The Beneficial Effects Of Cistanche Tubulosa Extract On Improving The Low Intestinal Permeability Of Echinacoside (ECH) And Acteoside (ACT).
Mar 25, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
Tadatoshi Taninoa , Noriaki Nagaib and Yoshinori Funakamib
* Faculty of Pharmaceutical Sciences, Tokushima Bunri University, Tokushima and b Faculty of Pharmacy, Kinki University, Osaka, Japan
Abstract
Objectives The objective of this study was to address the beneficial effects of Cistanche tubulosa extract on improving the low intestinal permeability of echinacoside (ECH) and acteoside (ACT). Methods Absorption of ECH and ACT in C. tubulosa extract was characterized using human intestinal Caco-2 cell monolayers with intact compounds. Glucose transporter-dependent absorption of ECH and ACT was confirmed by an in-situ intestinal perfusion technique. Key findings The apparent permeability (Papp) was not significantly different between intact ECH and intact ACT. In the presence of phloridzin, the Papp of the ECH and ACT at a high dose was reduced to 20% of the respective non-treatment but was not altered by phloretin and verapamil. C. tubulosa extract at low and high doses enhanced the Papp of ECH and ACT (both by threefold), resulting in their large participation in sodium-dependent glucose transporter-independent absorption. At a low concentration, concomitant ECH and ACT levels in portal blood were significantly suppressed by phloridzin. Conclusion The dietary and medicinal C. tubulosa extract enhancing the intestinal absorption of ECH and ACT may serve to better manage human health, although the involvement of phloridzin-sensitive transport should be reduced.
Keywords acteoside; Caco-2 cell monolayers; Cistanche tubulosa extract; echinacoside; phloridzin-sensitive glucose transporter
Introduction
The roots of Cistanche tubulosa have traditionally been used for medicine and food. C. tubulosa extract is known to possess pharmacological effects in various brain diseases, anti-aging functions, fat metabolism, and hair growth.[1–4] Recently, iridoids, monoterpenoids, phenylethanoid glycosides, and lignans have been isolated from C. tubulosa. [5,6] Phenylethanoid glycosides, a class of polyphenolic compounds, are the main chemical ingredients in Cistanche species,[7] although their amounts vary among different species. Echinacoside (ECH; Figure 1) is one of the major phenylethanoid glycosides in Herba Cistanchis. It is hydrolyzed to acteoside (ACT; also called verbascoside) by enzymes of bacterial origin in the large intestine.[8,9] ECH and ACT possess the beneficial activity of hepatoprotection[10] and anti-inflflammation[11] in rodent animals. Surprisingly, highly water-soluble ECH improves behavioral and neurochemical outcomes in a mouse model of Parkinson’s disease and inhibited caspase-3 and caspase-8 activation in cerebellar granule neurons.[9] It is well known that the blood-brain barrier strictly limits the entry and distribution of xenobiotics into the brain from the blood. Wu et al. [12] also showed that water-soluble ACT was rapidly distributed in brain tissues of rats. Therefore, ECH and ACT may be transported into the brain, intestines, and liver by specific system(s).

Figure 1 Chemical structures of echinacoside and acteoside.
Although there is strong evidence to suggest that consumption of C. tubulosa extract is beneficial to human health, the permeability of pure ECH across Caco-2 cell monolayers at an apical concentration of 8.4 ± 1.6 μg/ml is equal to or below that of the paracellular transport marker mannitol.[13] When pure ECH is orally administered to rats (dose, 100 mg/kg), absorption is extremely fast (Tmax, 15 min), and the maximum serum concentration is very low (Cmax, 0.61 ± 0.32 μg/ml).[14] The absolute bioavailability of ECH is only 0.83%. Similarly, when Caco-2 cells are incubated with a phenolic fraction partially purified from olive mill wastewater, pure ACT uptake is rapid with peak accumulation occurring after 30 min and a total accumulation efficiency of 0.1%, providing intracellular levels of 130 pmol/mg cell protein.[15] In rats, the maximal concentration (0.13 ± 0.03 μg/ml) of pure ACT was reached within 30 min after oral dosing with 100 mg/kg,[12] implying rapid intestinal absorption. The oral bioavailability of ACT, as well as ECH, is quite low (0.12 ± 0.04%), suggesting the possibility of first-pass effects in the intestinal tract and liver. In rat bile, methylation and glucuronidation conjugates of ECH are major metabolites,[16] although the extent of hepatic metabolism remains unclear. We preliminarily found that ECH and ACT were quite stable in the homogenates of rat intestinal mucosa and artificial gastric acid (data not shown). Najar et al. [17] demonstrated that ACT inhibits P-glycoprotein (P-GP)-ATPase activity in a manner similar to verapamil (a representative P-gp inhibitor), implying a P-gp modulator; however, it is uncertain whether ACT is available as a P-gp substrate. Interestingly, the recent findings of dietary flavonoid-D-glucosides showed that multidrug resistance protein (MRP2) masked the sodium-dependent glucose transporter (SGLT)1- mediated uptake of quercetin 4′-O-β-glucose,[18,19] which is responsible for very poor absorption. However, very little is known about the sensitivity of polyphenolic glucosides to absorptive transporters, including glucose transporters. Information on the absorption characteristics of quercetin 4′-glucoside and rapidly blood-brain barrier-permeable ECH prompted us to investigate the transporter-sensitive uptake of phenylethanoid glycosides in dietary C. tubulosa extract.
