Research On UHPLC-Based Extraction And Ultrafiltration Separation Process Optimization For Cistanche Extracts
Apr 01, 2026
Source link provided by user: https://www.xjcistanche.com/news/hplc-for-simultaneous-determination-of-four-ph-77351688.html
Note: The content below is translated and editorially optimized for factories and small workshops interested in herbal extract process optimization, while keeping the original technical meaning and structure as intact as possible.
Abstract
Objective: To establish a UHPLC multi-component quantitative analysis method for Cistanche and to optimize the extraction process and ultrafiltration membrane separation process.
Methods: A rapid UHPLC method was developed for simultaneous quantification of three phenylethanoid glycosides (PhGs) in Cistanche extract solution. The extraction process was optimized using a Box–Behnken response surface model. For ultrafiltration, the retention rates of the three marker compounds and membrane flux were selected as quality and efficiency indicators, respectively. The CRITIC method was applied to determine indicator weights, enabling a comprehensive evaluation of an orthogonal ultrafiltration experiment, and the optimal membrane separation conditions were selected.
Results: The established UHPLC method for Cistanche extract solution was stable, rapid, and efficient. Based on response surface analysis and industrial feasibility, the optimized extraction process was determined as:
"Extract twice using 7× solvent volume of 60% ethanol, 80 min each time."
Comprehensive evaluation of the ultrafiltration orthogonal experiment showed that membrane pore size significantly affected the overall score. The optimal ultrafiltration separation process was:
"100 kDa membrane module, 35°C, 0.5 MPa."
Conclusion: This study established a UHPLC multi-component quantitative method and comprehensively evaluated the extraction and ultrafiltration separation processes for Cistanche. The analytical method and process are stable and reliable, providing a practical reference for scale-up and industrial application of Cistanche extraction and membrane separation.
Keywords: Cistanche; UHPLC; extraction; ultrafiltration membrane; separation process; process optimization; phenylethanoid glycosides; quality control; how to make high quality extract.
Cistanche Tubulosa Extract Manufacturing Flow Chart (Source from wecistanche)


Supportive Service Of Wecistanche-For more details about cooperation
Email:wallence.suen@wecistanche.com
1. Background (Why this matters for making high-quality Cistanche extract)
Cistanche has a long history of use as a food and traditional herbal material in China, traditionally associated with functions such as "tonifying kidney-yang" and "nourishing essence and blood." Its bioactive constituents include phenylethanoid glycosides, iridoid terpenes, lignans, polysaccharides, etc. Among them, phenylethanoid glycosides (PhGs) are widely recognized as key active components, with reported activities including anti-inflammatory, anti-tumor, antioxidant, anti-aging, bone-density improvement, and neuroprotection.
Cistanche parasitizes the roots of Haloxylon (a Chenopodiaceae plant) and is mainly distributed in Xinjiang, Gansu, and other regions. With the expansion of cultivation, its use in pharmaceuticals and dietary supplements has grown rapidly.
In modern herbal manufacturing, multi-component evaluation is increasingly used for quality control across raw materials and extraction/separation steps. However, systematic research evaluating Cistanche extraction and membrane separation using multiple PhG markers-especially via fast and efficient quantitative methods-has been relatively limited. At the same time, membrane separation has become widely applied in TCM and supplement manufacturing because of high separation precision and lower-carbon, environmentally friendly processing, and it can improve the stability of liquid preparations.
This study therefore:
developed a rapid UHPLC method for three key PhGs in Cistanche extract solution;
optimized extraction using response surface methodology;
evaluated ultrafiltration using marker retention + membrane flux, with objective weighting by CRITIC, supporting process selection for industrialization.
2. Instruments and Materials
2.1 Instruments
Agilent 1290 Infinity II UHPLC (Agilent Technologies, USA)
ACQUITY UPLC HSS T3 column (100 mm × 2.1 mm, 1.8 μm; Waters, USA)
XS-105DU analytical balance (Mettler Toledo)
BONA-GM-18 microfiltration/ultrafiltration/nanofiltration membrane separation unit
TKF-18 ultrafiltration membranes (50, 100, 200 kDa; Shandong Bona Biotechnology Co., Ltd.)
