Preliminary Findings Of Elevated Inflammatory Plasma Cytokines in Children With Autism Who Have Co-Morbid Gastrointestinal Symptoms Part 2
Jul 25, 2023
2.2. Statistical Analyses
Cytokine and chemokine concentrations were natural-log-transformed for statistical analysis. Covariates of interest as possible confounders included the child’s age at the blood draw. Immune responses can change with age; thus, it is important to adjust for age as a confounder. Analyses of covariance (ANCOVA), adjusted for age at blood draw, were performed to compare analyte concentrations in cases and controls with and without GI symptoms.
Adjusted means and standard errors were presented as exponentiated values in pg/mL units. p-values were corrected for multiple comparisons using the Tukey–Kramer method and considered statistically significant if p < 0.05 after the corrections were applied. All analyses were performed using SAS version 9.2 (SAS Institute Inc., Cary, NC, USA).
Cytokines are important proteins in the body that can affect the growth, differentiation, and function of every cell in the body. Immunity in the body is also closely related to cytokines.
Immunity is the body's resistance to germs, viruses, and other pathogens. When the body is invaded by pathogens from the outside world, the immune system will produce a variety of cells and substances to protect the body. These substances include many cytokines, which can regulate the activity of white blood cells in the immune system, promote the production of antibodies, and thus enhance the body's immunity.
Cytokines can also act directly on pathogens, activating macrophages and NK cells to attack and kill pathogens. In addition, cytokines can also participate in the regulation of inflammatory response and immune suppression, and play an important role in the occurrence and development of autoimmune diseases.
Therefore, adequate cytokines play a very important role in the body's immunity. Scientists are also studying ways to use cytokines to enhance the body's immunity. Some immune modulators and immune enhancers have been widely used in immunotherapy.
In conclusion, cytokines are closely related to immunity, the development of immunity requires reasonable regulation of cytokines, and sufficient cytokines can also enhance immunity. Therefore, we should actively focus on maintaining the balance of cytokines in our body to maintain our physical health and immunity. From this point of view, we need to improve our immunity. Cistanche can significantly improve immunity because the polysaccharides in Cistanche can regulate the immune response of the human immune system, improve the stress ability of immune cells, and enhance the bactericidal effect of immune cells.

Click health benefits of cistanche
3. Results
We found no differences in any cytokines between TD controls who had GI symptoms and those TD controls that did not have GI symptoms, after adjusting for the child’s age at blood draw (Table 1).
Among AU cases, compared to those without GI symptoms those with GI symptoms had significantly higher innate immune cytokines, including; IFNα-2 levels (adjusted mean 86.574 (standard error 1.234) vs. 38.092 (1.241) pg/mL, p = 0.04); IL-1α levels (23.999 (1.496) vs. 5.028 (1.513) pg/mL, p = 0.04); TNFβ (21.672 (1.349) vs. 5.094 (1.358) pg/mL, p = 0.006); and, IL-15 (3.561 (1.405) vs. 0.690 (1.418) pg/mL, p = 0.006).
For cytokines mostly associated with adaptive lymphocyte responses, including; IL-2 (2.052 (1.391) vs. 0.525 (1.404) pg/mL, p = 0.03); IL-12p70 (5.989 (1.332) vs. 1.954 (1.342) pg/mL, p = 0.04); IL-4 (3.456 (1.432) vs. 0.669 (1.445) pg/mL, p = 0.01); and, IL-13 (1.747 (1.568) vs. 15.502 [1.551] pg/mL, p = 0.005), these were significantly elevated in AU cases with GI symptoms compared to those AU cases without GI symptoms. Interestingly, the regulatory cytokine IL-10 was decreased in AU cases with GI symptoms compared with those without GI symptoms (1.504 (1.516) vs. 9.365 (1.499) pg/mL, p = 0.01).
