Preparation And Application Of Cistanche Glycoside By Enzymatic Hydrolysis Method
May 26, 2023
Abstract: Exploring the preparation of Cistanche glycoside by enzymatic hydrolysis and its application research. Using the extraction rate of Cistanche glycoside as the evaluation indicator, the enzymatic hydrolysis method was used to extract Cistanche glycoside, and it was determined using high-performance liquid chromatography (HPLC). By constructing an aging model mouse, the glycosides of Cistanche deserticola prepared by enzymatic hydrolysis were applied to the aging model mice, and the learning and memory abilities of the aging model mice were observed, as well as the detection of brain tissue oxidation levels. The results showed that the optimal conditions for the enzymatic hydrolysis orthogonal experiment were: when the ratio of material to liquid was 1:10, the extraction times were 3 times, the extraction temperature was 80 ℃, and the extraction time was 1 minute, the extraction rate of Cistanche deserticola glycosides was the highest. Compared with the control group, the effect of Cistanche glycoside on aging model mice significantly improved their learning and memory abilities (P<0.05); After treatment with cistanche glycoside, the content of superoxide dismutase (SOD) and catalase (CAT) in brain tissue of aging model mice increased significantly (P<0.05), while the content of malondialdehyde (MDA) decreased significantly (P<0.05). In this study, the preparation method of Cistanche glycosides was optimized and the anti-aging ability of Cistanche glycosides was tested. This study laid a theoretical and practical foundation for the further development and clinical application of Cistanche glycosides in the future.
Phenylethanol glycoside is the main active component of Cistanche deserticola
Keywords: enzymatic hydrolysis method; Cistanche glycoside; Aging model mice; Antioxidant; application research
1 Experimental part
1.1 Main materials, reagents, and instruments
Healthy male ICR mice, Beijing Huizhi Heyuan Biotechnology Co., Ltd; Cistanche glycoside, Anyang Yishoutang Pharmaceutical Chain Co., Ltd; Cistanche glycoside standard (purity ≥ 99.0%), Wuhan Huashun Biotechnology Co., Ltd; Inner Mongolia Beiqi, Shanxi Beiqi, Inner Mongolia Cistanche glycoside special grade, Inner Mongolia Cistanche glycoside first class, Inner Mongolia Cistanche glycoside second class, Shanxi Heipi Qi AA grade, Shanxi Heipi Qi A grade, Gansu Cistanche glycoside, all purchased from local pharmacies; Methanol, ethanol, analytical alcohol, Nanjing Datang Chemical Co., Ltd; SOD, MDA, and CAT kits, Nanjing Jiancheng Biotechnology Co., Ltd.
DFY400 Swinging High-Speed Traditional Chinese Medicine Grinder, Zhejiang Wenling Dade Traditional Chinese Medicine Machinery Co., Ltd; SHZ-D circulating water vacuum pump, Gongyi Yuhua Instrument Co., Ltd; 101-1Y infrared drying oven, Hangzhou Lantian Laboratory Instrument Factory; U2001 Ultraviolet-Visible Spectrophotometer, Hitachi, Japan; Aglient 1100 LC High-Performance Liquid Chromatography, Agilent Technologies, USA; 6511 electric mixers, Shanghai Specimen Model Factory; PHS-3C Acidity Meter, Shanghai Ridao Scientific Instrument Co., Ltd; DF-500 freeze-drying machine, Baokun Vacuum Technology Co., Ltd.

Cistanche extract powder
Click here to view Cistanche products
【Ask for more】 Email:cindy.xue@wecistanche.com / Whats App: 0086 18599088692 / Wechat: 18599088692
1.2 Experimental methods
1. 2. 1. Treatment and Extraction of Cistanche Glycoside Samples
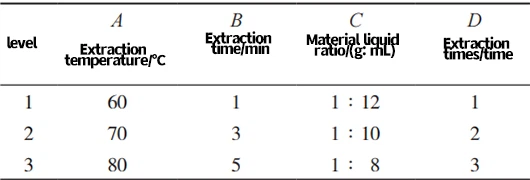
Using the extraction rate of Cistanche glycosides as the evaluation index, the L9 (34) orthogonal experimental design was used for investigation. The specific scheme design is shown in Table 1.
Tab. 1 Factor level table
Accurately weigh 100.0 g of Cistanche glycoside sample, dry it in a drying oven, crush it, pass through a 60 mesh sieve, and extract Cistanche glycoside from Scutellaria baicalensis self-enzyme hydrolysate according to the orthogonal experimental design scheme in Table 1 [13,14].