PURELAB classic ultrapure water system (ELGA, UK)

Note:1:Echinacoside;2:Verbascoside;3:Isoacteoside
2.2 Materials and Reagents
Reference standards (National Institutes for Food and Drug Control, China):
Echinacoside (111670-201907, 91.8%)
Acteoside / Verbascoside (111530-202315, 97.6%)
Isoacteoside (112112-202401, 97.0%)
Acetonitrile, HPLC grade (Fisher Chemical, USA)
Water: ultrapure water
Cistanche decoction pieces (batch no. 250221) purchased from Henan Dali Pharmaceutical Co., Ltd.
Identified by researcher Sun Daihua (Jingpai Chizhengtang Pharmaceutical Co., Ltd.)
Botanical origin: Cistanche tubulosa (Schenk) Wight
Verified to comply with Pharmacopoeia of the People's Republic of China (2020 edition) requirements.
Table 1. Linearity, correlation coefficient, linear range, and LOQ of three components
| Component | Regression equation (Y = peak area, X = concentration) | Correlation coefficient (R) | Linear range (µg·mL⁻¹) | LOQ (µg·mL⁻¹) |
|---|---|---|---|---|
| Echinacoside | Y = 1.873X + 49.049 | 0.9998 | 24.21–774.79 | 0.69 |
| Acteoside (Verbascoside) | Y = 2.038X − 6.483 | 0.9996 | 15.96–510.64 | 0.46 |
| Isoacteoside | Y = 2.039X − 3.105 | 0.9999 | 5.00–159.86 | 0.52 |
Table 2. Precision, repeatability, stability, and spike recovery results (n = 6)
| Component | Precision (RSD, %) | Repeatability (RSD, %) | Stability (RSD, %) | Spike recovery (%) | Recovery RSD (%) |
|---|---|---|---|---|---|
| Echinacoside | 0.93 | 1.05 | 1.38 | 98.47 | 1.27 |
| Acteoside (Verbascoside) | 0.86 | 1.39 | 1.54 | 101.35 | 1.04 |
| Isoacteoside | 0.62 | 0.89 | 1.07 | 99.58 | 0.93 |
Table 3. Experimental factors and levels (Box–Behnken design)
| Level | Factor A: Ethanol concentration (%) | Factor B: Solvent-to-material ratio (×) | Factor C: Extraction time (min) |
|---|---|---|---|
| 1 | 50 | 6 | 30 |
| 2 | 60 | 7 | 60 |
| 3 | 70 | 8 | 90 |
Table 4. Box–Behnken response surface experimental results
| Run | A: Ethanol concentration (%) | B: Solvent ratio (×) | C: Extraction time (min) | Y₁: Total extraction yield (%) |
|---|---|---|---|---|
| 1 | 70 | 8 | 60 | 8.40 |
| 2 | 70 | 7 | 90 | 8.31 |
| 3 | 60 | 6 | 90 | 9.09 |
| 4 | 60 | 7 | 60 | 9.56 |
| 5 | 60 | 8 | 30 | 8.69 |
| 6 | 60 | 7 | 60 | 9.46 |
| 7 | 50 | 6 | 60 | 8.24 |
| 8 | 60 | 7 | 60 | 9.57 |
| 9 | 70 | 7 | 30 | 7.74 |
| 10 | 50 | 7 | 30 | 7.81 |
| 11 | 60 | 7 | 60 | 9.54 |
| 12 | 60 | 8 | 90 | 9.53 |
| 13 | 50 | 8 | 60 | 8.84 |
| 14 | 50 | 7 | 90 | 8.99 |
| 15 | 70 | 6 | 60 | 7.77 |
| 16 | 60 | 6 | 30 | 8.03 |
| 17 | 60 | 7 | 60 | 9.51 |
Table 5. ANOVA for the response surface model
| Source | Sum of squares | df | Mean square | F value | P value | Significance |
|---|---|---|---|---|---|---|