Mean GM-CSF concentrations were significantly higher in the AU with GI symptoms group compared with TD controls with no GI symptoms after adjusting for the child’s age at blood draw (16.248 (1.298) vs. 4.568 (1.298) pg/mL, p = 0.04; Table 1). Moreover, levels of other innate cytokines were increased for IL-1α (23.999 (1.496) vs. 9.757 (1.383) pg/mL; p = 0.011); IFNα-2 (86.574 (1.234) vs. 50.3 (1.184) pg/mL; p = 0.026); TNFβ (21.672 (1.1.349) vs. 8.248 (1.271) pg/mL; p = 0.006) and IL-15 (3.561 (1.405) vs. 0.905(1.314) pg/mL; p = 0.01) in AU with GI symptoms compared with TD controls without GI symptoms.
AU children with GI symptoms had significantly higher IL-4 levels than TD controls with GI symptoms (0.542 [1.707], p = 0.03). Levels of IL-13 were also significantly higher in AU children with GI symptoms compared to levels in TD controls with GI symptoms (1.113 (1.923), p = 0.03). IL-15 levels were also significantly higher in AU children with GI symptoms compared with TD children with GI symptoms (0.499 (1.660), p = 0.01).
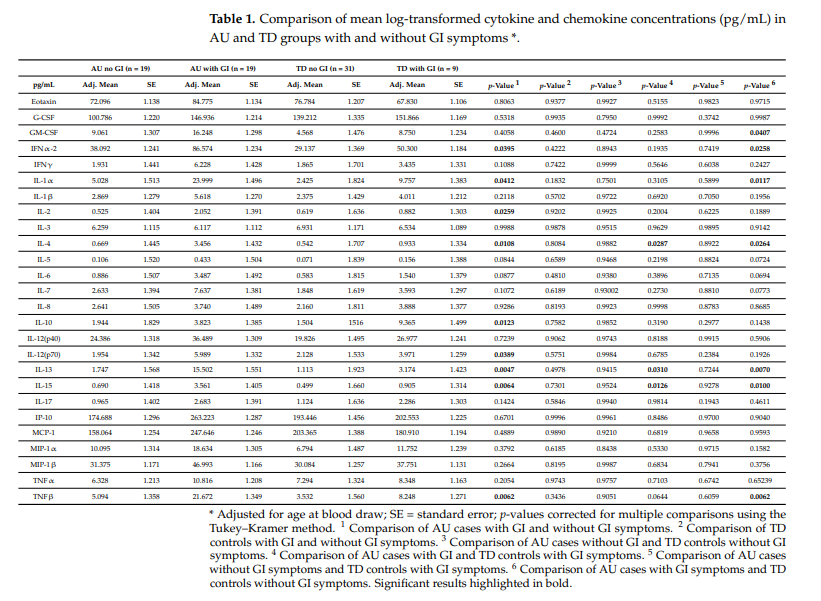
The concentrations of several other cytokines differed across cases and controls with and without GI symptoms but did not reach statistical significance at α = 0.05. Levels of IL-5 were higher in AU children with GI symptoms compared with AU children without GI symptoms (0.433 (1.504) vs. 0.106 (1.520) pg/mL, p = 0.08); moreover, IL-5 levels were higher in AU children with GI symptoms compared to TD children without GI symptoms (0.071 (1.839) pg/mL, p = 0.07). AU children with GI symptoms also had higher IL-6 concentrations than AU children without GI symptoms (3.487 (1.492) vs. 0.886 (1.507) pg/mL, p = 0.09), and AU children with GI symptoms had higher IL-6 concentrations than TD children without GI symptoms (0.583 (1.815) pg/mL, p = 0.07). Similarly, IL-7 levels were higher in AU children with GI symptoms compared with TD children without GI symptoms (7.637 (1.381) vs. 1.848 (1.619), p = 0.08).
4. Discussion
The immune system plays an essential role in protecting the host from infections and is continuously challenged by both external and internal stimuli. Inflammation is an important defense and survival response, triggered by innate and adaptive immune mechanisms.
However, persistent inflammation or dysregulated immune response could lead to impairment of physiological processes in both immune and non-immune systems. Moreover, inflammation leads to increased production of reactive oxygen species that can cause oxidative stress and tissue damage.
