Thyroid Hormone Replacement Therapy For Primary Hypothyroidism Leads To Significant Improvement Of Renal Function in Chronic Kidney Disease Patients
Mar 14, 2022
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791
Yuji Hataya et al
Abstract
Background The interactions between kidney and thyroid functions have been known for many years; however, there are few studies on the extent of the improvements and long-term changes of renal function after thyroid hormone replacement therapy (THRT) in chronic kidney disease (CKD) patients. The purpose of this study was to determine how THRT affects the estimated glomerular filtration rate (eGFR) in CKD (chronic kidney disease) patients with primary hypothyroidism. Methods A retrospective investigation was performed on 51 Japanese patients (15 men and 36 women) with primary hypothyroidism. The changes in eGFR after THRT were examined according to the existence of CKD and the severity of thyroid function.
Results eGFR increased rapidly over the first 6 months after THRT in CKD (chronic kidney disease) patients, which was followed by a plateau. There was a correlation between eGFR and the severity of hypothyroidism, which was independent of age, and eGFR in severely hypothyroid patients significantly increased up to levels that were similar to mildly hypothyroid patients after THRT. eGFR improved more in the lower initial eGFR group and increased about 30 % in CKD (chronic kidney disease) patients (47.5 ± 7.7 vs. 62.1 ± 9.5 ml/min/1.73 m2, P< 0.01). Moreover, eGFR in CKD (chronic kidney disease) patients with mild to moderate hypothyroidism was significantly increased compared to that in non-CKD patients.
Conclusion Our data suggested that hypothyroidism contributed to the reduction in eGFR, especially in CKD (chronic kidney disease) patients; therefore, patients with CKD (chronic kidney disease) should positively be examined for thyroid function, and appropriate THRT should be started if needed.
Keywords Thyroid hormone, Hypothyroidism, Chronic kidney disease, glomerular filtration rate

cistanche is good for chronic kidney disease
Introduction
The interactions between kidney and thyroid functions have been known for many years, and thyroid dysfunction causes significant changes in kidney function [1]. Patients with hypothyroidism, both overt and subclinical, are characterized by a decrease in the glomerular filtration rate (GFR) and renal plasma flow, resulting in increased serum creatinine [2–4]. Although it has been reported that these changes can be reversed with levothyroxine administration[5], there have been few studies of the extent of the improvements and long-term changes of renal function after thyroid hormone replacement therapy (THRT) in chronic kidney disease (CKD) patients.
On the other hand, CKD (chronic kidney disease) is associated with a high prevalence of primary hypothyroidism. Lo, et al. reported that the prevalence of hypothyroidism increased with progressively lower levels of kidney function in a nationally representative cohort of US adults and that about 20 % of subjects with estimated GFRs (eGFRs)<60 ml/min/1.73 m2 had laboratory or clinical evidence of hypothyroidism [6]. The kidney contributes to iodine clearance primarily through glomerular filtration. High serum iodine concentrations have been reported in CKD patients [7], and high exposure to iodine may facilitate the development of hypothyroidism [8]. Because most Japanese people consume excessive amounts of iodine, it is possible that iodine has a stronger influence on hypothyroidism in the Japanese population. A retrospective investigation was therefore performed to examine whether THRT affects eGFR in Japanese patients with primary hypothyroidism.
Materials and methods
Study population
Fifty-one patients (15 men and 36 women) with primary hypothyroidism who visited the Department of Endocrinology at Kyoto City Hospital during 2002–2010 were analyzed retrospectively. All patients received THRT with 25–150 lg levothyroxine daily to restore euthyroidism. We excluded subjects who had received thyroidectomies or radioiodine therapy, subjects who were taking antithyroid drugs (methimazole or propylthiouracil) for hyperthyroidism, and subjects who had already received THRT. Comorbid illness was considered positive if subjects met any of the following disease criteria or if they had been treated for any of the following: diabetes mellitus, hemoglobin A1c C6.5 %; dyslipidemia, fasting low-density lipoprotein C140 mg/dl, and/or fasting triglyceride C150 mg/dl; hypertension with systolic blood pressure C140 mmHg and/or diastolic blood pressure C90 mmHg. The backgrounds and characteristics of both groups are shown in Table 1.

