Part Ⅱ Adverse Clinical Outcomes Among Inflammatory Bowel Disease Patients Treated For Urinary Tract Infection
May 19, 2023
Results
1. Patient Characteristics
Out of 23,566 ER visits due to a UTI for the first time at Sheba Medical Center between 2012 and 2018, 1758 patients under 18 yo were excluded. The remaining 21,808 patients comprised the study cohort. Of them, 122 patients had a previous diagnosis of IBD (CD—52, UC—70), while there were 21,686 non-IBD patients (Figure 1). There were 6599 ER visits of IBD patients between 2012 and 2018; thus, the rate of UTI among IBD patients was 1.9% in this period.
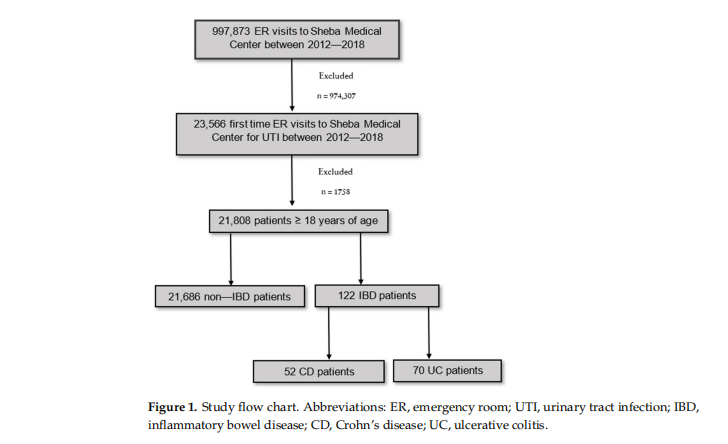
The baseline characteristics of patients with and without IBD are presented in Table 1. There was no difference in age (72.00 vs. 70.00 years, p = 0.351) or sex (p = 0.443) between the groups. BPH and urolithiasis were more prevalent among patients with IBD compared to patients without IBD (21% vs. 10%, p = 0.010; 11.5% vs. 3%, p < 0.001; respectively). On the other hand, urologic tumors and diabetes were slightly more common among the non-IBD group, but without statistical significance. The use of immunosuppressant and biological medications was more common among patients with IBD than patients without IBD. The IBD group had a higher rate of recent hospitalization (21% vs. 8%, p < 0.001), which was defined as hospitalization within three months before the index UTI ER visit.

The characteristics of CD and UC patients are shown in Table 2. Urolithiasis and BPH rates were similar between the groups (12% vs. 11%, p = 0.985; 21% vs. 21%; p = 0.950, respectively). The use of 5-ASA was more common among the UC group (25% vs. 56%, p = 0.001), while TNF-α inhibitors (21% vs. 4%, p = 0.004) and azathioprine (15% vs. 3%, p = 0.013) were more commonly used among CD patients. In total, 18 (35%) patients of the CD group and 7 (10%) patients of the UC group had a history of previous abdominal surgery as part of the management of disease natural history (data were missing for 18 and 36 patients, respectively). The rate of missing data regarding disease extent was 56% and 82% for CD patients and UC patients, respectively.

2. Microbiologic Characteristics
Figure 2 depicts the frequency distribution of uropathogens among the IBD group of 122 IBD patients, only 110 patients had an available urine culture result. Of them71 patients had a positive urine culture, and 39 patients had a negative urine culture without any pathogen detection (the missing data rate for urine culture was less than 10%)The most detected bacterium was E. coli (39%). Extended-spectrum beta-lactamase (ESBL)producing Enterobacteriaceae was grown on 17% of the urine cultures. Polymicrobial growth was detected in five urine cultures (7%). A total of 80 patients in the IBD group had an available blood culture result. Bacteria had grown on 13 of them, representing a positive blood culture (six-Escherichia coli; four-ESBL-producing Enterobacteriaceaetwo._Klebsiella Pneumonia oneEnterococcus faecalis).

