Plant Microbiome: An Ocean Of Possibilities For Improving Disease Resistance in Plants Part 1
Jun 09, 2023
Abstract:
Plant diseases pose a serious threat to crop production and the agricultural economy across the globe. Currently, chemical pesticides are frequently employed to combat these infections, which cause environmental toxicity and the emergence of resistant pathogens. Moreover, the genetic manipulation of plant defense pathways and the breeding of resistant genes has attained limited success due to the rapid evolution of pathogen virulence and resistance, together with host range expansion. Additionally, due to climate change and global warming, the occurrence of multiple stresses during disease outbreaks has further impacted overall crop growth and productivity, posing a serious threat to food security. In this regard, harnessing the plant-beneficial microbiome and its products can provide novel avenues for disease resistance in addition to boosting agricultural output, soil fertility, and environmental sustainability.
The relationship between disease outbreak period and immunity is very close. During a disease outbreak, many people become infected because their immunity is not strong enough to fight off the invading virus or bacteria. Immunity is the main line of defense of the human body against diseases. It can help the body resist the invasion and reproduction of germs. If the immunity is insufficient, the human body will be susceptible to diseases.
During a disease outbreak, we can reduce the risk of infection by strengthening our immunity. Ways to strengthen your immune system include paying attention to your diet, getting enough sleep, exercising, and avoiding stress and anxiety. In addition, according to the epidemic situation, timely vaccination is also an effective way to strengthen your immunity.
In short, the relationship between disease outbreaks and immunity is inseparable. Only by strengthening our immunity can we better protect ourselves from disease. Therefore, we need to improve immunity. Cistanche can significantly improve immunity. Cistanche also has anti-virus and anti-cancer effects, which can strengthen the immune system’s ability to fight and improve the body’s immunity.

Click cistanche deserticola supplement
In plant–beneficial microbiome interactions, induced systemic resistance (ISR) has emerged as a key mechanism by which a beneficial microbiome primes the entire plant system for better defense against a wide range of phytopathogens and pests. In this review, we provide the recent developments on the role of plant-beneficial microbiomes in disease resistance. We also highlight knowledge gaps and discuss how the plant immune system distinguishes pathogens and beneficial microbiota.
Furthermore, we provide an overview of how immune signature hormones, such as salicylic acid (SA), jasmonic acid (JA), and ethylene (ET), shape plant-beneficial microbiomes. We also discuss the importance of various high-throughput tools and their integration with synthetic biology to design tailored microbial communities for disease resistance. Finally, we conclude by highlighting important themes that need future attention to fill the knowledge gaps regarding the plant immune system and plant-beneficial-microbiome-mediated disease resistance.
Keywords:
pathogens; immunity; microbiome; hormones; induced systemic resistance; SynComs.
1. Introduction
Plants are constantly challenged by different microbial pathogens that endanger their survival and pose a constant threat to global food security. In the past, numerous disease outbreaks have significantly impacted our agricultural output and economy, especially in countries with food shortages [1]. The Irish potato famine in the nineteenth century was caused by Phytophthora infestans, which resulted in over two million fatalities and widespread migration from Ireland [2]. Similarly, Cochliobolus miyabeanus, causing brown spots on rice, was another significant plant disease and a disastrous epidemic that claimed the lives of over two million people. Food crops suffer significant yield losses worldwide due to microbial diseases and pests, with mean losses of 30.3% for rice, 22.6% for maize, 21.5% for wheat, 21.4% for soybeans, and 17.2% for potatoes [1].
Plant diseases can reduce crop yields by 50% in some regions, primarily among smallholder farmers, leading to severe economic challenges [3]. Plant diseases also hurt species diversity, downstream costs associated with control methods, and human health [3]. Emerging plant diseases and pest outbreaks have a major economic impact on agriculture, affecting food security, national security, and human health [4]. In the future, it is anticipated that variations in the geographic distribution of pathogens in response to climate change and growing global commerce would make emerging plant diseases more prevalent and severe [5,6]. Recently, coffee rust outbreaks caused by Hemileia vastatrix in Central America have also caused huge yield losses and economic crises [7].
Unlike endemic diseases, which can be managed, emerging diseases can have severe consequences on contemporary agricultural and other input systems, necessitating prompt mitigation strategies. Currently, a worldwide epidemic is endangering the health of millions of people worldwide. Hence, the availability of nutritious and healthy food is critical to helping people escape poverty and achieve better health outcomes.