Laboratory measurements and assessments of kidney function
Free triiodothyronine (FT3), free thyroxine (FT4), and thyrotropin (TSH) were measured by electrochemiluminescence immunoassays (ARCHITECT Free T3, ARCHITECT Free T4, and ARCHITECT TSH, respectively; Abbott Japan Co., Ltd., Tokyo, Japan). The normal reference ranges for FT3, FT4, and TSH in our institute are 1.71–3.71 pg/ml, 0.70–1.48 ng/dl, and 0.35–4.94 μIU/ml, respectively. Mild hypothyroidism was defined as FT4>0.8 ng/dl and TSH <30 μIU/ml; moderate hypothyroidism was defined as FT4 0.5–0.8 ng/dl and TSH 30–79μIU/ml; severe hypothyroidism was defined as FT4 <=0.4 ng/dl and TSH >=80 μIU/ml. Serum creatinine levels were also measured with an enzymatic method, and the GFR was estimated according to the Japanese Society of Nephrology CKD (chronic kidney disease) Practice Guide: eGFR (ml/min/1.73 m2) = 194 x (serum creatinine levels [mg/dl])-1.094 x (age [year])-0.287 x (0.739 if female) [9]. CKD (chronic kidney disease) was defined as eGFR<60 ml/min/1.73 m2. The association of CKD (chronic kidney disease) was explored according to the following eGFR categories: eGFR >=90, 60–89, and 30–59 ml/min/1.73 m2.
Statistical analysis
Continuous variables are expressed as the mean ± standard deviation. Categorical variables are presented as numbers. Continuous variables of groups were compared with the unpaired t-test, and categorical variables were compared with the chi-squared test. The paired t-test was used to compare eGFR between baseline and after treatment. The relationship between thyroid function and eGFR was explored with linear regression analyses. Multiple regression analysis was also performed in order to elucidate the effects of thyroid function on eGFR independent of age. For comparisons among different groups, statistical significance was determined using one-way ANOVA, followed by post hoc comparisons of the group means according to Tukey's method. P<0.05 was considered significant. All statistical analyses were performed using the IBM SPSS Statistics program, version 20 (IBM Corp., Armonk, NY, USA).
Results
Patients’ characteristics
Table 1 shows the clinical characteristics of the subjects according to eGFR. There were no patients with eGFR <30 ml/min/1.73 m2. There were no differences between the non-CKD (chronic kidney disease) and CKD groups for gender proportions, body mass index, comorbid illness, antithyroid antibodies, and levothyroxine doses. The subjects in the CKD (chronic kidney disease) group were significantly older than those in the non-CKD group (65.3 ± 14.0 vs. 54.7 ± 18.7 years, P\ 0.05). Serum FT3 and FT4 were significantly lower in the CKD group than in the non-CKD group (FT3: 1.79 ± 0.81 vs. 2.28 ± 0.76 pg/ ml, P< 0.05; FT4: 0.45 ± 0.20 vs. 0.63 ± 0.27 ng/dl, P< 0.01). Serum TSH was significantly higher in the CKD group than in the non-CKD group (128.9 ± 145.2 vs. 55.2 ± 56.1μIU/ml, P<0.05).