In this study, we investigated glucose transporter-mediated absorption of intact ECH and ACT using human intestinal Caco-2 cell monolayers. Simultaneously, the absorption transport of ECH and ACT concomitant in dietary C. tubulosa extract was characterized by an in-vitro model and in-situ intestinal perfusion system with portal blood sampling, which can easily distinguish between the extent of absorption and avoidance of hepatic first-pass disposition.
Materials and Methods
Materials
Intact ECH and ACT were generous gifts from Eishin Trading Co., Ltd (Osaka, Japan). Phloridzin and phloretin were purchased from Tokyo Kasei Co., Ltd. (Tokyo, Japan). Verapamil and p-coumaric acid, used as internal standards for high-performance liquid chromatography (HPLC) assay, were obtained from Sigma-Aldrich (St Louis, MO, USA). All other chemicals used were of analytical grade and commercially available.
Plant material and preparation of the methanolic extract
C. tubulosa (SCHRENK) R. WIGHT (Orobanchaceae) is a perennial parasitic plant growing on the roots of Salvadora or Calotropis species, and distributed in North African, Arabian, and Asian countries. Dried stems of the C. tubulosa were powdered and extracted three times with methanol under reflux for 3 h. Evaporation of the solvent under reduced pressure provided the methanolic extract. The methanolic extract (commercial grade, Batch No. 20070130;
register trade name, Sabaku Ninnjinn Kanka) was a generous gift from Eishin Trading Co., Ltd via Muraoka and Morikawa (Kinki University, Japan), and a botanical identifies- cation was undertaken by Professor Jia Xiaoguang in the Xinjiang Institute of Traditional Chinese and Ethnologic Medicines.
Plant extract analysis: chromatography
We determined ECH and ACT contents in the C. tubulosa extract (Batch No. 20070130) by an HPLC analysis described below. The data obtained are shown in Table 1.

Cell culture
Caco-2 cells, purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA), were used at passages 38–53. They were grown in a culture medium consisting of Dulbecco's modified Eagle's medium (DMEM, Nacalai Tesque Co., Kyoto, Japan) supplemented with 0.1 mM non-essential amino acids, 10% heat-inactivated fetal bovine serum, 100 U/ml penicillin G, and 0.1 mg/ml streptomycin sulfate.
Transport studies
Caco-2 cells were plated at a density of 6.4 × 103 cells/cm2 on polycarbonate filters. Monolayers were utilized for transport experiments 21–25 days after seeding. Intact ECH and ACT that were equivalent to their contents in Cistanche tubulosa extract (4.5 and 13.5 mg/ml) were mixed with DMEM medium containing 0.5% dimethylsulfoxide to maintain the integrity of the cell monolayer over the periods of the experiments. Intact ACT equivalent to ECH content in the extract was also dosed in the incubation medium. The extract was suspended in a DMEM medium and was centrifuged to remove insoluble components. Supernatants were loaded to the apical side. At the indicated times, an aliquot of the incubation medium was withdrawn from the basolateral side and was mixed with acetonitrile containing an internal standard for the assay. In separate experiments, phloridzin (fifinal concentration, 1 mM) and verapamil (fifinal concentration, 0.2 mM) was added to the apical side of the monolayer; however, phloretin (fifinal concentration, 0.3 mM) was treated on both sides of the monolayer. The integrity of monolayers was monitored by transepithelial electrical resistance (TEER) using Millicell-ERS (Millipore, Bedford, MA, USA) before and after transport experiments. TEER values of monolayers used were >300 Ω·cm2.