Over the last two decades, scientific breakthroughs have greatly benefited our efforts to manage plant infections. For example, genetically modifying plant immune components and breeding resistant genes are a few strategies that have been used to combat plant pathogens [8]. However, severe disease outbreaks are exacerbated by the rapid development of pathogen virulence and resistance, as well as host range expansion mainly under modern agricultural practices, which pose serious challenges to developing long-term disease-resistant cultivars. On the other hand, the application of chemical microbicides and fungicides has been partly successful, but their overuse hurts the environment, humans, and the emergence of newly resistant pathogens [9].
For instance, a
few examples of fungicide-resistant plant pathogens that represent a substantial danger
to commercially significant crops include Botrytis cinerea, Alternaria sp., Plasmopara viticola,
Pseudocercospora fijiensis, Ramularia collo-cygni, and powdery mildew pathogens [10]. Pesticides can also have an impact on the beneficial microbiota, which normally protects plants
from pathogens by competing with them, inhibiting their colonization, activating plant
immunological pathways, or secreting antimicrobial compounds [11]. At present, the rise
of multi-pesticide-resistant pathovars is one of the most serious negative consequences
of pesticide overuse in modern agriculture.
Due to the limitations of current mitigation
strategies for controlling disease outbreaks, there is a need to search for another alternative
that is more effective and eco-friendly. In this context, harnessing the potential of plant-beneficial microbiomes and their products is a viable strategy for the mitigation of plant
diseases in sustainable agriculture, owing to the multifaceted functions that they provide
to their hosts, such as promoting growth, providing nutrient availability, increasing soil
fertility, being eco-friendly and boosting multiple stress resilience. Here, we focus on the
recent developments on the role of plant-beneficial microbiomes and disease resistance. We
also discuss how plant immune signatures shape plant beneficial microbiomes. Furthermore, we provide an overview of how plant defense systems interact with beneficial and
pathogenic microbes. Recent studies have shown that individual- and community-level
features of plant microbiomes boost plant immunity in different crop systems.
2. Plant Microbiome and Disease Resistance: A New Sustainable Approach for Controlling Emerging Disease Outbreaks
Plants in nature coexist with diverse microbial communities that can be beneficial, commensal, and pathogenic. Plants interact with these diverse microbes in three key regions: the phyllosphere, endosphere, and rhizosphere. The plant microbiome comprises a variety of elegant species, including fungi, bacteria, protozoa, archaea, and viruses. Plant microbiomes can be beneficial in a variety of ways, including protecting the plant from harmful infections, improving tolerance to a wide range of abiotic stresses, increasing growth, health, and production, and giving plants a competitive advantage in response to climatic changes [12–14]. However, the complex and dynamic interactions between plants and microbiomes are greatly influenced by the host, microbe, and environment, forming a trio complex that influences their overall outcome [14].
The plant microbiome plays a multifaceted role in protecting plants from pathogen attacks using different strategies, such as activating immune responses, induced systemic resistance (ISR), and callose deposition. They are also responsible for the production and excretion of the following: antimicrobial compounds, such as 2,4-diacetyl phloroglucinol, proteases, chitinases, bacteriocins, and siderophores; lipopeptides, such as iturin A, bafilomycin D, and mycosubtilin; and volatile compounds. Moreover, they also inhibit pathogens by competition for nutrients and space, as shown in Figure 1. Interestingly, in plant–beneficial microbiome interactions, ISR has emerged as a key mechanism by which the beneficial microbiome primes the entire plant system for better defense against a wide range of phytopathogens and pests [15]. ISR is a generic term used for the induced resistance mechanism stimulated by both chemical and biological inducers that shield nonexposed plant organs against subsequent attacks by pathogenic bacteria and herbivorous pests.

Generally, ISR is regulated by a complex of interconnected signaling cascades in which plant hormones such as JA, ET, and SA, as well as their crosstalk, play an important regulatory role. However, in the majority of plant–beneficial microbiome interactions, ISR is predominantly regulated by the JA and ET hormonal cascades. This has been further proved in both mutant JA/ET and wild Arabidopsis plants, in which beneficial bacteria, such as Pseudomonas fluorescens WCS417r–ISR, Serratia marcescens, Pseudomonas protegens CHA0 and Pseudomonas fluorescens Q2-87, and beneficial fungi, such as Penicillium sp. GP16-2 and Trichoderma harzianum T39, trigger ISR in a JA/ET-dependent manner [16–19]. However, there are a few reports that show that beneficial microbes also trigger ISR in an SA-dependent manner. For example, beneficial bacteria such as Pseudomonas aeruginosa 7NSK2 fail to trigger ISR in SA mutant tomato plants [20].