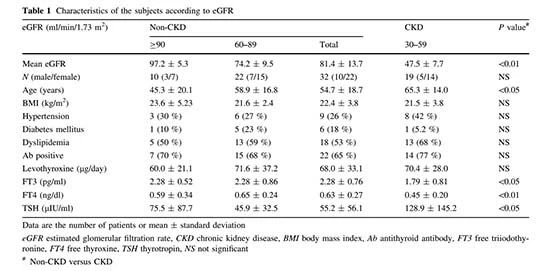
The natural course of eGFR after THRT in CKD (chronic kidney disease) patients
To clarify the natural course of eGFR after THRT, a longitudinal analysis was performed. Figure 1 shows the changes in eGFR from baseline to 36 months after treatment. The eGFR increased rapidly over the first 6 months with the decrease of TSH in CKD (chronic kidney disease) patients, which was followed by a plateau. Thus, we compared eGFR between baseline and 6 months after treatment in the following examinations.


Relationship between eGFR and thyroid hormone
Linear regression analysis was performed to explore the relationship between eGFR and thyroid function. There were positive relationships between eGFR and FT4 (eGFR = 26.120 9 FT4 ? 54.455, r = 0.350, P< 0.01, Fig. 2a). A negative relationship was found between eGFR and serum TSH (eGFR = -13.975 9 log TSH + 92.691, r = 0.337, P<0.05, Fig. 2b). When the analysis was performed using multiple regressions, eGFR was found to be positively related to serum FT4 and log TSH, respectively; this was independent of age (FT4: b = 0.344, P = 0.006, log TSH: b = -0.410, P = 0.001, Table 2). Figure 2c, d shows the difference in eGFR according to thyroid function at baseline and 6 months after treatment. eGFR significantly increased after THRT in patients with moderate and severe hypothyroidism. Moreover, the decreased eGFR at baseline in these patients increased up to the level of patients with mild hypothyroidism.
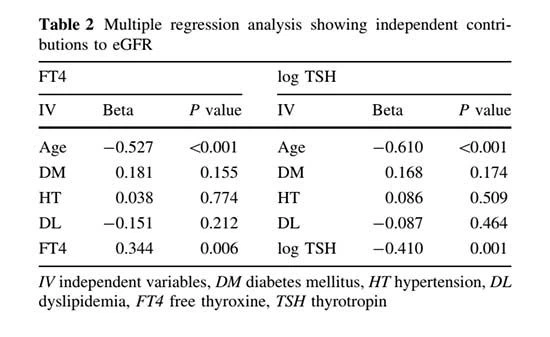


Fig. 2 Linear regression analyses between eGFR and free thyroxine (FT4) and b TSH at baseline. The effect of thyroid hormone replacement therapy (THRT) on eGFR according to thyroid function (c FT4; d TSH) at baseline (open bar) and 6 months after
treatment (filled bar). Values are the mean ± SD. r = correlation coefficient. **P <0.01 versus eGFR at baseline
Influence of THRT on eGFR
Figure 3 shows the difference in eGFR after treatment according to the initial eGFR values. The group with lower initial eGFR values had more improvement (>=90 ml/min/ 1.73 m2 group: 98.0 ± 5.0 vs. 100.9 ± 10.1 ml/min/ 1.73 m2, P = 0.24; 60–89 ml/min/1.73 m2 group: 74.2 ± 9.5 vs. 80.9 ± 12.7 ml/min/1.73 m2, P< 0.05; and 30–59 ml/min/1.73 m2 group: 47.5 ± 7.7 vs. 62.1 ± 9.5 ml/min/1.73 m2, P<0.01). Finally, Fig. 4 shows the changes in FT4, TSH, and eGFR after THRT according to CKD or non-CKD (chronic kidney disease) and severe hypothyroidism or mild to moderate hypothyroidism. There were positive relationships between changes in eGFR and FT4 (DeGFR =18.574 9 DFT4 - 1.919, r = 0.528, P< 0.01, Fig. 4a) and in eGFR and TSH (△eGFR = 8.558 9 log DTSH -3.963, r = 0.437, P<0.01, Fig. 4b). The changes in FT4 and TSH in the patients with severe hypothyroidism were significantly greater than in the patients with mild to moderate hypothyroidism; however, there was no significant change between CKD (chronic kidney disease) and non-CKD groups (Fig. 4c, d). eGFR in the non-CKD (chronic kidney disease) group with severe hypothyroidism was significantly increased compared to the group with mild to moderate hypothyroidism, and the changes of eGFR in the CKD (chronic kidney disease) group showed a similar tendency, but not significantly (Fig. 4e, f). Furthermore, eGFR in the CKD (chronic kidney disease) group with mild to moderate hypothyroidism was significantly increased compared to that in the non-CKD group (FT4>0.4 ng/dl: DeGFR, 11.3 ± 6.1 vs. 2.8 ± 10.8 ml/min/1.73 m2, P<0.05; TSH\80 μIU/ml: DeGFR, 11.7 ± 5.6 vs. 3.3 ± 14.2 ml/min/1.73 m2, P<0.01).