| Model | 7.76 | 9 | 0.8627 | 377.29 | < 0.0001 | **** |
| A | 0.3444 | 1 | 0.3444 | 150.65 | < 0.0001 | **** |
| B | 0.6786 | 1 | 0.6786 | 296.80 | < 0.0001 | **** |
| C | 1.6700 | 1 | 1.6700 | 728.35 | < 0.0001 | **** |
| AB | 0.0002 | 1 | 0.0002 | 0.0984 | 0.7629 | ns |
| AC | 0.0930 | 1 | 0.0930 | 40.69 | 0.0004 | *** |
| BC | 0.0121 | 1 | 0.0121 | 5.29 | 0.0550 | ns |
| A² | 3.5600 | 1 | 3.5600 | 1555.28 | < 0.0001 | **** |
| B² | 0.3702 | 1 | 0.3702 | 161.89 | < 0.0001 | **** |
| C² | 0.6619 | 1 | 0.6619 | 289.51 | < 0.0001 | **** |
| Residual | 0.0160 | 7 | 0.0023 | - | - | - |
| Lack of fit | 0.0081 | 3 | 0.0027 | 1.37 | 0.3712 | ns |
| Pure error | 0.0079 | 4 | 0.0020 | - | - | - |
| Total | 7.78 | 16 | - | - | - | - |
3. Methods and Results
3.1 Multi-Component Content Determination (UHPLC)
3.1.1 Preparation of Mixed Reference Solution
Accurately weigh appropriate amounts of echinacoside, acteoside (verbascoside), and isoacteoside standards and dissolve in methanol to prepare stock solutions at concentrations of 1936.980, 1702.144, and 799.280 μg·mL⁻¹, respectively. Accurately pipette 1.00 mL, 0.50 mL, and 0.50 mL of the three stock solutions into a 5 mL volumetric flask, dilute to volume with methanol, and mix well to obtain the mixed reference solution.

Figure 3. Three-dimensional response surface plot of the interactions among various factors.
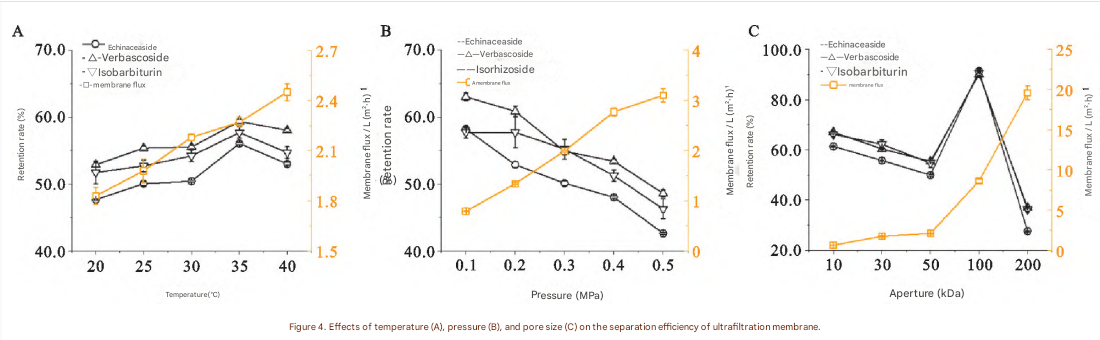
Figure 4. Effects of temperature (A), pressure (B), and pore size (C) on the separation efficiency of ultrafiltration membrane.
3.1.2 Preparation of Sample Solution
Accurately pipette 1.00 mL of Cistanche extract solution or ultrafiltrate into a 10 mL volumetric flask, dilute to volume with 50% ethanol, mix well, filter, and collect the subsequent filtrate as the test solution.
3.1.3 Chromatographic Conditions
Column: ACQUITY UPLC HSS T3 (100 mm × 2.1 mm, 1.8 μm)
Mobile phase: Acetonitrile (A) – 0.1% formic acid solution (B)
Gradient elution:
0–5 min: 15%–22% A
5–7 min: 22%–27% A
7–10 min: 27%–30% A
Detection wavelength: 330 nm
Flow rate: 0.3 mL/min
Column temperature: 30°C
Injection volume: 1 μL
Vacuum Tank (by Wecistanche)

Table 6. Ranges of single-factor experiments for ultrafiltration separation
| Factor | Level 1 | Level 2 | Level 3 | Level 4 | Level 5 |
|---|---|---|---|---|---|
| A: Temperature (°C) | 20 | 25 | 30 | 35 | 40 |
| B: Pressure (MPa) | 0.1 | 0.2 | 0.3 | 0.4 | 0.5 |
| C: MWCO (kDa) | 10 | 30 | 50 | 100 | 200 |
3.1.4 Specificity
Test solutions (S1), mixed reference solution, and blank solution (50% ethanol) were injected. Peak resolution for each component was > 1.5, and the blank showed no interference.
