New Prediction Model Can Help Patients With IgA Nephropathy Be Alert To The Risk Of Hyperuricemia in Advance
May 19, 2023
IgA nephropathy (IgAN) is currently considered to be one of the most common primary glomerular diseases worldwide, accounting for 30% of primary glomerulonephritis. It is one of the main causes of end-stage renal disease (ESRD). Hyperuricemia refers to elevated blood uric acid levels due to purine metabolic disorders and uric acid metabolic disorders. Compared with the general population, IgAN patients have a higher incidence of hyperuricemia and a worse prognosis. Therefore, it is of great scientific and clinical significance to determine the risk factors associated with the progression of IgAN with hyperuricemia.
Click to cistanche tubulosa extract for kidney disease
Existing studies only explored the influencing factors of hyperuricemia, but failed to fully evaluate the weight of specific factors. This study aims to explore the risk factors affecting IgAN hyperuricemia, establish a risk prediction model, provide a scientific basis and reference for early identification of high-risk groups of hyperuricemia, and provide a basis for clinically targeted diagnosis and treatment measures.
Method
The baseline data of IgAN patients were obtained from the electronic medical record system of the First Affiliated Hospital of Zhengzhou University Hospital. A total of 1184 IgAN patients were included according to the following inclusion and exclusion criteria, and their demographic and clinicopathological characteristics were collected.
Inclusion criteria:
(1) From January 2012 to June 2018, renal biopsy was diagnosed as IgAN;
(2) At least 18 years old;
(3) Complete data.

Exclusion criteria:
(1) There is no MEST-C pathological score available, or baseline estimated glomerular filtration rate (eGFR) data is missing;
(2) Before the start of the study, use corticosteroids and immunosuppressants for treatment, and take uric acid-lowering drugs and drugs that affect blood uric acid, such as diuretics, β-blockers, etc.;
(3) Secondary IgAN: including systemic lupus erythematosus, Henoch-Schonlein purpura, ankylosing spondylitis, psoriasis, liver disease, and tumors.
Seven out of ten patients were randomized to the training cohort (n=841) and three out of ten patients were randomized to the validation cohort (n=343). Univariate and multivariate logistic regression analyzes were used to screen risk factors for hyperuricemia and identify independent predictors.

The area under the receiver operating characteristic curve (AUC) was used to distinguish true positives from false positives in the hyperuricemia risk map. To evaluate its calibrating ability, a calibration curve was drawn. Decision curve analysis (DCA) was used to determine its clinical utility by calculating the net benefit of a hyperuricemia risk model in IgAN patients at different threshold probabilities.
Result
1. Screening predictors
Twelve potential predictors were selected by univariate Logistic regression analysis, and then 8 potential predictors were further screened by multivariate Logistic regression analysis, including gender, hypoalbuminemia, hypertriglyceridemia, blood Blood urea nitrogen (BUN), glomerular filtration rate (eGFR), 24-hour urine protein (24-hour TP), total hematuria (BUN), and tubular atrophy/interstitial fibrosis (T) (Table 1).
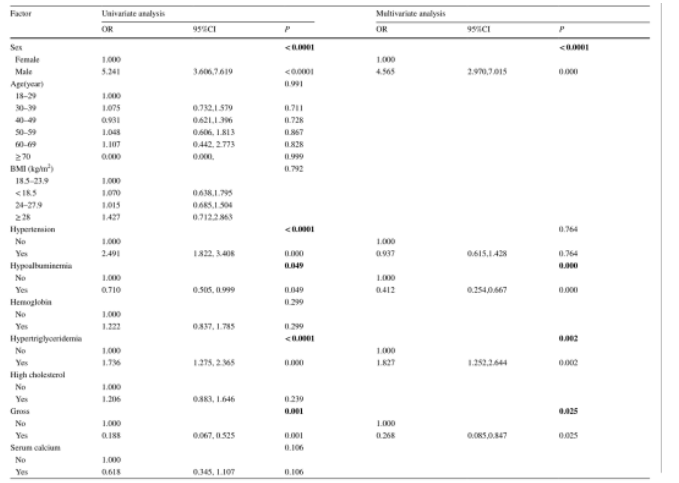

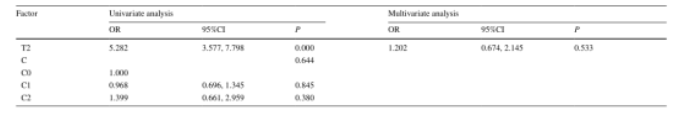
Table 1 Univariate and multivariate Logistic regression analysis using training cohort data
2. Prediction model establishment
Based on the above results, a prediction model for the risk of hyperuricemia in IgAN patients was constructed (Figure 1). The AUC of the nomogram to predict the risk of hyperuricemia was 0.834 (95% CI 0.804–0.864) (Fig. 2A), and the AUC of the validated AUC was 0.787 (95% CI 0.736–0.839) (Fig. 2B), indicating the predictive performance of the model Medium, with good recognition. Decision curve analysis showed that the hyperuricemia risk nomogram has clinical application value (Fig. 2E, Fig. 2F).