1. 2. 2. Determination of Cistanche glycoside content
Use a UV-visible spectrophotometer to determine the content of Cistanche glycoside. Accurately measure 0.5 mL of Cistanche glycoside extract and mix it with 4.5 mL of ultrapure water. Add 0.5 mL of 5% sodium nitrite solution and let it stand for 5 minutes. Then, add 0.5 mL of 10% aluminum nitrate solution. Gently mix and let it stand for 6 minutes. Then, add 4 mL of 1.0 mol/L sodium hydroxide solution. Gently mix and let it stand for 15 minutes. Measure at a wavelength of 510 nm between ultraviolet and visible light.

Cistanche deserticola experiment
1. 2. 3 HPLC analysis
Using Kromasil C18 column (250 mm × 4.6 mm,5 μ m) Room temperature, injection volume 20 μ L. Flow rate 1 mL/min, A-phase water, B-phase acetonitrile. Elution procedure: 0-5 minutes, 5% phase B, 95% phase A; 5-8 minutes, 5% -8% phase B, 95% -92% phase A; 8-15 minutes, 20% phase B, 80% phase A; 15~18 minutes, 20%~30% phase B, 80%~70% phase A; 18-35 minutes, 30% -45% phase B, 70% -55% phase A; 35~38 minutes, 45%~80% phase B, 55%~20% phase A. The detection wavelength is 254 nm.
1. 2. 4 Animal grouping and administration
72 healthy male ICR mice were selected and randomly divided into 4 groups, with 18 mice in each group: control group (Con group), model group (Mod group), positive control group (PC group), and cistanche glycoside group (HQ group). The control group mice received oral administration of double distilled water and subcutaneous injection of isotonic saline; The mice in the model group were orally administered with double distilled water and hypodermically injected with 400 mg/kg D-neneneba galactose; In the positive control group, 800 mg/kg piracetam was orally administered and 400 mg/kg D-neneneba galactose was subcutaneously injected; Mice in the cistanche glycoside group were given 400 mg/kg astragaloside (the dose was determined in the preliminary experiment; Shanxi Beiqi) orally, and 400 mg/kg D-neneneba galactose was injected subcutaneously. All experimental mice were administered orally and subcutaneously for 10 consecutive weeks. Weigh each mouse every week and closely monitor the physical condition of each group of mice. In the 9th week, after oral administration and subcutaneous injection for 30 minutes, each group of mice was subjected to platform jumping and darkness avoidance tests. In the 10th week, the Morris water maze test was performed on each group of mice. In the 11th week, the brain tissue of each group of mice was taken to determine the content of superoxide dismutase (SOD), catalase (CAT), and malondialdehyde (MDA) in brain tissue.
1. 2. 5 Measurement of Learning and Memory Ability in Aging Mice
1) Avoidance experiment
Before the experiment, the mice were placed on the instrument and acclimated for 3 minutes. After fully adapting to the dark instrument and environment, the mice were placed back in the breeding cage. Next, the mice were subjected to a continuous electric shock while standing on the floor. After the shock occurred, the mice instinctively fled from the floor to the platform, and the entire training process lasted for 5 minutes. After the experiment was completed, the total number of times the mice received electric shocks was counted, which is the number of dark avoidance errors.

Desert living cistanche
2) Jumping platform experiment
The jumping platform experimental device is 45 cm × 35 cm × A 25 cm organic glass box with a partition inside, and a rubber pad with a diameter of 9.5 cm and a height of 4.5 cm is placed on the left side of the left and right grids. The bottom of the box is covered with copper grids as stimulation electrodes. When subjected to electric shock stimulation, the mouse will escape to a safe platform and then jump off the platform. When the mouse's feet come into contact with the electric grid and receive an electric shock, it is an error response. Record the number of errors and latency of the mouse within 3 minutes. After 24 hours, the test again measured the learning and memory abilities of the mice based on the number of errors and latency.
3) Water maze experiment
Before the experiment, the mice were placed in a water maze device and allowed to swim freely for 2 minutes to adapt to the environment.
Positioning navigation experiment [15]: Record the time when mice find the platform.
Space exploration experiment [16]: record the times of mice crossing the platform.
1. 2. 6 Determination of antioxidant content in brain tissue of 6 aging mice
After separating mouse brain tissue according to conventional methods, the content of SOD, MDA, and CAT in the brain tissue of each group of mice was measured according to the operation steps of SOD, MDA, and CAT kits.
1.3 Data Statistics and Analysis
In this study, all data were statistically analyzed using Excel spreadsheets and SPSS19.0 software, and inter-group differences were evaluated using paired t-tests. When P<0.05, it indicates statistical significance; When P<0.01, it indicates significant statistical significance.
