (LDL) (mean 13.73; P=.008), and eGFR (mean 6.73; P=.02). We observed nonstatistically significant increases in BMI (mean −0.48; P=.05), systolic blood pressure (mean −1.77; P=.61), diastolic blood pressure (mean −3.42; P=.21), and the UACR (mean −18.63; P=.79). There were also nonstatistically significant decreases in the PHQ-9 score for depression (mean 1.30; P=.17), high-density lipoprotein (mean 1.70; P=.23), and triglycerides (mean 4.03; P=.62).
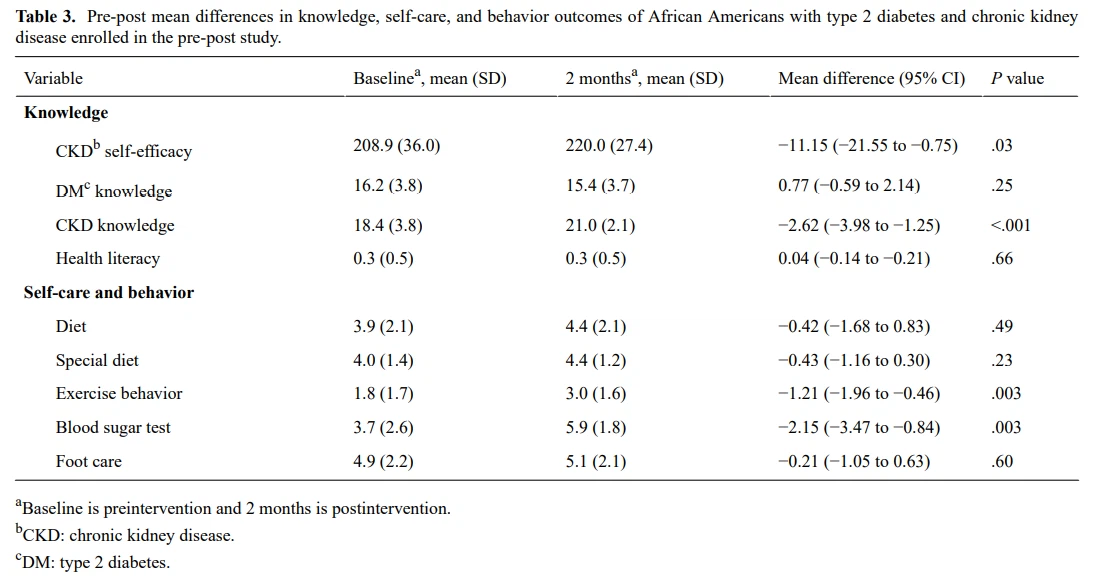
5. Preintervention and Postintervention Differences in Knowledge, Self-care, and Behavior Outcomes
Table 3 displays participant responses to questions related to knowledge, skills, self-care, and behavior outcomes. Significant preintervention and postintervention mean differences and increases were observed for CKD self-efficacy (mean −11.15; P=.03), CKD knowledge (mean −2.62; P<.001), exercise behavior (mean −1.21, P=.003), and blood sugar testing (mean −2.15; P=.003). We observed nonstatistically significant increases in diet (mean −0.42; P=.49), special diet (mean −0.43; P=.23), and foot care (mean −0.21; P=.60). A nonstatistically significant decrease in diabetes knowledge (mean 0.77; P=.25) was also observed, while no significant change in health literacy (mean 0.04; P=.66) was seen.
Discussion
1. Principal Findings
This study examined the feasibility and preliminary efficacy of a culturally tailored DKD-focused lifestyle intervention in African Americans with type 2 diabetes and CKD. With 100% recruitment, a 70% session attendance rate, and a 3% drop-out rate, the study findings suggest that the design, recruitment, and delivery of a culturally tailored lifestyle intervention for high-risk African Americans with type 2 diabetes and CKD are feasible. This study was also designed to examine preliminary changes in clinical outcomes, disease knowledge, self-care, and behavior outcomes. We observed statistically significant changes in the clinical outcomes of HbA1c, total cholesterol, LDL, and eGFR following the study intervention. In addition, there were statistically significant increases in CKD self-efficacy, CKD knowledge, and exercise. and blood sugar testing.