As a result, it would be interesting to investigate how plant-beneficial microbiomes activate ISR because plant microbiomes contain a variety of microflora that are adapted to various lifestyles. Therefore, more research is needed to determine how JA/ET and SA hormones, as well as their interactions, play a role in microbiome-mediated ISR activation. In plants, beneficial bacteria, pathogens, and insects all share the ISR-regulated linked signaling pathways. Consequently, more research is required to pinpoint the key participants in both pathogen- and microbiome-triggered ISR, which can reveal whether they utilize similar or dissimilar signaling or gene networks.
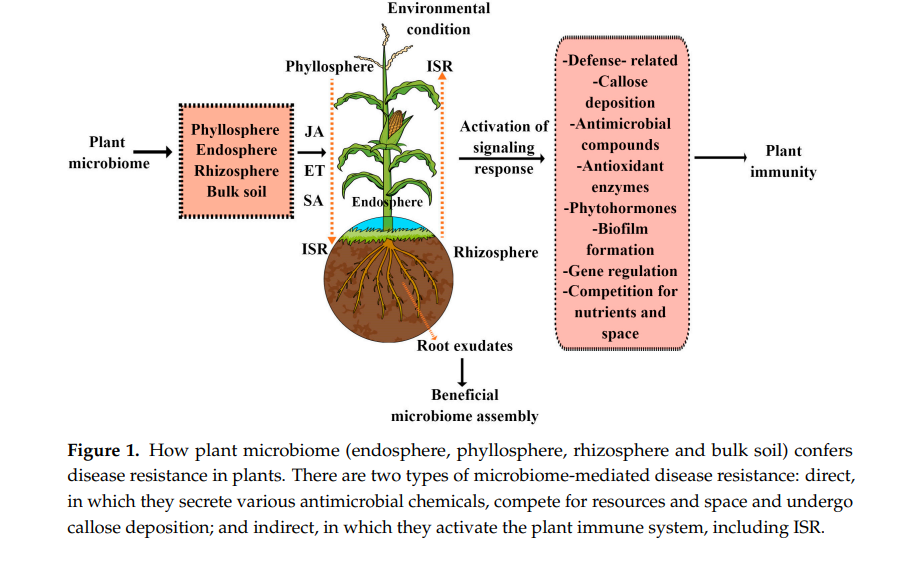
Many studies have reported that plant microbiota confers disease resistance against different pathogens of different lifestyles. For example, the activation of innate immunity in plants via root microbiota has been extensively characterized to provide resistance against numerous above-ground plant diseases via ISR [15,21]. ISR activation has been reported as an important driver of most plant-growth-promoting bacteria (PGPR)-mediated disease resistance in plants [15,22]. Previous studies have shown that bacterial and oomycete infections in Arabidopsis leave influence the root exudates to promote the assemblage of beneficial ISR-triggering microorganisms [23,24].
In plants, root exudates play a major role not only by shaping the beneficial microbiome but also by providing disease resistance by directly inhibiting pathogen growth or activating the plant immune system. Plants secrete diverse root exudates, such as organic acids, vitamins, flavonoids, polysaccharides, amino acids, and sugars that directly or indirectly confer host disease resistance. Many beneficial bacteria actively respond to root exudates by adjusting their transcriptional program toward traits involved in root colonization, activating ISR, chemotaxis, biofilm formation, and energy metabolism which are important for inhibiting pathogens and disease progression. Although the role of root exudates in shaping the plant microbiome is well addressed, how they trigger the immune system is largely unknown.
A growing body of studies on Triticum viaticum [25,26], Arabidopsis thaliana, and Beta vulgaris [27] has revealed that pathogen-infected plants’ roots can attract helpful bacteria to rescue or safeguard future generations. Endosphere and rhizosphere microbiome members have also been found to inhibit plant diseases, such as the damping-off and take-all [28–30]. Similarly, Kwak et al. [31] reported that rhizospheric microbiota in tomato plants enhance disease resistance against wilt disease caused by Ralstonia solanacearum. Mendes et al. [32] also reported that rhizobacteria from the Pseudomonadaceae, Bacillaceae, Solibacteraceae, and Cytophagaceae families are more prevalent in the rhizosphere of Fusarium-resistant bean cultivars. Similarly, another study revealed the role of the beneficial microbiome in mitigating the negative effects of potato scab disease, identifying key genera such as Geobacillus and Curtobacterium [33]. Numerous studies have shown that the beneficial microbiome directly suppresses pathogens through a variety of strategies, including the synthesis of antimicrobial compounds, hyperparasitism, and competition for nutrients and space [34,35]. All of these strategies result in pathogen limitation but may vary from microbe to microbe. For example, Fusarium-vulnerable cucumber plants often assemble helpful microorganisms, such as Comamonadaceae and Xanthomonadaceae, to inhibit the Fusarium pathogen by producing higher levels of organic acids [36]. A previous study reported that Pseudomonas fluorescens WCS417 promotes callose deposition at the pathogen entry site and also triggers the expression of defense signature genes (pathogenesis-related genes) that restrict pathogen entry and disease progression [37].