2 Results and Discussion
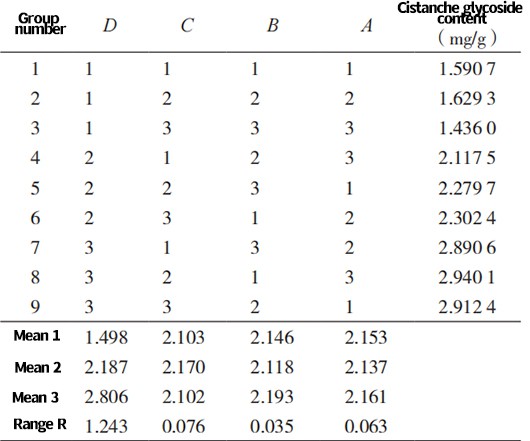
2.1 Orthogonal experiment
Table 2 shows the results of the orthogonal experiment for Cistanche glycosides. It can be seen from Table 2 that the optimal extraction process is A3B1C2D3, with an extraction temperature of 80 ℃, an extraction time of 1 minute, a solid-liquid ratio of 1:10, and three extraction times. The highest extraction rate of Cistanche glycosides is achieved.
Tab. 2 Orthogonal experiment ofCistanche glycoside
2.2 Determination of Cistanche glycoside content
The UV visible spectrophotometry was used to detect the extract of 8 cistanche glycoside samples. The results are shown in Figure 1, and there are significant differences in the content of cistanche glycoside among different regions or varieties. Among them, Shanxi Beiqi has the highest content of cistanche glycoside, while Shanxi Heipi Qi has the lowest content of AA grade cistanche glycoside.
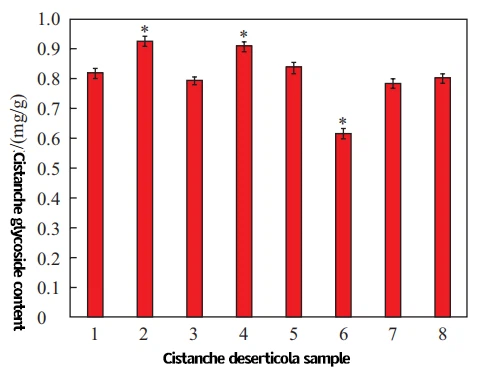
Fig. 1 Determination of different kinds of Cistanche glycoside
2.3 HPLC detection of Cistanche glycosides
According to the chromatographic conditions determined in Section 1.2.3, the HPLC diagram of Cistanche glycosides is shown in Figure 2. The retention time of Cistanche glycosides is 10.64 minutes, and the mass concentration is 1.0-40.0 μ G/mL, with good linearity and an R-value of 0.999 8.

Fig. 2 HPLC diagram of Cistanche glycoside
2.4 Precision and stability
Take the same batch of Cistanche glycoside test solution and inject 6 consecutive times to determine the peak area of Cistanche glycoside. The results showed that the precision RSD of Cistanche glycosides was less than 2.5% (n=6), indicating a good precision of Cistanche glycosides.
Take the same batch of Cistanche glycoside test solution and measure the peak area of Cistanche glycoside at 0, 3, 6, 9, 12, 15, 18, 21, and 24 hours respectively. The results showed that the RSD of Cistanche glycoside stability was less than 2.0% (n=6), indicating that Cistanche glycoside had good stability within 24 hours.
2. 5 Effect of Cistanche glycoside on body weight of aging model mice
Reference [17-19] found that the dosage of Cistanche glycosides used in this study did not produce systemic toxicity in mice. As shown in Figure 3, the weight of all groups of mice increased before the 6th week. Compared with the control group, the weight of the model group mice decreased significantly during the same period (P<0.01), while the weight of the positive control group and astragalus group mice slightly decreased. After the 6th week, the weight of the control group mice continued to increase, while the weight of the model group mice no longer increased. The positive control group and the Cistanche glycoside group slightly increased.