In-situ intestinal perfusion
Male Wistar rats (230–250 g) were obtained from SLC Japan (Hamamatsu, Japan). Animals were housed in an air-conditioned room under a 12 h light/dark cycle for 1 week before use. Rats were fed standard laboratory food (Oriental Yeast Co., Ltd., Tokyo, Japan) with water ad libitum and were fasted overnight before the test. The in-situ recirculating perfusion study was performed according to the modified procedure described by Mihara et al. [20] Briefly, rats were anesthetized with 25% urethane solution (1 mg/kg) to avoid decreases in blood pressure. A midline abdominal incision was made and the small intestine was exposed. The bile duct was ligated to avoid bile secretion into the perfusate. The whole small intestine as one segment (from the duodenum to the ileum) was rinsed with normal saline at 37°C for 10 min until the washing appeared clear. Glass tubings connected to silicone tubing were then cannulated into both ends of the small intestine and secured with suture thread. Then, the small intestine was replaced in the abdomen, and the cannulas were connected to a peristaltic pump. The portal vein was cannulated with polyethylene tubing (PE10). C. tubulosa extract available commercially was suspended in Krebs– Henseleit bicarbonate buffer (pH 7.4) to yield a fifinal concentration of 4.5 mg/ml and was centrifuged for 10 min at 8000 rpm to remove insoluble components. Supernatant in the absence or presence of phloridzin (1 mM) was recollected into a reservoir, which was maintained at a temperature of 37 ± 0.5°C throughout the course of the experiment. At the indicated times, blood was taken through the portal vein cannula. After centrifuging blood samples, the resulting plasma was deproteinized with acetonitrile containing the internal standard and was centrifuged at 3000 rpm. Supernatants were evaporated, and the residue was resolved with a mobile phase consisting of acetonitrile and 0.5% acetic acid. The mixed solution was loaded onto an HPLC column. Rats were used in accordance with ethical procedures following the Guidelines for the Care and Use of Laboratory Animals issued by the Japanese government and Kinki University.
HPLC analysis
HPLC analysis was performed on a system equipped with a Shimadzu SPD-10A, UV detector, Shimadzu LC-10A pump, and Shimadzu C-R4A chronotopic integrator (Kyoto, Japan). ECH and ACT were separated using an Inertsil ODS column (5 μm, 4.6 × 150 mm, GL Sciences Inc., Osaka, Japan). A mobile phase of acetonitrile and 0.5% acetic acid at a ratio of 15:85 (v/v) was used at a flow rate of 1.0 ml/ min. Detection was performed at 334 nm.
Kinetic analysis
Apparent permeability coefficients (Papp) were estimated from the slope of the linear portion of the time course of compound transport across Caco-2 cell monolayers, as follows:
Papp= (dQ/dt)/ A1C0)
where dQ/dt is the permeability rate, C0 is the initial concentration of the solute in the donor chamber, and A is the surface area of the membrane (4.7 cm2 ).
In the rat in-situ intestinal perfusion study, the area under the plasma concentration-time curve (AUC0–90) in the portal vein from time zero to the last measured was calculated according to the linear trapezoidal rule.
Physicochemical properties
The polar surface area and non-polar surface area of compounds were calculated using the program SAS (version 0.8, Olsson, T.; Sherbukhin, V., Synthesis and Structure Administration, 1997–2001, AstraZeneca, Cary, NC, USA). Experimentally determined log P and pKa values were obtained from the literature.

Statistical analysis
Data were analyzed by one-way analysis of variance followed by Tukey's posthoc test. Probability values less than 5% were considered significant.