2. Comparison With Prior Work
Behavior lifestyle intervention trials have conflicting results on the impact of lifestyle interventions on clinical outcomes [12]. Consistent with our study findings, a systematic review by Van Huffel et al evaluating the impact of exercise and diet on health outcomes in individuals with diabetes and CKD concluded that exercise and diet interventions have beneficial effects on glycemic control, BMI, and quality of life [13]. Similarly, large trials, such as the “Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan (RENAAL),” “Action to Control Cardiovascular Risk in Diabetes (ACCORD),” “Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT),” and “Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE),” demonstrated that aggressive risk factor control in African Americans using antihypertensive, antihyperglycemic, or lipid-lowering medications is beneficial [29-33]. However, these studies focused on risk factor control using medications and did not emphasize lifestyle modification, which is a core component of diabetes and CKD management [3,9,10].

Cistanche tubulosa
Contrary to our study findings, a systematic review and meta-analysis of self-management support interventions for people with diabetes and CKD showed that these interventions may improve self-care activities, HbA1c, and systolic blood pressure [34]. While we observed a significant increase in CKD self-efficacy, disease knowledge, exercise behavior, and blood sugar testing, our study did not show a significant difference in blood pressure; however, it was not powered to confirm or refute a hypothesis, which could explain the lack of statistical significance in most clinical outcomes. The impacts of lifestyle interventions on kidney function are also inconsistent, with some studies demonstrating no effect, or a negative or positive effect [12,13]. We observed a significant negative effect (decrease) in the eGFR postintervention in our study population. Glomerular hyperfiltration often mediated by hyperglycemia results in a high eGFR in type 2 diabetes and is a hallmark finding in DKD [35]. Nonpharmacological interventions, such as decreases in body weight, and salt and protein intake, have been shown to ameliorate diabetic hyperfiltration [35]. It is unclear why we observed these findings given the lack of a significant difference in BMI or dietary habits. Future large-scale and more rigorous behavior lifestyle randomized controlled trials in this population should explore measuring changes in salt and protein intake, and examine the impact on outcomes.
Recent evidence demonstrates that glucose-lowering medications, such as glucagon-like peptide 1 receptor agonists and sodium-glucose cotransporter-2 inhibitors, are of particular benefit in the prevention and treatment of CKD in patients with type 2 diabetes [36]. However, despite the strengths of these large clinical trials [37-44] and limited data on the efficacy of lifestyle interventions in African Americans [12,15,34], African Americans remain poorly represented. It is established that African Americans with CKD are poorly represented in clinical trials [45], and the low inclusion of African Americans in clinical trials limits the generalizability of study findings. This potentially propagates existing disparities in a high-risk high-cost population. Low participation of African Americans in clinical trials is often attributed to poverty, lack of accessibility, lack of information on clinical trials, and chronic disease–related stigma [45,46]. There is a need to overcome these barriers and increase the participation of African Americans in clinical trials. Ongoing clinical trials are exploring novel community-based screening recruitment methods for African Americans with CKD [46,47]. More intervention studies that focus on high-risk patients incorporating such novel recruitment strategies are needed. In addition, behavioral lifestyle interventions that account for contextual factors facing high-risk African American populations with diabetes and CKD are needed [48].

Cistanche powder
Strengths, Limitations, and Future Direction
The findings of this study are promising and have important
clinical implications. Significant changes observed in clinical
outcomes, such as a decrease in HbA1c, and improved CKD
knowledge, self-care, and behavior, can prevent or delay the
progression of CKD to renal failure, and improve the quality of life
and survival in this study population. This could potentially
reduce the economic burden associated with renal failure and
the life-threatening complications of renal failure. Despite these
promising findings, some limitations are worth noting. First,
the relatively small sample size, limited intervention duration,
and lack of a control group might have affected the findings.