3. Outcomes and Predictors of Outcomes
The outcomes of UTl among the cohort population are summarized in Table 3 Patients with IBD had a higher hospitalization rate compared to patients without IBD(68.9% vs. 59.3%, p = 0.032), while no difference in hospitalization duration was observed between the groups. Though the mortality rate within 30 days was almost equal between the groups, patients with IBD had worse secondary outcomes, such as higher rates of AKl(13.9% vs. 4.6%, p < 0.001) and 30-day recurrent hospitalization (15.6% vs. 7.3%, p = 0.001). No statistical significance was demonstrated between CD and UC patients regarding the pre-defined UTII outcomes. Performing sub-analyses of UTI outcomes across different age groups, we discovered UTI outcomes to be comparable among patients >70 with or without IBD. On the other hand, patients with IBD under 70 yo had higher rates of hospitalization (59.3% Vs. 44.5%, p = 0.030), AKI (16.7% vs. 1.7%, p < 0.001), and 30-day recurrent hospitalization (22.2% s. 5.7%, p < 0.001) compared to patients without IBD at the same age. Notably, among patients under 70 yo, there was a trend toward a higher rate of 30-day mortality in favor of patients with IBD compared to patients without IBD(3.7% Vs. 1.2%, p = 0.091).
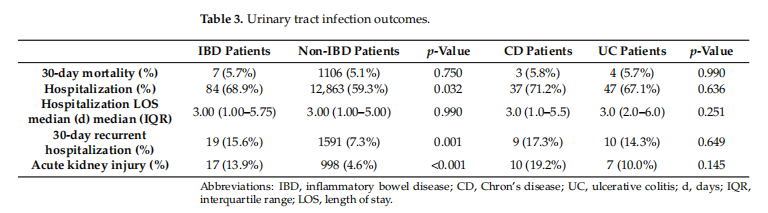
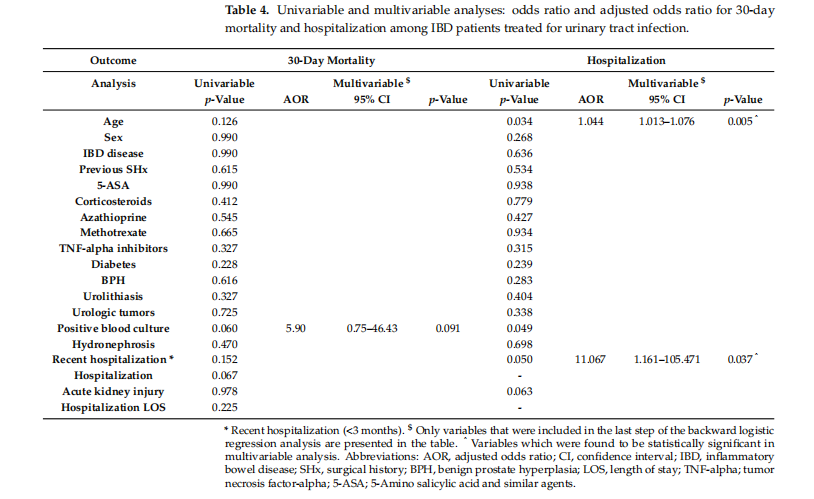
Tables 4 and 5 show the results of univariable and multivariable analyses related to the pre-defined UTI outcomes. We found advanced age (adjusted odds ratio (AOR) 1.044, 95% confidence interval (CI) 1.013–1.076, p = 0.005) and a history of recent hospitalization (AOR 11.067, 95% CI 1.161–105.471, p = 0.037) to be independently associated with increased risk of hospitalization among patients with IBD treated for UTIs. Similarly, we learned that the presence of hydronephrosis (AOR 10.383, 95% CI 2.039–52.865, p = 0.005), recent hospitalization (AOR 4.494, 95% CI 1.420–14.221, p = 0.011) and AKI (AOR 4.683, 95% CI 1.325–16.548, p = 0.017) were independently associated with the increased probability of 30-day recurrent hospitalization. Using multivariable analyses, we did not find any of the examined variables to be associated with increased mortality rate within 30 days or with AKI. Notably, no association was observed between a history of previous abdominal surgery and UTI outcomes.

Discussion
In this large, tertiary-center cohort, we examined the adverse outcomes of UTIs among patients with IBD compared to patients without IBD. We found higher rates of hospitalization, AKI, and 30-day recurrent hospitalization in the IBD group compared to the non-IBD group. We also discovered advanced age and history of recent hospitalization to be associated with an increased risk of hospitalization among patients with IBD treated for UTIs. History of recent hospitalization and urological complications such as hydronephrosis and AKI was associated with an increased risk of 30-day recurrent hospitalization in this group. To the best of our knowledge, this study is the first to evaluate outcomes and predictors of outcomes among patients with IBD treated for UTIs.