Results
Absorptive transport of echinacoside and acteoside through Caco-2 cell monolayers
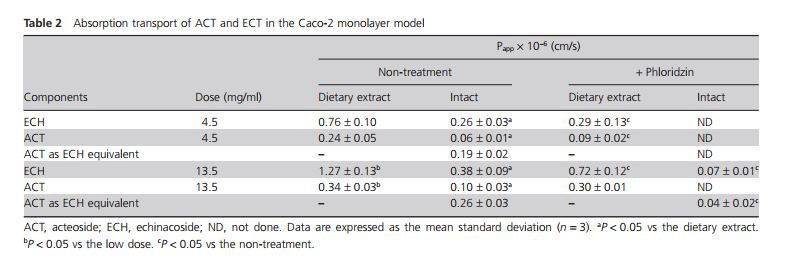
In mice and rats, intact ECH[10,14] and ACT[12,21] are orally administered at doses of 100–1000 mg/kg. The C. tubulosa extract used contained approximately 30% ECH and 15% ACT per dose. Since the extract changed osmotic pressure and pH in the incubation medium, concentrations of 4.5 and 13.5 mg/ml were determined based on the oral dosage (intact compounds: 2–20 mg/20 g body weight) in mice. The extract at low (4.5 mg/ml) and high doses (13.5 mg/ml) contained 2.0 and 6.1 mg for ECH and 1.0 and 3.0 mg for ACT, respectively. We applied C. tubulosa extract amounts that were much lower than the oral dosage of ECH and ACT reported in humans (recommended dietary allowance of extract: 150 mg containing approximately 45 mg for ECH and 22.5 mg for ACT). At low and high doses of intact compounds, absorption profiles (Figure 2) and Papp were not significantly different between ECH and ACT as an ECH equivalent (Table 2). When C. tubulosa extract at a high dose of 13.5 mg/ml was loaded into the medium, Papp values (1.27 ± 0.13 and 0.34 ± 0.03 × 10−6 cm/s, respectively) of ECH and ACT concomitants were threefold higher than those (0.38 ± 0.09 and 0.10 ± 0.03 × 10−6 cm/s, respectively) of intact ECH and ACT (Table 2). The extract, unlike intact compounds, significantly enhanced the absorption transport of ECH and ACT.

Figure 2 Absorptive transport of echinacoside and acteoside across Caco-2 cell monolayers in a transwell system. Apical to basolateral transport was monitored. Closed symbols are echinacoside (circle) and acteoside (square) from Cistanche tubulosa extract dosed at low and high concentrations of 4.5 (a) and 13.5 mg/ml (b). Open symbols are intact echinacoside (circle) and intact acteoside (square) corresponding to echinacoside and acteoside contents in Cistanche tubulosa extract dosed, respectively. Intact acteoside (open triangle) was also loaded in the medium as a dose equivalent to intact echinacoside (open circle). Results are given with standard deviations (n = 3).
Inhibitory effect of phloridzin, phloretin, and verapamil
To characterize the intestinal absorption of ECH and ACT, Caco-2 cell monolayers were incubated with representative inhibitors. Apical glucose transporter 1-sensitive phloridzin dramatically reduced the Papp of intact ECH and ACT to 20% of non-treatment at the high dose (Table 2). Basolateral glucose transporter (GLUT) 2-sensitive phloretin did not decrease the transport of intact ECH and ACT (Figure 3). In this study, higher concentrations (>0.3 mM) of phloretin could not be used because of noticeable cell toxicity. Additionally, P-gp has been identifified as an important player responsible for the interaction between herbal medicines and clinically important P-gp substrates. Verapamil did not enhance the absorptive transport of intact compounds (Figure 3).
The absorptive transport of ECH and ACT in the extract (low dose) was significantly inhibited by phloridzin (Table 2 and Figure 4). The extract at the high dose suppressed phloridzin-sensitive inhibition, although the transport of intact ECH and ACT was more sensitive to phloridzin (Table 2).

Figure 3 Effect of phloretin and verapamil on the absorptive transport of intact echinacoside and acteoside. Apical to basolateral transport was monitored after applying intact echinacoside corresponding to echinacoside content in a 13.5 mg/ml extract on the apical side (n = 3). Acteoside (closed square) was equivalent in dosage to intact echinacoside (closed circle) in the absence of inhibitors (n = 3). Open and closed diamonds show transport in the presence of 0.2 mM verapamil and 0.3 mM phloretin, respectively. Inhibition experiments were performed in duplicate.
The in-situ intestinal perfusion study
In an in-situ study, we tested whether ECH and ACT in C. tubulosa extract were transported by SGLT1 located on the apical side of the small intestine. When the dietary extract at the low dose (4.5 mg/ml) was perfused, ECH and ACT quickly appeared in portal blood (Figure 5). The AUC was determined as 2702.8 ± 384.1 μm·min for ECH and 698.3 ± 197.2 μm·min for the ACT. After the AUC was normalized with content from C. tubulosa extract, the absorbed amount was not significantly different between ECH and ACT. SGLT1-sensitive phloridzin, unlike phloretin, significantly suppressed the absorptive transport of concomitant ECH (AUC, 649.4 ± 248.2 μm·min) and ACT (not detected).