However, the goal of this feasibility pilot study was to generate
information needed for planning and designing a future
large-scale study. Second, eGFR was estimated using creatinine
and cystatin C equations with race. Recent evidence suggests
that the inclusion of race in eGFR estimation overestimates
measured eGFR, which potentially exacerbates health disparities
and contributes to systemic racism. While it is unlikely that the
eGFR equation used for this study influenced the study findings,
future studies will use new creatinine and cystatin C equations
without race to ensure accuracy. Third, although the majority of
the study participants completed all intervention sessions, some
weekly intervention sessions were delayed. The main reasons
for delayed intervention sessions were travel abroad,
hospitalization, and death in the family. Future studies will
incorporate a run-in period to establish expectations and
processes for the timely completion of intervention sessions in the
event of hospitalization or unanticipated events. In addition, we
will account for the loss of information due to dropout when
calculating the sample size. Fourth, the study findings may not be
generalizable to other populations since the study was primarily
designed for African American/non-Hispanic Black populations.

Herba Cistanche and Cistanche extract
Conclusion
This study clarifies the feasibility and preliminary efficacy of
a culturally tailored DKD-focused lifestyle intervention in
African Americans with type 2 diabetes and CKD in terms of
clinical, knowledge, self-care, and behavior outcomes. Statistically significant changes in the clinical outcomes of
HbA1c, total cholesterol, LDL, and eGFR were observed
following the study intervention. In addition, there were
statistically significant increases in CKD self-efficacy, CKD
knowledge, exercise, and blood sugar testing. Based on the
results of this study, a trial to determine the efficacy of this
intervention would be feasible in African Americans with type
2 diabetes and CKD. The findings from this study will also
serve as preliminary data to inform the design of a large-scale
appropriately powered randomized controlled trial to examine
the efficacy of a culturally tailored lifestyle intervention in
African Americans with comorbid diabetes and CKD in terms
of clinical, knowledge, self-care, and behavior outcomes.
Acknowledgments
The effort for this study was partially supported by the National Institute of Diabetes and Digestive Kidney Disease (R21DK131356, PI: MNO) and the National Institute of Diabetes and Digestive Kidney Disease (K24DK093699, R01DK118038, R01DK120861, PI: LEE).
Authors' Contributions
MNO and LEE designed the study. LEE analyzed the data. MNO drafted the manuscript. All authors were involved in the critical revision of the manuscript content. The final manuscript was approved by all the authors. LEE and MNO are guarantors of this work.
References
29. Appel LJ, Wright JT, Greene T, Agodoa LY, Astor BC, Bakris GL, AASK Collaborative Research Group. Intensive blood-pressure control in hypertensive chronic kidney disease. N Engl J Med 2010 Sep 02;363(10):918-929 [FREE Full text] [doi: 10.1056/NEJMoa0910975] [Medline: 20818902]
30. de Zeeuw D, Ramjit D, Zhang Z, Ribeiro AB, Kurokawa K, Lash JP, et al. Renal risk and renoprotection among ethnic groups with type 2 diabetic nephropathy: a post hoc analysis of RENAAL. Kidney Int 2006 May;69(9):1675-1682 [FREE Full text] [doi: 10.1038/sj.ki.5000326] [Medline: 16572114]
31. Action to Control Cardiovascular Risk in Diabetes Study Group, Gerstein HC, Miller ME, Byington RP, Goff DC, Bigger JT, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008 Jun 12;358(24):2545-2559 [FREE Full text] [doi: 10.1056/NEJMoa0802743] [Medline: 18539917]
32. Rahman M, Pressel S, Davis BR, Nwachuku C, Wright JT, Whelton PK, et al. Renal outcomes in high-risk hypertensive patients treated with an angiotensin-converting enzyme inhibitor or a calcium channel blocker vs a diuretic: a report from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). Arch Intern Med 2005 Apr 25;165(8):936-946. [doi: 10.1001/architect.165.8.936] [Medline: 15851647]