Table 7. Factors and levels for the orthogonal experiment
| Level | A: Temperature (°C) | B: Pressure (MPa) | C: MWCO (kDa) | D: Blank |
|---|---|---|---|---|
| 1 | 30 | 0.3 | 50 | 1 |
| 2 | 35 | 0.4 | 100 | 2 |
| 3 | 40 | 0.5 | 200 | 3 |
3.1.5 Linearity and Limit of Quantitation (LOQ)
Prepare a mixed reference solution containing echinacoside, acteoside, and isoacteoside at 774.792, 510.643, and 159.856 μg·mL⁻¹, respectively, then dilute 2×, 4×, 8×, 16×, 32× to obtain six concentration levels. Under the chromatographic conditions above, use concentration (X) vs. peak area (Y) to establish calibration curves. Correlation coefficients (R) were ≥ 0.9996, indicating excellent linearity. (Regression equation, linear range, and LOQ are reported in the original tables.)
Table 8. Orthogonal experimental design and results for ultrafiltration membrane separation
Note: Retention rate WWW is reported for three analytes:
W1W_1W1 Echinacoside; W2W_2W2 Acteoside (Verbascoside); W3W_3W3 Isoacteoside.
Membrane flux W4W_4W4 unit: [L⋅(m2⋅h)−1][ \mathrm{L \cdot (m^2 \cdot h)^{-1}} ][L⋅(m2⋅h)−1]. Comprehensive score Y2Y_2Y2 unit: (%).
| Run | A: Temp (°C) | B: Pressure (MPa) | C: MWCO (kDa) | D (blank) | W1W_1W1 (%) | W2W_2W2 (%) | W3W_3W3 (%) | W4W_4W4 [L⋅(m2⋅h)−1][L·(m²·h)⁻¹][L⋅(m2⋅h)−1] | Y2Y_2Y2 (%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 30 | 0.3 | 50 | 1 | 3.49 | 1.12 | 0.36 | 2.05 | 39.88 |
| 2 | 30 | 0.4 | 100 | 2 | 5.72 | 1.62 | 0.50 | 11.14 | 67.71 |
| 3 | 30 | 0.5 | 200 | 3 | 1.59 | 0.61 | 0.20 | 32.14 | 52.85 |
| 4 | 35 | 0.3 | 100 | 3 | 6.65 | 1.89 | 0.62 | 9.38 | 76.48 |
| 5 | 35 | 0.4 | 200 | 1 | 1.65 | 0.63 | 0.21 | 25.00 | 46.27 |
| 6 | 35 | 0.5 | 50 | 2 | 3.36 | 1.08 | 0.35 | 3.04 | 39.64 |
| 7 | 40 | 0.3 | 200 | 2 | 1.74 | 0.68 | 0.22 | 26.79 | 49.36 |
| 8 | 40 | 0.4 | 50 | 3 | 3.23 | 1.04 | 0.33 | 2.96 | 37.94 |
| 9 | 40 | 0.5 | 100 | 1 | 5.52 |
Factor level totals / ranges (as shown in the figure):
| Statistic | A | B | C | D |
|---|---|---|---|---|
| k1k_1k1 | 53.48 | 55.24 | 39.15 | 52.39 |
| k2k_2k2 | 54.13 | 50.64 | 71.74 | 52.24 |
| k3k_3k3 | 52.78 | 54.51 | 49.49 | 55.76 |
| RRR | 1.35 | 4.60 | 32.59 | 3.52 |
Table 9. Analysis of variance (ANOVA) results
| Factor | DfD_fDf | Adj SS | Adj MS | F | P |
|---|---|---|---|---|---|
| A: Temperature | 2 | 2.75 | 1.37 | 0.12 | 0.90 |
| B: Pressure | 2 | 36.65 | 18.32 | 1.54 | 0.39 |
| C: MWCO | 2 | 1663.72 | 831.86 | 70.12 | 0.01 |
| D: Blank | 2 | 23.73 | 11.86 | - | - |
Table 10. Verification results of the orthogonal experiment
| Run | Echinacoside retention (%) | Acteoside retention (%) | Isoacteoside retention (%) | Membrane flux [L⋅(m2⋅h)−1][L·(m²·h)⁻¹][L⋅(m2⋅h)−1] |
|---|---|---|---|---|
| 1 | 94.85 | 92.54 | 91.04 | 10.42 |
| 2 | 94.71 | 93.03 | 92.54 | 10.04 |
| 3 | 94.71 | 92.54 | 91.04 | 10.61 |
| Mean | 94.76 | 92.70 | 91.54 | 10.36 |
| RSD (%) | 0.1 | 0.3 | 0.9 | 2.8 |
3.1.6 Precision, Repeatability, Stability, and Recovery
Method validation was carried out according to the analytical method validation guidelines in the Chinese Pharmacopoeia (2020), including precision, repeatability, 48-hour stability, and spike recovery. Results indicated that the method is suitable for content determination of Cistanche extract solution.