Discussion
Some herbal ingredients are substrates of P-gp highly expressed in the liver, intestine, brain, and kidneys. P-gp is a determining factor for in-vivo bioavailability, disposition, and distribution of herbal remedies, including St John’s wort, curcumin, echinacea, ginseng, ginkgo, and ginger.[22,23] The bioavailability of genistein-7-glucoside, a flavonoid derivative, was also limited by the intestinal MRP2 transporter.[24] Therefore, this study was designed to investigate the absorption properties of ECH and ACT concomitant in dietary and medicinal C. tubulosa extract.
Polarized Caco-2 cell monolayers, as well as the intestine,[25], express major intestinal drug efflflux transporters, such as P-gp, MRPs, and breast cancer resistance protein.[26] Dietary flavonoids of quercetin[27] and myricetin[28] have been shown to inhibit P-gp-mediated efflflux both in the cell lines and animal models. Verapamil, a P-gp inhibitor, did not alter the permeability of ACT and ECH across Caco-2 cell monolayers (Figure 3), indicating that intact ECH and ACT were not limited by the P-gp efflflux pump. Our previous studies showed that MRP2 proteins were not expressed in Caco-2 cell monolayers.[29] P-gp and MRP2-mediated efflflux could be excluded in ECH and ACT transport. Some glycosides of quercetin with low lipophilicity were more efficiently absorbed than quercetin itself.[30] It is also important to note that ACT with a sugar moiety is rapidly distributed in brain tissues. Our attention has been focused on the combined action of two glucose transporters in enterocytes: SGLT in the brush-border membrane and facilitated diffusion glucose transport (GLUT) in the basolateral membrane. Caco-2 cell culture can be used as a model for studying phloretin-sensitive GLUT2, and phloridzin-sensitive SGLT1 and 2 transporters.[31–34] Glucose is transported from the apical to the basolateral side of Caco-2 monolayers at a high rate with a Papp of 36.8 ± 1.1×10−6 cm/s.[35] It possesses a higher Papp than the transcellular transport marker propranolol (23.4 ± 2.8 × 10−6 cm/s). As shown in Table 2, intact ECH and ACT had much lower Papp than that reported in glucose and passive propranolol. We calculated the logarithm of the partition coeffificient (octanol-water), log P, was calculated to be −2.32 and 0.077 for ECH and ACT, respectively. Polar or hydrophilic compounds are believed to be transported via a paracellular pathway (across tight junctions). The two phenylethanoid glycosides, like mannitol, appear to be transported through a paracellular route. However, phloridzin dramatically reduced the absorptive permeability of intact ECH and ACT (Table 2), suggesting that apical SGLT1 plays a major role in the intestinal absorption of intact ECH and ACT. At an equivalent dose, higher hydrophobic ACT permeability was close to ECH permeability (Figure 2 and Table 2). Yoshikawa et al. [36] have demonstrated that facilitative transporters (GLUT 1 and 2), as well as phloridzin-sensitive SGLT1, are intensively expressed in the small intestine. Since absorbed amounts of compounds are based on the mass balance between uptake and elimination, we evaluated the participation of GLUT2. Glucose crosses the apical membranes of enterocytes by SGLT1 with high affinity and low capacity and exits across the basolateral membrane through GLUT2 with low affinity and high capacity. Phloretin (a specific inhibitor of GLUT2) did not abolish the transport of intact ECH and ACT (Figure 3). Funes et al. [37] demonstrated that ACT strongly interacted with the phosphate groups of phospholipid membranes. As hydroxyl groups are abundant in the ACT structure, hydrogen bonds between these groups and the glycerol polar heads or phosphate groups of phospholipids are the most likely interactions to take place. When intact ECH and its equivalent ACT were incubated with Caco-2 monolayers for 11 h, cellular accumulation of ACT (0.24 ± 0.04 nmol/cm2 ) was threefold greater than that of ECH (0.07 ± 0.01 nmol/cm2 ). We thought that SGLT1- sensitive ECH and ACT were slowly moved from enterocytes to the bloodstream, possibly leading to the low Papp observed. Compared with highly hydrophilic ECH, the low permeability of ACT may be due to intercalation into cell membranes.