Although both UC and CD mainly involve the GI tract, extraintestinal manifestations (EIMs) are common in both IBD phenotypes [14]. These manifestations can affect almost any organ system, including the urinary system. Urolithiasis, a well-known risk factor for UTIs [15], is common among IBD patients—8–19% compared to only 0.1% in the general population, with a higher risk in CD patients [12,16]. Moreover, disease anatomic features such as perianal involvement [11] and entero-vesical fistulas [9] are associated with a higher risk for UTIs among patients with CD. Surprisingly, we found that UTI outcomes were comparable between UC patients and CD patients. This finding may be explained by the small size of each subgroup or by the compatible rates of urolithiasis between the groups (Table 2), though a previous study by L. Peyrin-Biroulet [11] et al. showed no significant difference between the groups as well. However, further research will probably clarify this issue.

Click here to know the benefits of Cistanche for Kidney
For the first time, our study demonstrated a higher rate of BPH in male patients with IBD compared to male patients without IBD [17]. Notably, no difference between baseline demographic characteristics (age and sex) of the IBD group compared to the non-IBD group was observed (Table 1). Next to urolithiasis, BPH is the most common cause of urinary outflow obstruction leading to UTIs [13]. Even though BPH was more prevalent among the IBD group, no association with adverse UTI outcomes among patients with IBD was demonstrated (Tables 4 and 5). Thus, we could assume that BPH did not influence the worse outcomes among the IBD group. Interestingly, previous studies showed a high prevalence of prostate cancer among IBD patients [17,18]. However, in our cohort, we noticed a similar rate of urological tumors between the groups. Further studies should be performed to confirm an association between IBD and BPH because this cohort might be biased by the selected diagnosis—UTI. Considering the high prevalence of BPH among the IBD group and the higher risk for hydronephrosis, obstructive AKI, and UTIs among patients with BPH, it seems reasonable to screen IBD patients for this medical condition. Early diagnosis could allow appropriate management (e.g., alpha receptor antagonists [13]) to prevent unwanted adverse outcomes.
Previous studies assessing the risk of AKI among patients treated for UTIs indicated that AKI occurs in 12.3–27.8% of the cases [19,20]. As mentioned, patients with IBD were more likely to develop AKI than patients without IBD. Not surprisingly, we found AKI as a predictor of increased risk for a 30-day recurrent hospitalization rate. AKI is associated with short-term complications such as life-threatening electrolyte abnormalities (e.g., hyperkalemia), metabolic acidosis, fluid overload, mechanical ventilation, the need for renal replacement therapy, and even mortality (16.2–23.8%) [21–23]. Moreover, patients with AKI are more likely to develop chronic kidney disease (CKD), and patients who have CKD may rapidly progress to end-stage renal disease following an episode of AKI [24–26]. The dangers are overemphasized among patients with IBD who have a high prevalence of urolithiasis, as noted above [10]. Additionally, urolithiasis is one of the most common causes of hydronephrosis. Consequently, patients with IBD are at risk of hydronephrosis [27,28] and post-renal AKI [29]. BPH is another cardinal risk factor accounting for hydronephrosis [29]. Thus, it seems crucial to detect any renal impairment early and, in particular, urine outflow obstruction in these patients. Given the virtues of ultrasonography (high availability, lack of ionized exposure risk, and its usefulness in the detection of flow obstruction [30,31]), we think that it is reasonable to perform a urinary system ultrasonography for each IBD patient admitted due to a UTI. Additionally, supporting the fluid status is an important measure to maintain normal renal function among patients with IBD treated for UTIs [22,23,32].
Cistanche pills
This study demonstrated a higher rate of hospitalization among the IBD group treated for UTI. Our findings are consistent with previous studies—J. Burisch et al. [33] described a higher rate of all-cause hospitalization among IBD patients, particularly in the early years of diagnosis. Additionally, in a previous study by our group about IBD patients treated for pneumonia, we found a higher rate of hospitalization, though no difference in adverse outcomes was observed [34]. This study indicates that patients with IBD who had had a history of recent hospitalization for any reason before a UTI episode had an increased risk of hospitalization and 30-day recurrent hospitalization for any reason. It may be explained by the various complications associated with hospitalization state (e.g., nosocomial infection [35,36], AKI [37], deep vein thrombosis [38], etc.) that may lead to a further one.