Fig. 3 The eGFR according to initial eGFR at baseline (open bar) and 6 months after treatment (filled bar). Values are the mean ± SD. *P<0.05 and **P <0.01 versus eGFR at baseline

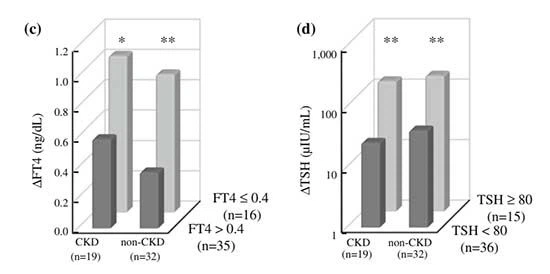

Fig. 4 Linear regression analyses between changes in eGFR and a FT4 and b TSH after THRT (a DeGFR = 18.574 9 DFT4 - 1.919, r = 0.528, P <0.01; b DeGFR = 8.558 9 log DTSH - 3.963, r = 0.437, P<0.01). Changes in FT4 (c), TSH (d), and eGFR (e, f) after THRT according to the CKD (chronic kidney disease) or non-CKD (chronic kidney disease) groups and severe hypothyroidism (FT4 B0.4 ng/dl and TSH C80 μIU/ml) or mild to moderate hypothyroidism (FT4 [0.4 ng/dl and TSH <80 μIU/ml). Values are the mean ± SD. *P<0.05 and **P <0.01 versus in mild to moderate hypothyroidism. # P<0.05 and ##P <0.01 versus in the non-CKD group.
Discussion
This study showed three significant findings with regard to eGFR in patients with hypothyroidism. First, eGFR increased after thyroxine supplementation in CKD (chronic kidney disease) patients within 6 months. Although eGFR in CKD (chronic kidney disease) patients did not improve further after 6 months in longitudinal analysis, it is possible that various causes other than hypothyroidism, including aging, may have influenced renal dysfunction. Second, there was a correlation between eGFR levels and the severity of hypothyroidism, which was independent of age. Furthermore, eGFR in severely hypothyroid patients significantly increased up to a level similar to mildly hypothyroid patients after THRT. These findings indicate that the decrease of eGFR was caused by decreased thyroid hormone and that renal dysfunction due to hypothyroidism can be mostly improved by THRT. Finally, although the change in thyroid function after treatment was similar in CKD (chronic kidney disease) and non-CKD patients, eGFR in CKD (chronic kidney disease) patients with mild to moderate hypothyroidism was significantly increased after treatment compared to that in non-CKD patients. These findings indicate that hypothyroidism contributes to reducing eGFR in CKD (chronic kidney disease) patients more than in non-CKD (chronic kidney disease) patients.