Polyphenolic compounds are consumed in herbal mixtures during their clinical application and are commercially available as dietary supplements. In an in-vitro study, it was shown that the absorption of phenolic epicatechin was not influenced by the ingredient composition of beverage food materials.[38] In contrast, Hypericum perforatum L. product matrices affect the transport of quercetin glucosides (rutin and isoquercitrin) and hyperoside across Caco-2 cells due to differences in matrix phytochemical composition and transport characteristics, i.e. paracellular transfer and carrier-mediated or active transport.[39] In this study, C. tubulosa provided a threefold higher transepithelial transport than intact ECH and ACT (Figure 2 and Table 2). We speculate that components in the C. tubulosa extract activate the phloridzin-sensitive transporter and/or accelerate the elimination of intracellular ECH and ACT. C. tubulosa extract at the high dose seemed to greatly mask the potency of phloridzin-sensitive transport (Table 2). Dietary carbohydrates[40] and proteins[41] interact with some polyphenols in the gastrointestinal tracts. Morikawa et al. [10] demonstrated that five iridoids, kankanosides A-D, and kankanol, a monoterpene glycoside, kankanoside E, two phenylethanoid oligoglycosides, kankanosides F and G, and an acylated oligo sugar, kankanose, could be isolated from the C. tubulosa extract presently used. Other ingredients, including proteins in the C. tubulosa extract, remain unclear. Together with the above speculation, we are designed to examine whether other components interact with SGLT1 and inhibit the absorption of ECH and ACT.
In-vivo experiments cannot easily distinguish between the extent of absorption and avoidance of first-pass disposition through the liver. The in-situ intestinal perfusion model has an advantage over in-vivo and in-vitro models due to the easy control of experiment parameters exclusion of the impact of other organs and maintenance of an intact intestinal blood supply.[22] The involvement of the phloridzin-sensitive glucose transporter was evaluated in an in-situ intestinal perfusion system. As shown in Figure 5, absorbed amounts of ECH and ACT concomitants in C. tubulosa extract (low dose) were greatly abolished by phloridzin, which agrees with our in-vitro data (Figure 4). Using peptides and 20 drugs passively absorbed, a good correlation is obtained between in-vivo drug absorption and the drug permeability of Caco-2 monolayers.[42] Drugs with a Papp of >1 × 10−6 cm/s are completely absorbed in humans, while poorly absorbed drugs and peptides (<1% of dose) have Papp values of <1 × 10−7 cm/s. Surprisingly, the Papp of the ECH concomitant (high dose) was >1 × 10−6 cm/s (Table 2), suggesting high oral bioavailability in animals and humans. Crespy et al. [43] demonstrated that efflflux in an in-situ intestinal perfusion study was not significantly different between phloridzin and phloretin. They[44] also showed that oral bioavailability of phloridzin with high sensitivity to SGLT1 was only 10% in rats. Future studies need to evaluate the bioavailability and hepatic first-pass effect of the ECH concomitant after oral administration of the dietary extract at the high dose. In-situ results imply that the intake of C. tubulosa extract may improve the low oral absorption of intact ECH and ACT.

Figure 4 Inhibitory effect of phloridzin on the absorptive transport of echinacoside and acteoside in Cistanche tubulosa extract. Apical to basolateral transport was monitored. Closed circles and squares are echinacoside (a) and acteoside (b) in the 4.5 mg/ml extract without phloridzin, respectively. Closed diamonds show the treatment with 4.5 mg/ml extract including 1 mM phloridzin. Results are given with standard deviations (n = 3).

Figure 5 Time courses of echinacoside and acteoside concentrations in portal blood during in-situ recirculating rat intestinal perfusion. Circle and square symbols are echinacoside and acteoside, respectively. Cistanche tubulosa extract at a concentration of 4.5 mg/ml was perfused in the absence (closed symbols) or presence (open symbols) of 1 mM phloridzin at 37°C. Results are given with standard deviations (n = 3–4). *P < 0.05 vs the dietary extract in the presence of phloridzin.
Conclusion
The dietary and medicinal C. tubulosa extract enhancing the intestinal absorption of ECH and ACT may serve to better manage human health, although the involvement of phloridzin-sensitive transport should be reduced.
Declarations Conflict of interests
The Author(s) declare(s) that they have no conflicts of interest to disclose.
Funding
This work was in part supported by the High-Tech Research Center from Kinki University.