IBD treatments including TNF-α inhibitors, corticosteroids, and immunomodulators alter the immune system, leading to an increased risk for infectious disease [39]. These characteristics may be related to the results demonstrated in this study (Table 3). However, we did not find a significant association between IBD treatment agents and the primary or secondary outcomes (Tables 4 and 5). The latter may be explained by a modest sample size; otherwise, it is conceivable that other risk factors had a more significant impact on the prognosis. Moreover, corticosteroid use patterns vary among IBD patients (e.g., continuous, intermittent, or short-term, as needed by the patient [40]) and reliable follow-up is often unavailable. In our study, we could not trace those patterns based on its retrospective design. Further prospective research which will address this issue is needed to better explore it. We could not examine the effect of Vedolizumab on UTI outcomes, because no patient in the IBD group had been treated with it, based on retrospective data extraction. Further research may include Tofacitinib and Ustekinumab medications which had not commonly been used in our IBD group between 2012 and 2018.

Herba Cistanche and Cistanche extract
Our study has some limitations. First, UTI cases were extracted using the ICD-10 coding system based on electronic records. Typing errors might have contaminated the study population with other diseases (e.g., UTI cases that have been categorized as non-UTI cases by typing mistakes, and vice versa). This is true also in cases where the physician has not documented a precise diagnosis due to a lack of attention to this issue. Second, due to the retrospective design of this study, data regarding blood cultures and urine cultures of patients without IBD were not available; thus, we could not make a reliable comparison of microbiological characteristics between the IBD group and the non-IBD group at the same period and under the influence of the same regional antibiotic resistance profile. Therefore, we could not estimate the association between the IBD state and the probability to have bacteremia during a urinary tract infection episode, among other contributing factors. Third, we could not evaluate the association between IBD-related features and UTI outcomes, probably because of the small sample size in each subgroup (e.g., CD group and UC group); this was accentuated by substantial missing data regarding disease extent due to the retrospective nature of this study. Disease extent by Montreal Classification was extracted from free-text summaries following gastroenterologist visits. Because most of the IBD patients in the cohort were treated out of the gastroenterology department at Sheba Medical Center, this information was not available. Fourth, the IBD group in this study was relatively small. A larger sample size may have allowed us to better investigate predictors of mortality and other outcomes of UTI among patients with IBD. Fifth, as a tertiary medical center, patients who are referred to our ER may have a worse illness compared to community-treated patients. This selection bias might influence the study outcomes, but because the whole cohort (patients with and without IBD) was composed of patients who were referred to our ER, and the comparison was between the above-mentioned populations, we think it was well-balanced. Sixth, as mentioned above, we could not examine the effect of Vedolizumab, Tofacitinib, and Ustekinumab on UTI outcomes, because no patient was treated with these agents during the study period. Seventh, the prevalence of ER visits for UTIs was around 2% of all ER visits, among both IBD and non-IBD groups; this is lower than the results of a previously published study, which demonstrated that UTI cases account for 3.3–4% of the hospitalizations [11] of IBD and non-IBD patients. We think that conceivable explanations for that are the different denominators (ER visits vs. admissions) and the different local epidemiologic features. Unfortunately, due to the retrospective design of this study, data regarding the hospitalization rate among all causes of hospitalization in our institute were not available. Eighth, because of the retrospective design of this study, data regarding potential confounding factors might have been missing.
Conclusions
This is the first study to evaluate clinical outcomes among patients with IBD treated for UTIs. We demonstrated that in this population, UTIs encompass a greater risk for hospitalization, AKI, and re-hospitalization within 30 days. Therefore, renal function monitoring, fluid maintenance, and performing a renal ultrasonography scan to eliminate the existence of obstructive impairment should be considered. Notably, neither immunosuppressants nor biologics were found to influence UTI outcomes among IBD patients.