In the previous report, in patients with hypothyroidism who underwent renal biopsies, all patients showed uniform changes that consisted of thickening of the glomerular and tubular basement membranes and the accumulation of various types of inclusions in the cell cytoplasm. Furthermore, two patients who underwent rebiopsy after the administration of thyroid hormones showed improvement in the anatomical lesions [10]; however, currently, the histological renal changes in hypothyroid patients are not well understood. The previous report and the present data showing that decreased GFR was corrected after treatment with thyroid hormone may indicate that renal dysfunction is caused mainly by functional changes rather than permanent histological damage [5]. The mechanisms of hypothyroidism-associated kidney dysfunction are thought to have several causes. First, hypothyroidism is associated with decreased cardiac output and circulating blood volume, impaired activity of the renin-angiotensin-aldosterone system, and decreased atrial natriuretic factor levels, which could lead to decreased renal perfusion [11–14]. Second, filtrate overloads caused by deficient sodium and water reabsorption in the proximal tubule could lead to adaptive preglomerular vasoconstriction [15]. Third, hypothyroidism causes a decrease of insulin-like growth factor 1 (IGF1) and vascular endothelial growth factor (VEGF). IGF1 increases forearm blood flow and creatinine clearance in humans, and VEGF increases endothelial nitric oxide synthase activity, contributing to the relaxing capacity of the renal vasculature [16].

It is known that thyroid hormone physiology is altered in CKD (chronic kidney disease) patients. These alterations may include an increased basal TSH value, reduced TSH response to TRH, diminished or absent TSH diurnal rhythm, abnormal TSH glycosylation, and impaired TSH and TRH clearance rate [17]. In addition, serum-free T3 and T4 may be reduced, free reverse T3 may be increased, and serum binding protein concentrations may be altered [17]. Uremia and chronic metabolic acidosis associated with CKD (chronic kidney disease) may contribute to these effects [18]. Furthermore, high serum iodine concentrations have been reported in CKD (chronic kidney disease) patients [7], and this iodide excess may lead to hypothyroidism through impairments in sodium iodide transport, iodine organification, and thyroid hormone synthesis and secretion via the Wolff-Chaikoff effect [8]. Indeed, there was a report that restriction of dietary iodine could correct hypothyroidism in uremic patients on hemodialysis [19].
Previous studies have demonstrated that hypothyroidism leads to decreased renal function [2–5], and, conversely, renal dysfunction leads to decreased thyroid function [6]. Our findings suggest that hypothyroidism affects eGFR in CKD (chronic kidney disease) patients more than in non-CKD patients; therefore, hypothyroidism is considered to be one of the risk factors for the progression of CKD (chronic kidney disease). It has been reported that the prevalence of hypothyroidism, including subclinical, is very high in patients with CKD (chronic kidney disease) [6]. Consequently, patients with CKD (chronic kidney disease) should be positively examined for thyroid function, and appropriate THRT should be started if needed.
Patients with CKD (chronic kidney disease), even those in the early stages of the disorder, are not only at increased risk for progression to end-stage kidney disease but also at increased risk for cardiovascular disease (CVD). Many factors, such as dyslipidemia, diabetes, and hypertension, which coexist with CKD, are associated with an increased risk of CVD [20]. In addition to the classical CVD risk factors, CKD is also associated with newly recognized risk factors for the development of atherosclerosis, including chronic inflammation and oxidative stress [20], erythropoietin resistance and anemia [21], vitamin D deficiency [22], and vascular calcification [23]. On the other hand, even a minor elevation of TSH was demonstrated to increase the risk for the development of CVD [24]. Thyroid hormone affects nearly all organ systems in the body. The number of risk factors for CVD, including hypertension, dyslipidemia, and hyperhomocysteinemia, is increased in hypothyroid patients [25]. Furthermore, Lekakis et al. [26] showed that flow-mediated, endothelium-dependent vasodilatation, a marker of endothelial function, was impaired not only in patients with mild hypothyroidism but also in subjects with subclinical hypothyroidism. Because the risk factors for CVD overlap between CKD and hypothyroidism, hypothyroidism may increase the risk for the development of CVD with worsening of CKD (chronic kidney disease). Further examination of the risk for CVD in CKD patients with hypothyroidism will be required.