Acknowledgments
The authors wish to thank Osamu Muraoka (Kinki University, Osaka, Japan) and Toshio Morikawa (Kinki University, Osaka, Japan) for the supply of Cistanche tubulosa extract and pure constituents. We are very grateful to Masahiro Iwaki (Kinki University) for their study support.

References
1. Tanaka J et al. Effect of Cistanche tubulosa extract on various brain diseases. Food Style 21 2008; 12: 24–26.
2. Tanaka J et al. Anti-aging functions of Cistanche tubulosa extract. Food Style 21 2008; 12: 27–29.
3. Tanaka J et al. Beauty and hair growth functions of Cistanche tubulosa extract. Food Style 21 2008; 12: 29–32.
4. Tanaka J et al. Fat metabolizing effect of Cistanche tubulosa extract. Food Style 21 2008; 12: 30–33.
5. Yoshizawa F et al. The constituents of Cistanche tubulosa Schrenk (Hook) f.II. isolation and structure of a new phenylethanoid glycoside and a new neolignan glycoside. Chem Pharm Bull 1990; 38: 1927–1930.
6. Yoshikawa M et al. Phenylethanoid aminoglycosides and acylated oligosugars with vasorelaxant activity from Cistanche tubulosa. Bioorg Med Chem 2006; 14: 7468–7475.
7. Tu PF et al. Analysis of phenylethanoid glycosides of Herba cistanche by RP-HPLC. Yao Xue Xue Bao 1997; 32: 294–300.
8. Lei L et al. Metabolic regulation of phenylethanoid glycosides from Herba cistanches in dog’s gastrointestinal. Yao Xue Xue Bao 2001; 36: 432–435.
9. Geng X et al. Neuroprotective effects of echinacoside in the mouse MPTP model of Parkinson’s disease. Eur J Pharmacol 2007; 564: 66–74.
10. Morikawa T et al. Acylated phenylethanoid aminoglycosides with hepatoprotective activity from the desert plant Cistanche tubulosa. Bioorg Med Chem 2010; 18: 1882–1890.
11. Paola RD et al. Effects of verbascoside, biotechnologically purified by syringa Vulgaris plant cell cultures, in a rodent model of periodontitis. J Pharm Pharmacol 2011; 63: 707–717.
12. Wu YT et al. Determination of acteoside in Cistanche deserticola and Boschniakia rossica and its pharmacokinetics in freely-moving rats using LC-MS/MS. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 844: 89–95.
13. Matthias A et al. Permeability studies of alkylamides and caffeic acid conjugates from echinacea using a caco-2 cell monolayer model. J Clin Pharm Therapeut 2004; 29: 7–13.
14. Jia C et al. Determination of echinacoside in rat serum by reversed-phase high-performance liquid chromatography with ultraviolet detection and its application to pharmacokinetics and bioavailability. J Chromatogr 2006; 844: 308–313.
15. Cardinali A et al. Verbascosides from olive mill water: assessment of their bioaccessibility and intestinal uptake using an in vitro digestion/caco-2 model system. J Food Sci 2011; 176: H48–H54.
16. Jia C et al. Metabolism of echinacoside, a good antioxidant, in rats: isolation and identification of its biliary metabolites. Drug Metab Dispos 2009; 37: 431–438.
17. Najar IA et al. Modulation of P-glycoprotein ATPase activity by some phytoconstituents. Phytother Res 2009; 24: 454–458.
18. Walgren RA et al. Efflux of dietary flavonoid quercetin 4′-beta-glucoside across human intestinal caco-2 cell monolayers by apical multidrug resistance-associated protein-2. J Pharmacol Exp Ther 2000a; 294: 830– 836.
19. Walgren RA et al. Cellular uptake of dietary flavonoid quercetin 4′-beta-glucosidase by sodium-dependent glucose transporter SGLT1. J Pharmacol Exp Ther 2000b; 294: 837– 843.
20. Mihara K et al. Intestinal first-pass metabolism of eperisone in the rat. Pharm Res 2001; 18: 1131–1137.
21. Isacchi B et al. Antihyperalgesic activity of verbascoside in two models of neuropathic pain. J Pharm Pharmacol 2011; 63: 594–601.
22. Cook TJ et al. Intestinal permeability of chlorpyrifos using the single-pass intestinal perfusion method in the rat. Toxicology 2003; 184: 125–133.23. Kumar YS et al. P-glycoprotein- and cytochrome P-450-mediated herbal drug interaction. Drug Metabol Drug Interact 2010; 25: 3–16.