In AU, we and others have observed alterations of immune-related genes, inflammatory markers, oxidative stress, immune cell activation, and response to pathogens [3,5,10,34]. In this preliminary study, we found that children with AU and GI symptoms have elevated levels of innate cytokines IFNα, IL-1a, IL-15, and TNFβ, and adaptive cytokines IL-2, IL-4, IL-12 (p70), and IL-13, but decreased regulatory cytokine IL-10 compared with children with AU without GI symptoms.

In TD controls we found no differences based on GI symptoms. AU children with GI symptoms had significantly higher IL-4 and IL-13 levels than TD controls with GI symptoms. Moreover, several innate cytokines were increased in children with AU with GI symptoms compared with TD controls without GI symptoms including, GM-CSF, IL-1α, IFNα-2, and TNFβ.
Finally, the mucosal-related cytokine IL-15 was also increased in AU with GI symptoms compared to both TD controls and those without GI symptoms. These data suggest that there may be different patterns of plasma cytokines in children with AU dependent on the presence of co-morbidities such as GI symptoms.
The premise/utility of biological markers or signatures in AU is simple; to aid in diagnoses, to help monitor treatments/interventions, and to point to pathological pathways involved in causation(s). However, the implementation of biological markers into research or clinical settings in AU is far from simple and has so far been largely understudied.
For instance, in this study differences in plasma biomarkers were only apparent in the AU group with GI symptoms and not those without GI symptoms, after adjusting for age at child blood draw and statistical correction for multiple comparisons. This may reflect a true difference in immune activation in the AU group with GI or that the AU group without GI is more heterogeneous.
Based on the tools available in the CHARGE study (i.e., assessments using VABS, MSEL, ADOS, and ADI-R;), we were unable to reveal further co-morbidities in AU such as anxiety, attention deficit hyperactivity disorders, or enlarged brain growth that may also have an immune basis in the participants in the study. These co-morbidities could be present in either AU group, however, based on the current assessments there were no substantial differences in scores between the two AU groups.
Further studies with larger sample sizes are warranted to investigate whether other co-morbidities could also be identified using plasma cytokines. As well as differences within the AU groups, there were also differences between AU with GI and TD controls, with and without GI issues in plasma cytokine levels; this added further potential for plasma cytokines as biological signatures for the GI co-morbidities.
The increased production of IL-15 in AU with GI compared with AU without GI and both TD (with and without GI symptoms) has several implications for mucosal immune health. IL-15 is produced by epithelial cells in the gut and innate immune cells such as macrophages, and dendritic cells. IL-15 promotes T cell proliferation and cytokine production, influences the expression of the mucosal adhesion integrin—αEβ7—on intraepithelial T cells, and it can also induce the proliferation of intestinal epithelial cells [35].
In the GI tract, IL-15 is overexpressed in the gut mucosa of patients with celiac disease and is thought to contribute to epithelial damage [36]. The health of the GI tract is highly dependent on an intact gut barrier function in part regulated by tight junctions located in between the enterocytes. Using the lactulose: mannitol test, decreased intestinal permeability has been shown in autism [37,38].
Moreover, a previous study demonstrates that 75% of intestinal samples isolated from individuals with autism have reduced expression of barrier-forming tight junction components, and 66% had increased pore-forming claudins when compared to controls [39]. We have also shown altered levels in the genes controlling zonulin levels, a molecule that regulates intestinal permeability, in children with AU with GI symptoms but not AU without GI symptoms or controls [10].

IL-15 is also involved in the activation of NK cells a finding previously seen in autism [5]. Furthermore, IL-15 can block regulatory T-cell generation by inducing IL-12p70 production in dendritic cells [40]. Decreased regulatory T-cell formation has been observed in autism [22,41]. In the current study, we also saw increased IL-12p70 in AU with GI symptoms.
Differences in other innate cytokines were also noted in AU with GI symptoms. The innate immune system acts as a first line of defense and is triggered through pattern recognition receptors such as Toll-like receptors (TLR). In the GI tract, these interactions are important as there is potential to respond to commensal bacteria, foodborne pathogens, or bacterial by-products.