Figure 1 Nomogram for predicting the risk of hyperuricemia in IgAN patients
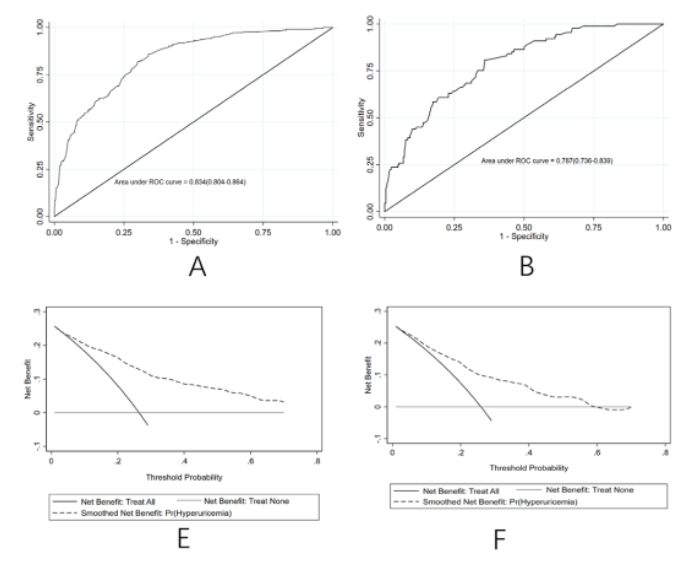
Figure 2 Curves of training queue and validation queue
A. ROC curve for the training cohort B. ROC curve for the validation cohort C. Decision curve analysis for the training cohort D. Decision curve analysis for the validation cohort
Discussion
At present, many studies have shown that hyperuricemia will aggravate the prognosis of IgAN. Therefore, we should pay more attention to the risk factors of IgAN patients with hyperuricemia and establish a comprehensive risk prediction model of hyperuricemia for early intervention, delay disease progression, and prevent the occurrence of ESRD.
Compared with previous studies, this study provides a complete set of prediction models for IgAN with hyperuricemia and verifies its feasibility. In addition, the sample size of this study is larger than previous studies, which provides a more valuable reference for clinical practice.
However, the current research still has certain limitations. First, the nomogram model was developed and validated by retrospective analysis of clinicopathological data from a single institution, which may have led to some bias in the conclusions. Secondly, this study did not conduct statistical analysis on the patient's eating habits, exercise patterns, and other indicators, and other important parameters may be missed. Finally, included patients may have an unavoidable bias in the results due to different treatment regimens. Therefore, before the nomogram model can be widely used, it needs to use data from multiple centers and conduct external validation to further evaluate its accuracy and reliability.

In conclusion, the nomogram developed in this study can effectively predict the risk of hyperuricemia in IgAN patients based on clinicopathological factors. This practical new approach provides strong support for the assessment of hyperuricemia risk in IgAN patients.
How does Cistanche treat kidney disease?
Cistanche is an herb that has been used for centuries in traditional Chinese medicine to treat various health conditions, including kidney disease. It is believed to help improve kidney function by increasing blood flow to the kidneys and promoting the regeneration of kidney cells. Cistanche contains several active compounds, including phenylethanoid glycosides, iridoids, and polysaccharides, which have been shown to have beneficial effects on the kidneys. These compounds have anti-inflammatory and antioxidant properties that help protect the kidneys from damage caused by inflammation and oxidative stress. Additionally, cistanche has been found to help regulate blood pressure and improve blood sugar control, both of which are important factors in the development and progression of kidney disease. Overall, cistanche is a natural and effective way to support kidney health and may be beneficial for those with kidney disease or at risk of developing it.
Reference:
Geng Y H, Zhang Z, Zhang J J, et al. Established the first clinical prediction model regarding the risk of hyperuricemia in adult IgA nephropathy[J]. Int Urol Nephrol, 2023 Feb 08.