Standardized Cistanche
References
14. Guillo, L.; D’Amico, F.; Serrero, M.; Angioi, K.; Loeuille, D.; Costanzo, A.; Danese, S.; Peyrin-Biroulet, L. Assessment of extraintestinal manifestations in inflammatory bowel diseases: A systematic review and a proposed guide for clinical trials. United Eur. Gastroenterol. J. 2020, 8, 1013–1030.
15. Yongzhi, L.; Shi, Y.; Jia, L.; Yili, L.; Xingwang, Z.; Xue, G. Risk factors for urinary tract infection in patients with urolithiasis—Primary report of a single center cohort. BMC Urol. 2018, 18, 45.
16. Sato, S.; Sasaki, I.; Naito, H.; Funayama, Y.; Fukushima, K.; Shibata, C.; Masuko, T.; Ogawa, H.; Ueno, T.; Hashimoto, A.; et al. Management of Urinary Complications in Crohn’s Disease. Jpn. J. Surg. 1999, 29, 713–717.
17. Hammami, M.B.; Mahadevan, U. Men with Inflammatory Bowel Disease: Sexual Function, Fertility, Medication Safety, and Prostate Cancer. Am. J. Gastroenterol. 2020, 115, 526–534.
18. Ge, Y.; Shi, Q.; Yao, W.; Cheng, Y.; Ma, G. The association between in fl ammatory bowel disease and prostate cancer risk: A meta-analysis. Prostate Cancer Prostatic Dis. 2020, 23, 53–58.
19. Hsiao, C.-Y.; Chen, T.-H.; Lee, Y.-C.; Hsiao, M.-C.; Hung, P.-H.; Chen, Y.-Y.; Wang, M.-C. Urolithiasis Is a Risk Factor for Uroseptic Shock and Acute Kidney Injury in Patients With Urinary Tract Infections. Front. Med. 2019, 6, 288.
20. Hsiao, C.-Y.; Yang, H.-Y.; Hsiao, M.-C.; Hung, P.-H.; Wang, M.-C. Risk Factors for Development of Acute Kidney Injury in Patients with Urinary Tract Infection. PLoS ONE 2015, 10, e0133835.
21. Koza, Y. Acute kidney injury: Current concepts and new insights. J. INJ Violence Res. 2014, 8, 58–62.
22. Connell, A.; Laing, C. Acute kidney injury. Clin. Med. J. R. Coll. Phys. Lond. 2015, 15, 581–584.
23. Nagalingam, K. Acute Kidney Injury: The Hidden Killer in the Ward. J. Ren. Care 2020, 46, 72–73.
24. Wald, R.; Quinn, R.R.; Luo, J.; Li, P.; Scales, D.C.; Mamdani, M.M.; Ray, J.G.; the University of Toronto Acute Kidney Injury Research Group. Chronic dialysis and death among survivors of acute kidney injury requiring dialysis. JAMA J. Am. Med. Assoc. 2009, 302, 1179–1185.
25. Lo, L.J.; Go, A.S.; Chertow, G.M.; McCulloch, C.E.; Fan, D.; Ordoñez, J.D.; Hsu, C.Y. Dialysis-requiring acute renal failure increases the risk of progressive chronic kidney disease. Kidney Int. 2010, 76, 893–899.
26. Hsu, C.-Y.; Chertow, G.M.; McCulloch, C.E.; Fan, D.; Ordoñez, J.D.; Go, A.S. Nonrecovery of kidney function and death after acute on chronic renal failure. Clin. J. Am. Soc. Nephrol. 2009, 4, 891–898.
27. Okumus, M.; Inci, M.F.; Ozkan, F.; Bozkurt, S.; Sucakli, M.H.; Altunoluk, B. Correlation of volume, the position of the stone, and hydronephrosis with microhematuria in patients with solitary urolithiasis. Med. Sci. Monit. 2013, 19, 295–299.
28. Sasmaz, M.I.; Kirpat, V. The relationship between the severity of pain and stone size, hydronephrosis and laboratory parameters in the renal colic attack. Am. J. Emerg. Med. 2019, 37, 2107–2110.
29. Sacks, S.H.; Aparicio, S.A.; Bevan, A.; Oliver, D.O.; Will, E.J.; Davison, A.M. Late renal failure due to prostatic outflow obstruction: A preventable disease. BMJ 1989, 298, 156–159.