There were some limitations of this study. First, this was a retrospective study with a small sample size. Second, as in many other studies, we used creatinine-based estimates of GFR. Serum creatinine levels may be influenced by the creatinine generation from myopathy and rhabdomyolysis in hypothyroidism; however, studies using inulin or 51CrEDTA clearance to estimate GFR, whose methods do not depend on creatinine levels, have reported that glomerular function is reduced during hypothyroidism, and a rise in creatinine levels in these patients is not a consequence of impaired creatinine metabolism [12, 27, 28]. Third, CKD (chronic kidney disease) was categorized using only eGFR values, and other findings of kidney damage, such as proteinuria or hematuria, were not used in this study. Fourth, because this study was conducted in Japan, which is considered an iodine-sufficient area, the results may not be applicable to another country with insufficient dietary iodine intake. Despite these limitations, it should be noted that eGFR in the CKD (chronic kidney disease) group with hypothyroidism was significantly increased after treatment compared to that in the non-CKD group in this study. To the best of our knowledge, there have been no reports on the influence of hypothyroidism on eGFR in CKD (chronic kidney disease) patients compared to non-CKD patients; however, further studies are needed to clarify whether hypothyroidism affects renal function in CKD (chronic kidney disease) patients.
In conclusion, hypothyroidism contributed to reductions in eGFR, especially in CKD (chronic kidney disease) patients. Because of the high prevalence of primary hypothyroidism in CKD (chronic kidney disease) patients, they should be positively examined for thyroid function, and appropriate THRT should be started if needed.
Conflict of interest
All the authors have declared no competing interests.

Cistanche extract is good for chronic kidney disease
From: ' Thyroid hormone replacement therapy for primary hypothyroidism leads to significant improvement of renal function in chronic kidney disease patients ' by Yuji Hataya et al
---Clin Exp Nephrol (2013) 17:525–531 DOI 10.1007/s10157-012-0727-y
References
1. Kaptein EM, Feinstein EI, Massry SG. Thyroid hormone metabolism in renal diseases. Contrib Nephrol. 1982;33:122–35.
2. Verhelst J, Berwaerts J, Maresca B, Abs R, Neels H, Mahler C, et al. Serum creatine, creatinine, and other guanidino compounds in patients with thyroid dysfunction. Metabolism. 1997;46:1063–7.
3. den Hollander JG, Wulkan RW, Mantel MJ, Berghout A. Correlation between severity of thyroid dysfunction and renal function. Clin Endocrinol (Oxf). 2005;62:423–7.
4. Asvold BO, Bjøro T, Vatten LJ. Association of thyroid function with estimated glomerular filtration rate in a population-based study: the HUNT study. Eur J Endocrinol. 2011;164:101–5.
5. Capasso G, De Tommaso G, Pica A, Anastasio P, Capasso J, Kinne R, et al. Effects of thyroid hormones on heart and kidney functions. Miner Electrolyte Metab. 1999;25:56–64.
6. Lo JC, Chertow GM, Go AS, Hsu CY. Increased prevalence of subclinical and clinical hypothyroidism in persons with chronic kidney disease. Kidney Int. 2005;67:1047–52.
7. Ramirez G, O’Neill W Jr, Jubiz W, Bloomer HA. Thyroid dysfunction in uremia: evidence for thyroid and hypophyseal abnormalities. Ann Intern Med. 1976;84:672–6.
8. Bando Y, Ushiogi Y, Okafuji K, Toya D, Tanaka N, Miura S. Non-autoimmune primary hypothyroidism in diabetic and nondiabetic chronic renal dysfunction. Exp Clin Endocrinol Diabetes. 2002;110:408–15.
9. Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
10. Salomon MI, Di Scala V, Grishman E, Brener J, Churg J. Renal lesions in hypothyroidism: a study based on kidney biopsies. Metabolism. 1967;16:846–52.