Major cells of the innate immune system include macrophages, dendritic cells, NK cells, and neutrophils. Perivascular macrophages and microglia (a specialized type of macrophage) are resident immune cells in the brain and protect against damage or infections. Alterations in innate response and microglia function are linked to several neurodevelopmental disorders, including autism [5].
Brain inflammation with activated microglia and astrocytes that affects neuronal connectivity with loss of synaptic connection and neuronal cell death has been described in ASD [42]. Studies also show elevated levels of pro-inflammatory cytokines such as IL-1β, IFN, TNF, and IL-8 both systemically and in the brain [5]. Meanwhile, anti-inflammatory cytokines, such as transforming growth factor beta1 (TGFβ1), and IL-35 are decreased in AU [10,25,27,42].
Interestingly, multiple studies reported strong associations between the severity of AU-related behaviors and cytokine levels, [3,13,43]. Of note, innate cytokine IL-1 can affect the hypothalamic-pituitary-adrenal (HPA) axis, and IL-6 has important roles in early neurodevelopment and neuro-immune communication [5].
As well as a change in the balance of innate cytokines and IL-12, we also saw increases in cytokines associated with atopy in AU with GI symptoms. IL-4 and IL-13 are associated with humoral responses and are drivers of inflammation in atopy and food allergies [40,46]. Autism has previously been associated with increased food allergies and asthma [44–48]. Increased IL-4 has also been found in neonatal blood spots from children who later developed severe AU [49]. Further studies to investigate biomarkers and food allergies in AU are warranted.
Furthermore, another difference between the AU groups was the reduced plasma levels of IL-10 which may suggest an imbalance in immune regulation. We, and others, have observed decreased plasma levels of other regulatory cytokines such as active TGFβ1 and IL-35 levels in adults and children with AU [25,27,50]. Several reports have also shown decreased IL-10 levels in T cells in children with autism [17,18,22,51]. Maintaining immune homeostasis is a balance between providing regulation/tolerance to self-proteins and important beneficial commensal microbes, and responding to pathogenic microbes. Previous studies investigating stimulated responses in immune cells in AU children found that those who have marked behavioral fluctuations and GI symptoms, have decreased IL-10 to certain immune stimuli [23,24].
While increased inflammatory mediators help mount an effective defense against pathogens, disruption in regulation can lead to excessive inflammation and has been implicated in many autoimmune disorders including those that affect the GI such as Crohn’s disease and ulcerative colitis [52–55].
Recent studies indicate that host immune responses can influence the gut microbiome, as well as the nervous system, and may be involved in the pathology of AU [4]. For instance, the innate immune response can shape the microbiome composition through the production of antimicrobial molecules including α-defensins and β-defensins, whereas adaptive responses can lead to IgA, and T cells that are specific for commensal bacteria [56–58]. Furthermore, the production of reactive oxygen species due to inflammatory responses can lead to overgrowth of specific bacterial species, altering the microbiome composition.
Altered microbiota composition is commonly found in AU; however, the causes of this are unknown and may relate to food sensitivities or dietary preferences [59]. Moreover, it was recently shown that GI symptoms in AU are not associated with microbiome changes in AU [60], whereas we show here and previously, that immune activation and plasma cytokines are associated with GI symptoms in AU [10,22,25]. Further studies are needed to clarify the role of the immune–gut–brain axis, including the interplay between the immune system, intestinal barrier function, microbiome, and the vagal nerve and peripheral nervous system.
As a preliminary study, there are several limitations. Our study is limited by small sample sizes that impacted any behavioral analyses within groups and restricted how we could stratify our study population.
We did not find evidence for predictive markers of state and trait features with any cytokine using area under the curve (AUC) analyses (data not shown) a fact that most likely reflects the large number of variables that need correcting for and the small group sizes. As with other studies, the recruitment of TD children who experience GI issues was difficult due to their low frequency in the general population in this age group. We focused on symptoms of irregular bowel movements as these previously have been the most associated with GI symptoms in AU [1,3].