24. Walle UK et al. Transport of genistein- 7-glucoside by human intestinal CACO-2 cells: a potential role for MRP2. Res Commun Mol Pathol Pharmacol 1999; 103: 45–56.
25. Ito K et al. Apical/basolateral surface expression of drug transporters and its role in vectorial drug transport. Pharm Res 2005; 22: 1559–1577.
26. Laitinen L et al. Caco-2 cell cultures in the assessment of intestinal absorption: effects of some co-administered drugs and natural compounds in biological matrices. (University of Helsinki, Finland, 2006) Academic Dissertation, pp. 1–66.
27. Scambia G et al. Quercetin potentiates the effect of adriamycin in a multidrug-resistant MCF-7 human breast cancer cell line: P-glycoprotein as a possible target. Cancer Chemother Pharmacol 1994; 34: 459– 464.
28. Choi DH et al. Effect of myricetin, an antioxidant, on the pharmacokinetics of losartan and its active metabolite, EXP-3174, in rats: possible role of cytochrome P450 3A4, cytochrome P450 2C9 and P-glycoprotein inhibition by myricetin. J Pharm Pharmacol 2010; 62: 908–914.
29. Tanino T et al. Paclitaxel-2′- ethyl carbonate prodrug can circumvent P-glycoprotein-mediated cellular efflux to increase drug cytotoxicity. Pharm Res 2007; 24: 555–565.
30. Hollman PC et al. Absorption of dietary quercetin glycosides and quercetin in healthy ileostomy volunteers. Am J Clin Nutr 1995; 62: 1276– 1282.
31. Kellett GL et al. The diffusive component of intestinal glucose absorption is mediated by the glucose-induced recruitment of GLUT2 to the brush board membrane. Biochem J 2000; 350: 155–162.
32. Matter K et al. Sorting of endogenous plasma-membrane proteins occurs from two sites in cultured human intestinal epithelial cells (Caco-2). Cell 1990; 60: 429–437.
33. Mahraoui L et al. Presence and differential expression of SGLT1, GLUT1, GLUT2, GLUT3, and GLUT5 hexose transporter mRNAs in Caco-2 cell clones in relation to cell growth and glucose consumption. Biochem J 1994; 298: 629–633.
34. Mesonero J et al. Sugar-dependent expression of the fructose transporter GLUT 5 in Cac-2 cells. Biochem J 1995; 312: 757–762.
35. Walgren RA et al. Transport of quercetin and its glucosides across human intestinal epithelial Caco-2 cells. Biochem Pharmacol 1998; 55: 1721–1727.
36. Yoshikawa T et al. Comparative expression of hexose transporters (SGLT1, GLUT1, GLUT2, and GLUT5) throughout the mouse gastrointestinal tract. Histochem Cell Biol 2011; 135: 183–194.
37. Funes L et al. Effects of verbascoside, a phenylpropanoid glycoside from lemon verbena, on phospholipids model membranes. Chem Phys Lipids 2010; 163: 190–199.
38. Neilson AP et al. Influence of chocolate matrix composition on cocoa flavan-3-ol bioaccessibility in vitro and bioavailability in humans. J Agric Food Chem 2009; 57: 9418–9426.
39. Gao S et al. Highly variable contents of phenolics in St. John’s wort products affect their transport in the human intestinal Caco-2 cell model: pharmaceutical and biopharmaceutical rationale for product standardization. J Agric Food Chem 2010; 58: 6650–6659.
40. Schramm DD et al. Food effects on the absorption and pharmacokinetics of cocoa flavanols. Life Sci 2003; 73: 857–869.
41. Laurent C et al. Ethanol and polyphenolic free wine matrix stimulate the differentiation of human intestinal Caco-2 cells. Influence of their association with a procyanidin-rich grape seed extract. J Agric Food Chem 2005; 53: 5541–5548.
42. Artursson P et al. Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human internal epithelial (Caco-2) cells. Biochem Biophys Res Commun 1991; 175: 880–885.
43. Crespy V et al. Comparison of the intestinal absorption of quercetin, phloretin, and their glucosides in rats. J Nutr 2001a; 131: 2109–2114.
44. Crespy V et al. Bioavailability of phloretin and phloridzin in rats. J Nutr 2001b; 131: 3227–3230.