3.2 Extraction Process Study (How to extract for high quality and high yield)
3.2.1 Single-Factor Experiments
Each extraction used 30 g crushed Cistanche raw material. Using the extraction yields of echinacoside, acteoside, and isoacteoside as evaluation indicators, the following factors were studied:
Ethanol concentration: 40%, 50%, 60%, 70%, 80%
Extraction times: 1, 2, 3, 4, 5
Solvent-to-material ratio: 5×, 6×, 7×, 8×, 9×
Extraction time: 30, 60, 90, 120, 150 min
When studying one factor, others were fixed (baseline: 60% ethanol, 7× solvent, 3 times, 1.5 h). Extraction method: reflux extraction (water bath at 80°C). Each condition was performed in triplicate.
Findings:
Total extraction yield of the three markers increased then decreased with ethanol concentration; differences were significant (P < 0.05). Best ethanol concentration: 60%.
Solvent ratio, extraction times, and time showed similar trends: yield increased and then plateaued. After 7× solvent, 2 extractions, and 1 h, differences were no longer significant.
Considering scale-up cost and efficiency, the response surface design used: ethanol 50–70%, solvent 6–8×, time 30–90 min, with extraction times set to 2.
Membrane Separation Equipment (by Wecistanche)

3.2.2 Box–Behnken Response Surface Design and Results
A Box–Behnken response surface model was used for further optimization. The response variable was the total extraction yield (Y1) of the three phenylethanoid glycosides.
The fitted regression equation was:
Y1=9.53−0.21A+0.29B+0.46C+0.01AB−0.15AC−0.06BC−0.92A2−0.30B2−0.40C2Y1 = 9.53 - 0.21A + 0.29B + 0.46C + 0.01AB - 0.15AC - 0.06BC - 0.92A^2 - 0.30B^2 - 0.40C^2Y1=9.53−0.21A+0.29B+0.46C+0.01AB−0.15AC−0.06BC−0.92A2−0.30B2−0.40C2
with R2=0.9979R^2 = 0.9979R2=0.9979.
ANOVA showed:
The model was highly significant (P < 0.0001)
Lack of fit was not significant (P = 0.3712), indicating good model suitability
Factor influence order: C > B > A
A, B, C and quadratic terms A2A^2A2, B2B^2B2, C2C^2C2 were extremely significant (P < 0.0001)
Interaction term AC was extremely significant (P < 0.001)
Using Design-Expert 12, predicted optimum conditions were:
Ethanol: 58.41%
Solvent ratio: 7.44×
Time: 77.26 min
Considering industrial control convenience and cost, the final practical condition was adjusted to:
60% ethanol, 7× solvent, 80 min, extracted twice.
3.2.3 Process Verification
Three parallel validation experiments were conducted under the selected optimum conditions. The average total extraction yield was 9.66%, matching the model prediction well (relative error 0.11%). The extraction yields of all three marker compounds were at a high level, indicating good agreement between predicted and actual results.