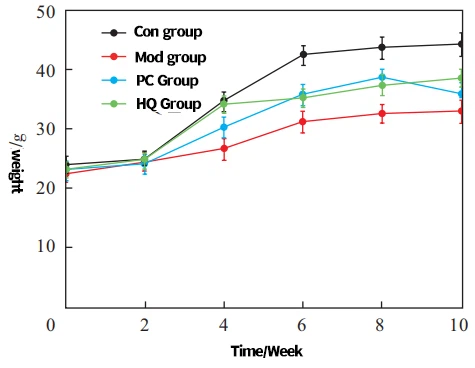
Fig. 3 The effect of Cistanche glycoside on the body weight of the aging model mice
2.6 Behavioral Experiment Results
2. 6. Observation of the Effect of Cistanche Cistanche Glycoside on Aging Model Mice through Darkness Avoidance Experiment
As shown in Figure 4A, the latency period of mice in each group was prolonged on the second day compared to the first day. Compared with the control group, the latency time of the model group mice was significantly reduced (P<0.05); Compared with the model group, the latency time of mice in the positive control group and the Cistanche glycoside group was significantly increased (P<0.05). As shown in Figure 4B, the number of errors in each group of mice decreased on the second day compared to the first day. Compared with the control group, the number of errors in the model group mice significantly increased (P<0.05); Compared with the model group, the positive control group and the Cistanche glycoside group significantly reduced the number of errors in mice (P<0.05).
2. 6. Observation of the Effect of Cistanche Cistanche Glycoside on Aging Model Mice through a Two-Step Platform Experiment
As shown in Figure 5, the latency time of the jumping platform in each group of mice was extended on the second day compared to the first day. Compared with the control group, the jumping stage time of the model group mice was significantly reduced (P<0.05); Compared with the model group, the latency time of the jumping platform in the positive control group and the Cistanche glycoside group of mice was significantly increased (P<0.05). As shown in Figure 4B, the number of errors in each group of mice decreased on the second day compared to the first day. Compared with the control group, the number of errors in the model group mice significantly increased (P<0.05); Compared with the model group, the positive control group and the Cistanche glycoside group significantly reduced the number of errors in mice (P<0.05).
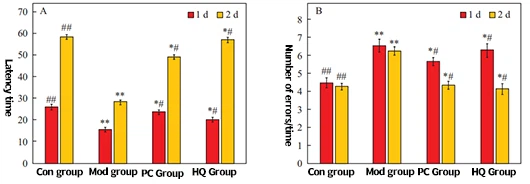
Compared with the control group, P<0.05; Compared with the control group, P<0.01; Compared with the model group, the '#' is P<0.05; Compared with the model group, P<0.01, the same below
Fig. 4 Dark avoidance experiments to observe the effect of Cistanche glycoside on the aging model mice. Latency time (A); Number of errors (B)

Fig. 5 The effect of Cistanche glycoside on the aging model mice observed by platform jumping experiment. Latency time (A); Number of errors
2. 6. 3 Morris water maze experiment to observe the effect of Cistanche glycosides on aging model mice
As shown in Figure 6, as the number of training days increases, the time for each group of mice to find the platform gradually decreases. Compared with the control group, the time for the model group mice to find the platform significantly increased (P<0.05), while the time for the positive control group and the Cistanche glycoside group mice to find the platform slightly increased.
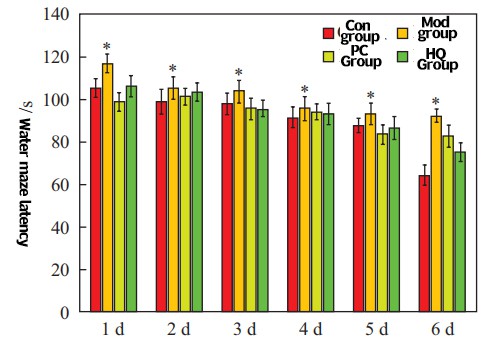
Compared with the control group, P<0.05
Fig. 6 The effect of Cistanche glycoside on the aging model mice observed by positioning navigation experiment
As shown in Figure 7, as the number of training days increases, the number of times each group of mice crosses the platform gradually increases. Compared with the control group, the number of times the model group mice crossed the platform significantly decreased (P<0.05), while the number of times the positive control group and the Cistanche glycoside group mice crossed the platform slightly decreased.

Fig. 7 The effect of Cistanche glycoside on the aging model mice observed by space exploration experiment
2. 7 Effect of Cistanche Glycosides on antioxidant levels in brain tissue of aging model mice
As shown in Figure 8, compared with the control group, the SOD and CAT enzyme activities in the brain tissue of the model group mice were significantly reduced (P<0.05). The SOD and CAT enzyme activities in the brain tissue of the positive control group and the Cistanche glycoside group mice were slightly increased, while the MDA content in the brain tissue of the model group mice was significantly increased (P<0.05). The MDA content in the brain tissue of the positive control group and the Cistanche glycoside group mice was slightly reduced.