11. Crowley WF Jr, Ridgway EC, Bough EW, Francis GS, Daniels GH, Kourides IA, et al. Noninvasive evaluation of cardiac function in hypothyroidism. Response to gradual thyroxine replacement. N Engl J Med. 1977;296:1–6.
12. Villabona C, Shaun M, Roca M, Mora J, Gomez N, Gomez JM, et al. Blood volumes and renal function in overt and subclinical primary hypothyroidism. Am J Med Sci. 1999;318:277–80.
13. Asmah BJ, Wan Nazaimoon WM, Norazmi K, Tan TT, Khalid BA. Plasma renin and aldosterone in thyroid diseases. Horm Metab Res. 1997;29:580–3.
14. Zimmerman RS, Gharib H, Zimmerman D, Heublein D, Burnett JC Jr. Atrial natriuretic peptide in hypothyroidism. J Clin Endocrinol Metab. 1987;64:353–5.
15. Zimmerman RS, Ryan J, Edwards BS, Klee G, Zimmerman D, Scott N, et al. Cardiorenal endocrine dynamics during volume expansion in hypothyroid dogs. Am J Physiol. 1988;255:R61–6.
16. Schmid C, Bra¨ndle M, Zwimpfer C, Zapf J, Wiesli P. Effect of thyroxine replacement on creatinine, insulin-like growth factor 1, acid-labile subunit, and vascular endothelial growth factor. Clin Chem. 2004;50:228–31.
17. Kaptein EM. Thyroid hormone metabolism and thyroid diseases in chronic renal failure. Endocr Rev. 1996;17:45–63.
18. Wiederkehr MR, Kalogiros J, Krapf R. Correction of metabolic acidosis improves thyroid and growth hormone axes in hemodialysis patients. Nephrol Dial Transpl. 2004;19:1190–7.
19. Sanai T, Inoue T, Okamura K, Sato K, Yamamoto K, Abe T, et al. Reversible primary hypothyroidism in Japanese patients undergoing maintenance hemodialysis. Clin Nephrol. 2008;69:107–13.
20. Locatelli F, Pozzoni P, Tentori F, Del Vecchio L. Epidemiology of cardiovascular risk in patients with chronic kidney disease. Nephrol Dial Transpl. 2003;18(Suppl 7):vii2–9.
21. Kazory A, Ross EA. Anemia: the point of convergence or divergence for kidney disease and heart failure. J Am Coll Cardiol. 2009;53:639–47.
22. Levin A, Li YC. Vitamin D and its analogs: do they protect against cardiovascular disease in patients with kidney disease? Kidney Int. 2005;68:1973–81.
23. Mizobuchi M, Towler D, Slatopolsky E. Vascular calcification: the killer of patients with chronic kidney disease. J Am Soc Nephrol. 2009;20:1453–64.
24. Kahaly GJ. Cardiovascular and atherogenic aspects of subclinical hypothyroidism. Thyroid. 2000;10:665–79.
25. Vanhaelst L, Neve P, Chailly P, Bastenie PA. Coronary-artery disease in hypothyroidism. Observations in clinical myxoedema. Lancet. 1967;2:800–2.
26. Lekakis J, Papamichael C, Alevizaki M, Piperingos G, Marafelia P, Mantzos J, et al. Flow-mediated, endothelium-dependent vasodilation is impaired in subjects with hypothyroidism, borderline hypothyroidism, and high-normal serum thyrotropin (TSH) values. Thyroid. 1997;7:411–4.
27. Allon M, Harrow A, Pasque CB, Rodriguez M. Renal sodium and water handling in hypothyroid patients: the role of renal insufficiency. J Am Soc Nephrol. 1990;1:205–10.
28. Karanikas G, Schu¨tz M, Szabo M, Becherer A, Wiesner K, Dudczak R, et al. Isotopic renal function studies in severe hypothyroidism and after thyroid hormone replacement therapy. Am J Nephrol. 2004;24:41–5.