3.3 Ultrafiltration Separation Process Study (How to improve clarity/stability while retaining actives)
3.3.1 Preparation of Ultrafiltration Feed Solution
Twelve kilograms of crushed Cistanche decoction pieces were extracted using the optimized extraction process above, and the extract solution was used as the ultrafiltration feed. The measured concentrations were:
Echinacoside: 6.99 mg·mL⁻¹
Acteoside: 2.01 mg·mL⁻¹
Isoacteoside: 0.67 mg·mL⁻¹
3.3.2 Ultrafiltration Procedure and Evaluation Indicators
For each run, 2 L of feed solution was processed in the membrane separation unit. Temperature and transmembrane pressure were adjusted per the experimental design. After equilibrium (0.2 L), membrane flux was calculated as:
Flux=VtA\text{Flux} = \frac{V}{tA}Flux=tAV
where VVV is permeate volume, ttt is filtration time, and AAA is effective membrane area.
UHPLC was used to quantify the three markers in the permeate. The ultrafiltration effect was evaluated using:
Retention rate of each active component
Retention (%)=Content in ultrafiltrateContent in feed×100%\text{Retention (\%)} = \frac{\text{Content in ultrafiltrate}}{\text{Content in feed}} \times 100\%Retention (%)=Content in feedContent in ultrafiltrate×100%
Membrane flux (efficiency indicator)
3.3.3 Single-Factor Design
Temperature, pressure, and MWCO (pore size) were studied as single factors with baseline conditions set to 30°C, 0.3 MPa, 50 kDa. The ranges selected for the orthogonal experiment were determined based on single-factor outcomes.
3.3.4 Single-Factor Findings (Practical meaning for production)
Temperature: retention trends were similar; 35°C gave the best overall effect; flux tended to increase with temperature → selected 30, 35, 40°C
Pressure: retention decreased with pressure, but flux strongly depended on pressure and determines throughput → selected 0.3, 0.4, 0.5 MPa
MWCO: strongly affected both retention and flux. 100 kDa achieved the highest retention for the three markers, while 200 kDa gave lower retention → selected 50, 100, 200 kDa
Comprehensive Evaluation and Optimal Ultrafiltration Conditions
Because flux varied more strongly than retention across conditions, the study applied the CRITIC method with a dynamic adjustment mechanism to avoid overweighting highly dispersed indicators (flux). Based on overall scoring, membrane pore size had a significant influence on the score.
Optimal ultrafiltration process:
100 kDa membrane module, 35°C, 0.5 MPa
This process is described as low-carbon and environmentally friendly, with high retention of active markers while also considering separation efficiency, making it suitable for broader application.
4. Discussion (Implementation guidance for factories & workshops)
4.1 Multi-marker QC is the foundation of "high quality extract"
If you want to make a consistent high-quality herbal extract, you need:
A fast assay method that can be run frequently (in-process QC)
Multiple active markers (not only one compound)
This study demonstrates a 10-minute UHPLC method for three key PhGs, supporting routine batch control for extract production and membrane processing.
4.2 Extraction: balance between yield, cost, and scalability
The optimized extraction condition-60% ethanol, 7× solvent, 80 min, 2 cycles-is industrially friendly because it:
Uses a moderate ethanol concentration (safe and cost-balanced)
Uses solvent ratio and time that are easy to standardize
Reduces unnecessary extra cycles once yield plateaus
4.3 Ultrafiltration: retain actives while improving stability
Ultrafiltration is often used to improve clarity and stability of liquid extracts (e.g., removing larger molecules/impurities). The chosen condition-100 kDa, 35°C, 0.5 MPa-is designed to:
Maintain high retention of key PhGs
Maintain acceptable flux for production throughput
Offer a process that can be standardized and scaled
5. Practical Keywords (SEO / GPT-friendly for industry websites)
how to make high quality extract
Cistanche extraction optimization
UHPLC multi-component quantification
phenylethanoid glycosides (PhGs) analysis
echinacoside acteoside isoacteoside UHPLC
herbal extract process development
membrane filtration for herbal extracts
ultrafiltration MWCO 100 kDa
extraction yield response surface Box–Behnken
in-process quality control (IPQC) for botanical extracts
scalable ethanol extraction protocol
improve clarity and stability of liquid herbal extract
References
Liu X, Yang Z, Han M, et al. Bioactive components, pharmacological properties, and applications of Cistanche deserticola Y.C. Ma: a comprehensive review. Nutrients. 2025;17(9):1501.