Due to limited numbers, we were unable to further break down groups to examine differences between specific GI symptom clusters (e.g., constipation vs. diarrhea vs. IBS) but this warrants further study. As the recruitment of males and females was consistent with AU diagnosis, we did not have sufficient statistical power to analyze sex differences due to the low number of females per group.
Finally, our study included a narrow (young) age group, in follow-up studies it would be warranted to compare plasma cytokines in older age groups to see how GI symptoms change and how they are associated with plasma cytokines across age. In addition, in this study we cannot determine the direction of elevated cytokine levels, i.e., is it generated in the gut epithelium, lamina propria, mesenteric lymph nodes, or liver? However, even with these limitations, we feel that this study provides invaluable clues for plasma cytokines and GI symptoms in children with AU.
Due to the heterogeneity of AU and the varying types of immune dysfunction reported, we sought to investigate differences in plasma cytokines within a subgroup of children based on the comorbidity of GI symptoms. The main aim of this study was to identify differences in inflammatory and regulatory plasma cytokines in AU with and without GI symptoms compared with TD children. Children with AU and GI symptoms displayed the largest number of differences with elevated inflammatory cytokines and decreased regulatory IL-10 compared with AU with no GI symptoms. Mucosal-relevant IL-15 was increased in AU with GI symptoms compared with all groups. We previously reported altered immune responses in children with AU who experience GI symptoms. Peripheral blood mononuclear cells from children with AU and GI symptoms produced increased mucosa-related cytokines but decreased active TGFβ1 after stimulation in vitro [10], suggesting a net imbalance away from a regulated response.
These data, along with the current study, illustrate the need to find common subgroups within AU, that may help define more targeted treatments to benefit individuals across the spectrum [1]. The field of AU requires further investigation to elucidate the complex pathogenesis of this wide spectrum of conditions and co-morbidities. This leads to the question of how we can ameliorate intestinal disorders in the context of AU. For example, can immune modulation help restore intestinal homeostasis, and which immune-related factors should be targeted? Our data suggest two possible areas, either decreasing inflammatory cytokines or increasing immune regulation. Future studies could focus on immune cell activation in the gut in AU to help unravel signaling pathways, and immune activation in children with AU with GI co-morbidities.

Funding:
This study was funded by Autism Speaks Foundation (Grant #7567), The National Institutes of Health, grants R21HD086669, P01 ES011269-11, R01MH118209, R01HD090214, R01ES015359, NARSAD Foundation, Michael and Barbara Bass Foundation, Jane Botsford Johnson Foundation, Jonty Foundation, and Brain Foundation. This work was supported by the Department of Defense, through the Autism Research Program under Award No. W81XWH-18-1-0681. Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the Department of Defense or NIH.
Institutional Review Board Statement:
The CHARGE Study protocol was approved by institutional
review boards of the University of California in Davis and the State of California Committee for the
Protection of Human Subjects (IRB ID: 226028-33).
Informed Consent Statement:
Written informed consent was obtained before participation.
Data Availability Statement:
Data is available on request.
Acknowledgments:
We would like to thank the participants and their families for their participation in the study and the staff of both the University of California Davis M.I.N.D. Institute and the CHARGE project for their technical support. We would like to thank Paula Krakowiak for her advice and statistical support.
Conflicts of Interest:
The authors declare no conflict of interest.