Chinese herb cistanche
3 Conclusion
This study used the extraction rate of Cistanche glycoside as the evaluation indicator and extracted Cistanche glycoside using the enzymatic hydrolysis method. The content of Cistanche glycoside was determined by HPLC. The following conclusions were drawn:
1) When the ratio of material to liquid is 1:10, the extraction frequency is 3 times, the extraction temperature is 80 ℃, and the extraction reagent is 1 minute, the extraction rate of Cistanche glycosides is the highest. The content of cistanche glycoside in 8 different varieties was determined using a UV visible spectrophotometer. The results showed significant differences in cistanche glycoside content among different regions or varieties. Among them, Shanxi Beiqi had the highest content of cistanche glycoside, while Shanxi Heipi Qi had the lowest content of cistanche glycoside in the AA grade. In order to further establish a separation and purification system for Cistanche glycoside, a fingerprint of Cistanche glycoside was established by HPLC. The results showed that the retention time of Cistanche glycoside was 10.64 minutes, the mass concentration was 1.0~40.0 μ G/mL, with good linearity and an R-value of 0.999 8, and the HPLC method has good stability and precision.
2) Effect of Cistanche deserticola glycoside prepared by enzymatic hydrolysis on aging model mice, observe the learning and memory abilities of age model mice and detect brain tissue oxidation levels. The results showed that compared with the control group, Cistanche glycosides significantly improved the learning and memory abilities of age model mice after treatment (P<0.05); Compared with the control group, the activity of SOD and CAT enzymes in the brain tissue of aging model mice significantly increased (P<0.05) after treatment with Cistanche glycosides, while the content of MDA significantly decreased (P<0.05).
reference:
[1 ] Wang Xiaoju. Extraction, separation and activity study of the active components ofCistanche glycoside [D]. Xi’an: Shanxi University, 2005.
[ 2 ] Wang Guangzhong, Hu Di, Chen Jingbing. Orthogonal design to optimize the extraction process of total flavonoids and total saponins ofCistanche glycoside [J]. Chinese Patent Medicine, 2004, 26(10): 3-4.
[ 3 ] Liu Jinhua, Chen Jingran, You Guangfu. Advances in pharmacological research on myocardial protection of Astragalus [J]. Chinese Patent Medicine, 2002, 24(8): 623-626.
[ 4 ] Guo Wei. Pharmacology and clinical research overview of traditional Chinese medicineCistanche glycoside [J]. Shanxi Traditional Chinese Medicine, 2011, 27(11): 52-54.
[ 5 ] Lu Yanqi, He Xueli. A review of the chemical constituents and pharmacological effects ofCistanche glycoside [J]. Journal of Baoding Teachers College, 2004, 17(4): 40-42.
[ 6 ] Li Qin, Liu Hong. Pharmacological effects and clinical application ofCistanche glycoside [J]. Chinese Medicine of Traditional Chinese Medicine, 2004, 10(10): 633-635.
[7 ] Jin Jizi. Research progress on the pharmacological effects and clinical application ofCistanche glycoside [J]. Chinese Medicine of Traditional Chinese Medicine, 2008, 17(20): 134-135. [ 8 ] Chen Jianzhen, Lv Guiyuan, Ye Lei. Research progress on chemical constituents and pharmacological effects ofCistanche glycoside flavonoids [J]. Medical Herald, 2009, 28(10): 1314-1316.
[ 9 ] Liang Liansheng, Yu Jing. Pharmacological effects of flavonoids inCistanche glycoside [J]. Journal of Integrative Chinese and Western Medicine Cardiovascular and Cerebrovascular Diseases, 2005, 3(12): 1085-1087.
[10] Zhang Dongqing, Wang Deqing. Research progress on biological activity of total flavonoids ofCistanche glycoside [J]. China Important Journal, 2010, 35(2): 253-256.
[11] Li Y, Guo S, Zhu Y, Yan H, et al. Flowers ofCistanche glycoside membranaceus var. mongholicus as a novel high potential by product: phytochemical characterization and antioxidant activity [J]. Molecules, 2019, 24(3): 434.
[12] Butkutė B, Dagilytė A, Benetis R, et al. Mineral and phytochemical profiles and antioxidant activity of herbal material from two temperateCistanche glycoside species [J]. Biomed. Res. Int., 2018.
[13] Sharifi-Rad M, Pohl P, Epifano F, et al. Green synthesis of silver nanoparticles usingCistanche glycoside tribuloides delile. Root extract: characterization, antioxidant, antibacterial, and anti-Inflammatory activities [J]. Nanomaterials (Basel), 2020, 10(12): 2383.
[14] Wang D, Zhuang Y, Tian Y, et al. Study of the effects of total flavonoids ofCistanche glycoside on atherosclerosis formation and potential mechanisms [J]. Oxid. Med. Cell. Longev., 2012.