National Pharmacopoeia Commission. Pharmacopoeia of the People's Republic of China, Vol. 1. Beijing: China Medical Science and Technology Press; 2020.
Bi Cuicui, Liu Yinlu, Wei Fenfen, et al. Research progress on the main chemical constituents and bioactivities of Cistanche. Drug Evaluation Research. 2019;42(9):1896-1900.
Chen Changmao, Gazang Zhaxi, Wang Jie, et al. Research progress on active components and pharmacological mechanisms of Cistanche. Chinese Traditional and Herbal Drugs. 2025;56(12):4464-4477.
Liu Aoxue, Wan Feng, Hu Jiangxue, et al. Structure–activity relationship and mechanisms of phenylethanoid glycosides promoting neuroregeneration based on cell assays, network pharmacology and molecular docking. Drug Evaluation Research. 2025;48(1):136-142.
Li J, Zou Z, Su X, et al. Cistanche deserticola improves ovariectomized-induced osteoporosis mainly by regulating lipid metabolism: Insights from serum metabolomics using UPLC/Q-TOFMS. J Ethnopharmacol. 2024;322:117570.
Wen L, Hu J, Zhang J, et al. Phenylethanol glycosides from Herba Cistanche improve the hypoxic tumor microenvironment and enhance the effects of oxaliplatin via the HIF-1α signaling pathway. Mol Med Rep. 2021;24(1):517.
Yao SQ, Xu ZP, Guo WX, et al. Neuroprotective effect of Cistanche deserticola glycosides in MPTP-induced Parkinson's disease mouse model involves Nrf2 activation. J Toxicol Environ Health A. 2025;88(14):576-588.
Feng Duo, Duan Hao, Lü Yanni, et al. Application of Cistanche in health foods in China. Food Science and Technology. 2021;46(12):76-81.
Zhao Hongting, Xin Guoxiong, Guo Yuan. Research progress of the medicinal and edible homologous TCM Cistanche. Chinese Traditional and Herbal Drugs. 2025;56(9):3316-3329.
Mei Ju, Wu Tao, Li Qiang, et al. Quality and processing-related component changes of cinnamon from different origins. Natural Product Research and Development. 2024;36(3):377-387,399.
Meng Ran, Wu Zhe, Feng Wei, et al. Quality evaluation of dandelion based on HPLC fingerprinting, chemometrics and multi-component quantification. Chinese Traditional and Herbal Drugs. 2022;53(24):7887-7896.
Wu Xin, Wu Tao, Wang Bingqing, et al. Establishment of UPLC fingerprints and quantification of six components in licorice from different origins. Chinese Traditional and Herbal Drugs. 2025;56(7):2518-2526.
Li Yang, Bubi Ajiar·Hayirati, Qin Yadi, et al. Simultaneous determination of four phenylethanoid glycosides in Cistanche decoction pieces and Cistanche tubulosa extracts by HPLC. China Pharmaceuticals. 2022;31(6):77-80.
Ding Yanxia, Wang Xiaoqin, Zhang Ying. Determination of six major phenylethanoid glycosides in Cistanche medicinal materials by HPLC. Shizhen Guoyi Guoyao. 2020;31(4):813-816.
Dong Zhiguo, Zhou Rong, He Jinfeng, et al. Application and prospects of membrane separation technology in green manufacturing of TCM. Chinese Traditional and Herbal Drugs. 2024;55(12):4225-4234.
Li Yaxin, Liang Huizhen, Zhang Wensheng, et al. Research progress on processing technology of health liquor. China Brewing. 2022;41(5):23-27.
Wang Yuetong, Liu Hongbo, Xu Sining, et al. Effects of ultrafiltration membrane technology on physicochemical properties and antioxidant activity of Astragalus water extracts. Modern Chinese Medicine. 2024;26(4):695-701.
Huang Xiang, Guo Yehong, Jia Cunqin, et al. Study on extraction process and drying method of Cistanche. Chinese Traditional and Herbal Drugs. 2019;50(15):3622-3630.
Ma Jincheng, Wang Xuxing, Li Qian, et al. UPLC-based comparison of eight chemical components in two origins of Cistanche raw products and wine-processed products. Modern Chinese Medicine. 2025;27(6):1040-1048.