References
Coury, D.L.; Ashwood, P.; Fasano, A.; Fuchs, G.; Geraghty, M.; Kaul, A.; Mawe, G.; Patterson, P.; Jones, N.E. Gastrointestinal conditions in children with autism spectrum disorder: Developing a research agenda. Pediatrics 2012, 130 (Suppl. S2), S160–S168. [CrossRef] [PubMed]
2. Gesundheit, B.; Rosenzweig, J.P.; Naor, D.; Lerer, B.; Zachor, D.A.; Procházka, V.; Melamed, M.; Kristt, D.A.; Steinberg, A.; Shulman, C.; et al. Immunological and autoimmune considerations of Autism Spectrum Disorders. J. Autoimmun. 2013, 44, 1–7. [CrossRef] [PubMed]
3. Hughes, H.K.; Mills Ko, E.; Rose, D.; Ashwood, P. Immune Dysfunction and Autoimmunity as Pathological Mechanisms in Autism Spectrum Disorders. Front. Cell Neurosci. 2018, 12, 405. [CrossRef] [PubMed]
4. Hughes, H.K.; Rose, D.; Ashwood, P. The Gut Microbiota and Dysbiosis in Autism Spectrum Disorders. Curr. Neurol. Neurosci. Rep. 2018, 18, 81. [CrossRef]
5. Hughes, H.K.; Moreno, R.J.; Ashwood, P. Innate immune dysfunction and neuroinflammation in autism spectrum disorder (ASD). Brain Behav. Immun. 2023, 108, 245–254. [CrossRef]
6. Restrepo, B.; Angkustsiri, K.; Taylor, S.L.; Rogers, S.J.; Cabral, J.; Heath, B.; Hechtman, A.; Solomon, M.; Ashwood, P.; Amaral, D.G.; et al. Developmental-behavioral profiles in children with autism spectrum disorder and co-occurring gastrointestinal symptoms. Autism. Res. 2020, 13, 1778–1789. [CrossRef]
7. Sanctuary, M.R.; Kain, J.N.; Chen, S.Y.; Kalanetra, K.; Lemay, D.G.; Rose, D.R.; Yang, H.T.; Tancredi, D.J.; German, J.B.; Slupsky, C.M.; et al. Pilot study of probiotic/colostrum supplementation on gut function in children with autism and gastrointestinal symptoms. PLoS ONE 2019, 14, e0210064. [CrossRef]
8. Kanner, L. Autistic disturbances of affective contact. Nerv. Child 1943, 2, 217–250.
9. Ashwood, P.; Murch, S.H.; Anthony, A.; Pellicer, A.A.; Torrente, F.; Thomson, M.A.; Walker-Smith, J.A.; Wakefield, A.J. Intestinal lymphocyte populations in children with regressive autism: Evidence for extensive mucosal immunopathology. J. Clin. Immunol. 2003, 23, 504–517. [CrossRef]
10. Rose, D.R.; Yang, H.; Serena, G.; Sturgeon, C.; Ma, B.; Careaga, M.; Hughes, H.K.; Angkustsiri, K.; Rose, M.; Hertz-Picciotto, I.; et al. Differential immune responses and microbiota profiles in children with autism spectrum disorders and co-morbid gastrointestinal symptoms. Brain Behav. Immun. 2018, 70, 354–368. [CrossRef]
11. Torrente, F.; Anthony, A.; Heuschkel, R.B.; Thomson, M.A.; Ashwood, P.; Murch, S.H. Focal-enhanced gastritis in regressive autism with features distinct from Crohn’s and Helicobacter pylori gastritis. Am. J. Gastroenterol. 2004, 99, 598–605. [CrossRef]
12. Chaidez, V.; Hansen, R.L.; Hertz-Picciotto, I. Gastrointestinal problems in children with autism, developmental delays or typical development. J. Autism Dev. Disord. 2014, 44, 1117–1127. [CrossRef]
13. Careaga, M.; Rogers, S.; Hansen, R.L.; Amaral, D.G.; Van de Water, J.; Ashwood, P. Immune Endophenotypes in Children With Autism Spectrum Disorder. Biol. Psychiatry 2017, 81, 434–441. [CrossRef]
14. Corbett, B.A.; Kantor, A.B.; Schulman, H.; Walker, W.L.; Lit, L.; Ashwood, P.; Rocke, D.M.; Sharp, F.R. A proteomic study of serum from children with autism showing differential expression of apolipoproteins and complement proteins. Mol. Psychiatry 2007, 12, 292–306. [CrossRef]
15. Goines, P.E.; Ashwood, P. Cytokine dysregulation in autism spectrum disorders (ASD): Possible role of the environment. Neurotoxicol. Teratol. 2013, 36, 67–81. [CrossRef]
For more information:1950477648nn@gmail.com