30. Gottlieb, R.H.; Voci, S.L.; Cholewinski, S.P.; Hartley, D.F.; Rubens, D.J.; Orloff, M.S.; Bronsther, O.L. Sonography: A Useful Tool to Detect the Transplant Dysfunction. J. Clin. Ultrasound. 1999, 27, 325–333.
31. Havard, J.D.J. Regular Review Ultrasonography in the diagnosis of renal obstruction. BMJ 1990, 301, 944–946. [CrossRef] 32. Bellomo, R.; Kellum, J.A.; Ronco, C. Acute kidney injury. Lancet 2012, 380, 756–766.
33. Burisch, J.; Jess, T.; Martinato, M.; Lakatos, P.L. The burden of inflammatory bowel disease in Europe. J. Crohns Colitis 2013, 7, 322–337.
34. Ukashi, O.; Barash, Y.; Segel, M.J.; Ungar, B.; Soffer, S.; Ben-Horin, S.; Klang, E.; Kopylov, U. Predictors of mortality in inflammatory bowel disease patients treated for pneumonia. Ther. Adv. Gastroenterol. 2020, 13, 1756284820939453.
35. Kaye, K.S.; Anderson, D.J.; Cook, E.; Huang, S.S.; Siegel, J.D.; Zuckerman, J.M.; Talbot, T.R. Guidance for Infection Prevention and Healthcare Epidemiology Programs: Healthcare Epidemiologist Skills and Competencies. Infect. Control Hosp. Epidemiol. 2015, 36, 369–380.
36. Kollef, M.H. Health Care—Associated Pneumonia: Perception versus Reality. Clin. Infect. Dis. 2009, 49, 1875–1877.
37. Wang, H.E.; Muntner, P.; Chertow, G.M.; Warnock, D.G. Acute kidney injury and mortality in hospitalized patients. Am. J. Nephrol. 2012, 35, 349–355.
38. Heit, J.A.; Melton, L.J.; Lohse, C.M.; Petterson, T.M.; Silverstein, M.D.; Mohr, D.N.; O’Fallon, W.M. Incidence of venous thromboembolism in hospitalized patients vs community residents. Mayo Clin. Proc. 2001, 76, 1102–1110.
39. Grace, R.; Bownik, H.; Scott, F.; Lichtenstein, G. Infectious Complications in IBD Patients on Immunomodulators, Corticosteroids, and Vedolizumab: Is Older Age a Predictor of Higher Complication Rates or Worsened Response?: 1940. Am. J. Gastroenterol. 2015, 110, S823.
40. Waljee, A.K.; Wiitala, W.L.; Govani, S.; Stidham, R.; Saini, S.; Hou, J.; Feagins, L.A.; Khan, N.; Good, C.B.; Vijan, S.; et al. Corticosteroid use and complications in a US inflammatory bowel disease cohort. PLoS ONE 2016, 11, e0158017.
Offiir Ukashi 1,2,3, Yiftach Barash 3,4,5, Eyal Klang 3,4,5, Tal Zilberman 3,6, Bella Ungar 1,3, Uri Kopylov 1,3, Shomron Ben-Horin 1,3 and Ido Veisman 1,3
1. Department of Gastroenterology, Sheba Medical Center, Tel Hashomer, Ramat Gan 52620, Israel; bella.geyshis.ungar@gmail.com (B.U.); ukopylov@gmail.com (U.K.); shomron.benhorin@gmail.com (S.B.-H.); idoweiss37@gmail.com (I.V.)
2. Department of Internal Medicine A, Sheba Medical Center, Tel Hashomer, Ramat Gan 52620, Israel
3. Sackler School of Medicine, Tel-Aviv University, Tel Aviv-Yafo 67011, Israel; yibarash@gmail.com (Y.B.); eyalkla@hotmail.com (E.K.); ztaltal@gmail.com (T.Z.)
4. Department of Diagnostic Imaging, Sheba Medical Center, Tel Hashomer, Ramat Gan 52620, Israel
5. DeepVision Lab, Sheba Medical Center, Tel Hashomer, Ramat Gan 52620, Israel
6. Infectious Disease Unit, Sheba Medical Center, Tel Hashomer, Ramat Gan 52620, Israel