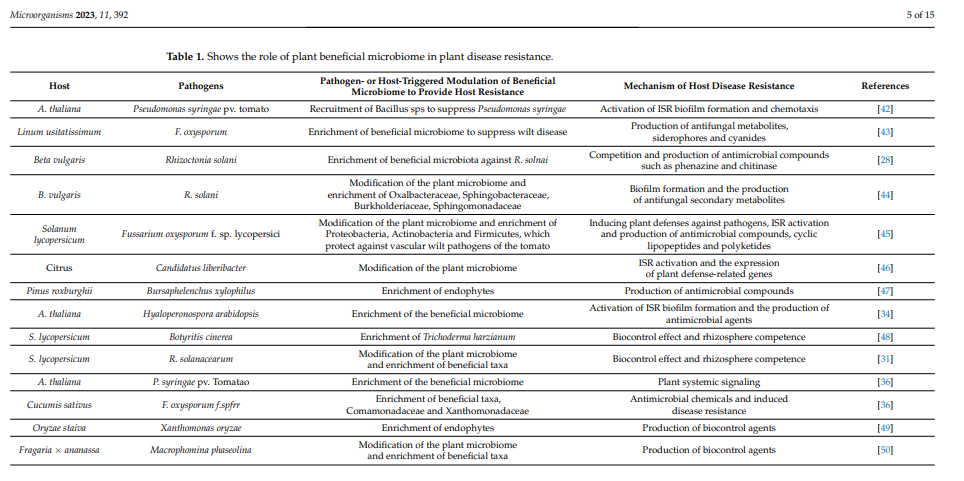
Recently, a seed-endophytic Sphingomonas melonis strain was reported to provide disease resistance against Burkholderia plantarii, which causes seedling blight in rice [38]. Arbuscular mycorrhiza fungi (AMF) have also been shown in numerous studies to increase plant resilience to a variety of diseases. For example, mycorrhiza colonization in different crop plants provides disease resistance against numerous phytopathogens, such as Pyrenochaeta terrestris, Fusarium oxysporum f. sp. Lycopersici, Phytophthora nicotianae var. parasitic, P. parasitica and Pseudomonas syringae [39–41]. Additionally, AMF activates ISR and systemic acquired resistance (SAR), which provides disease resistance to pathogens from various lifestyles. Deciphering how AMF and a beneficial microbiome work together can therefore provide novel perspectives on plant disease resistance in sustainable agriculture. Furthermore, we summarize the role of plant-beneficial microbiomes and their products in disease resistance in Table 1.
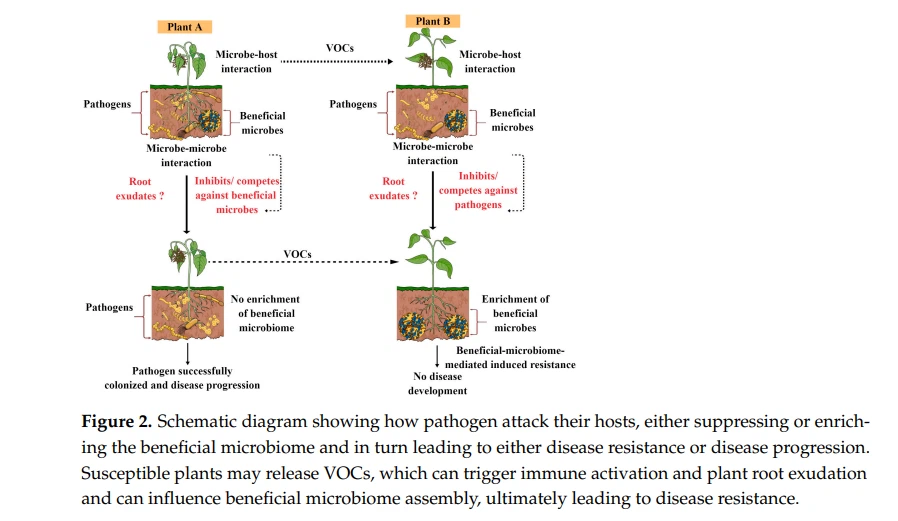
Recently, a new hypothesis has emerged in plants in the context of stress and plant-beneficial microbiome recruitment, known as the ‘cry for help’. According to this cry for help hypothesis, plants actively recruit or enrich specific microorganisms during stress conditions that can protect them from detrimental effects and that can provide an array of growth-promoting benefits. This hypothesis was further studied under diverse stressful conditions in different plants, and the results were astounding, demonstrating how plants recruit their beneficial microbiomes to fend off pathogen attacks and gain further growth benefits from them [26]. In this review, we made one model showing how pathogen attacks in their hosts either suppress or enrich the beneficial microbiome, which in turn leads to either disease resistance or disease progression, as shown in Figure 2. However, many factors govern the overall success or failure of the plant-beneficial microbiome and pathogen battles.