33. ADVANCE Collaborative Group, Patel A, MacMahon S, Chalmers J, Neal B, Billot L, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008 Jun 12;358(24):2560-2572. [doi: 10.1056/NEJMoa0802987] [Medline: 18539916]
34. Zimbudzi E, Lo C, Misso ML, Ranasinha S, Kerr PG, Teede HJ, et al. Effectiveness of self-management support interventions for people with comorbid diabetes and chronic kidney disease: a systematic review and meta-analysis. Syst Rev 2018 Jun 13;7(1):84 [FREE Full text] [doi: 10.1186/s13643-018-0748-z] [Medline: 29898785]
35. Tonneijck L, Muskiet MHA, Smits MM, van Bommel EJ, Heerspink HJL, van Raalte DH, et al. Glomerular Hyperfiltration in Diabetes: Mechanisms, Clinical Significance, and Treatment. J Am Soc Nephrol 2017 Apr;28(4):1023-1039 [FREE Full text] [doi: 10.1681/ASN.2016060666] [Medline: 28143897]
36. Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. The Lancet 2019 Jan 05;393(10166):31-39 [FREE Full text] [doi: 10.1016/S0140-6736(18)32590-X] [Medline: 30424892]
37. Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JFE, Nauck MA, LEADER Steering Committee, LEADER Trial Investigators. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 2016 Jul 28;375(4):311-322 [FREE Full text] [doi: 10.1056/NEJMoa1603827] [Medline: 27295427]
38. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, SUSTAIN-6 Investigators. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med 2016 Nov 10;375(19):1834-1844. [doi: 10.1056/NEJMoa1607141] [Medline: 27633186]
39. Hernandez AF, Green JB, Janmohamed S, D'Agostino RB, Granger CB, Jones NP, Harmony Outcomes committee investigators. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): a double-blind, randomized placebo-controlled trial. Lancet 2018 Oct 27;392(10157):1519-1529 [FREE Full text] [doi: 10.1016/S0140-6736(18)32261-X] [Medline: 30291013]
40. Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, EMPA-REG OUTCOME Investigators. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med 2015 Nov 26;373(22):2117-2128. [doi: 10.1056/NEJMoa1504720] [Medline: 26378978]
41. Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, CANVAS Program Collaborative Group. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med 2017 Aug 17;377(7):644-657. [doi: 10.1056/NEJMoa1611925] [Medline: 28605608]
42. Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, DECLARE–TIMI 58 Investigators. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 2019 Jan 24;380(4):347-357. [doi: 10.1056/NEJMoa1812389] [Medline: 30415602]
43. Holman RR, Bethel MA, Mentz RJ, Thompson VP, Lokhnygina Y, Buse JB, EXCEL Study Group. Effects of Once-Weekly Exenatide on Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 2017 Sep 28;377(13):1228-1239 [FREE Full text] [doi: 10.1056/NEJMoa1612917] [Medline: 28910237]
44. Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, ELIXA Investigators. Lixisenatide in Patients with Type 2 Diabetes and Acute Coronary Syndrome. N Engl J Med 2015 Dec 03;373(23):2247-2257 [FREE Full text] [doi: 10.1056/NEJMoa1509225] [Medline: 26630143]
45. Harding K, Mersha TB, Vassalotti JA, Webb FA, Nicholas SB. Current State and Future Trends to Optimize the Care of Chronic Kidney Disease in African Americans. Am J Nephrol 2017;46(2):176-186 [FREE Full text] [doi: 10.1159/000479481] [Medline: 28787720]
46. Mitchell S, Bragg A, Moldovan I, Woods S, Melo K, Martin-Howard J, et al. Stigma as a Barrier to Participant Recruitment of Minority Populations in Diabetes Research: Development of a Community-Centered Recruitment Approach. JMIR Diabetes 2021 May 03;6(2):e26965 [FREE Full text] [doi: 10.2196/26965] [Medline: 33938811]
47. Wesson DE, Kitzman H, Montgomery A, Mamun A, Parnell W, Vilayvanh B, et al. A population health dietary intervention for African American adults with chronic kidney disease: The Fruit and Veggies for Kidney Health randomized study. Contemp Clin Trials Commun 2020 Mar;17:100540 [FREE Full text] [doi: 10.1016/j.conctc.2020.100540] [Medline: 32090186]
Mukoso N Ozieh* , MD, MSCR; Leonard E Egede, MD, MS
Department of Medicine, Division of Nephrology, Medical College of Wisconsin, Milwaukee, WI, United States