For instance, the lifestyle of plant pathogens, plant species, environmental factors, root exudate chemistry, the plant immune system, and signatures can have a significant effect (both individually and in combination) on plant-beneficial microbiome and pathogen interactions. We are still in the early stages of understanding the complexity of plant-beneficial microbiomes and host disease resistance. Hence, future research should concentrate on determining how the plant-beneficial microbiome influences disease resistance in both susceptible and resistant cultivars and how it varies between the two, which can provide novel opportunities to develop disease-resistant cultivars and control future disease outbreaks.

Moreover, it would be interesting to explore how susceptible plants release volatile compounds (VOCs) during pathogen attacks or disease development that can trigger the plant immune system or microbiome assembly in neighboring plants, making them alert and resistant to desired pathogen attacks. VOCs function as airborne signals in plant–plant or plant–microbe communication, which can assist plants in developing and coping with stress in both direct and indirect ways. It is well documented that plants release a wide range of volatile organic molecules when they become infected by pathogenic microorganisms, such as aromatics, terpenes, fatty acid derivatives, and nitrogen-containing compounds, as well as volatile phytohormones, methyl jasmonate, and methyl salicylate, which seem to provide disease resistance either directly or indirectly [51]. In the future, the integration of analytical and molecular tools is required to decipher the role of VOCs produced by plants or the beneficial microbiome in disease resistance in different crop pathology systems. Moreover, how VOCs from plants and beneficial microbiomes share or differ in triggering the plant immune system is one of the most exciting research areas in the field of plant microbiomes. Microorganisms 2023, 11, x FOR PEER REVIEW 6 of 15 recruit their beneficial microbiomes to fend off pathogen attacks and gain further growth benefits from them [26]. In this review, we made one model showing how pathogen attacks in their hosts either suppress or enrich the beneficial microbiome, which in turn leads to either disease resistance or disease progression, as shown in Figure 2. However, many factors govern the overall success or failure of the plant-beneficial microbiome and pathogen battles.
For instance, the lifestyle of plant pathogens, plant species, environmental factors, root exudate chemistry, the plant immune system, and signatures can have a significant effect (both individually and in combination) on plant-beneficial microbiome and pathogen interactions. We are still in the early stages of understanding the complexity of plant-beneficial microbiomes and host disease resistance. Hence, future research should concentrate on determining how the plant-beneficial microbiome influences disease resistance in both susceptible and resistant cultivars and how it varies between the two, which can provide novel opportunities to develop disease-resistant cultivars and control future disease outbreaks. Moreover, it would be interesting to explore how susceptible plants release volatile compounds (VOCs) during pathogen attacks or disease development that can trigger the plant immune system or microbiome assembly in neighboring plants, making them alert and resistant to desired pathogen attacks. VOCs function as airborne signals in plant–plant or plant–microbe communication, which can assist plants in developing and coping with stress in both direct and indirect ways.

It is well documented that plants release a wide range of volatile organic molecules when they become infected by pathogenic microorganisms, such as aromatics, terpenes, fatty acid derivatives, and nitrogen-containing compounds, as well as volatile phytohormones, methyl jasmonate, and methyl salicylate, which seem to provide disease resistance either directly or indirectly [51]. In the future, the integration of analytical and molecular tools is required to decipher the role of VOCs produced by plants or the beneficial microbiome in disease resistance in different crop pathology systems. Moreover, how VOCs from plants and beneficial microbiomes share or differ in triggering the plant immune system is one of the most exciting research areas in the field of plant microbiomes.
For more information:1950477648nn@gamil.com






