Analysis Of Molecular Mechanism Of Cistanche Deserticola in The Treatment Of Spinal Cord Injury
Jul 10, 2023
Abstract: Spinal cord injury (SCI) is a fatal injury of the central nervous system, and there is no effective repair method at present. Cistanche.
deserticola deserticola extract can treat spinal cord injury, but the mechanism of action is still unclear. Use network pharmacology and Macromolecular docking technology to explore the molecular mechanism of Cistanche deserticola deserticola in treating SCI. The active components and targets of Cistanche deserticola deserticola are predicted by the Chinese Medicine Systems pharmacology database and analysis platform (TCMSP). The targets related to SCI are from GeneCards, Drug Bank PharmGkb, and OMIM databases, intersection targets were obtained by Venny2.1.0. The protein interaction (PPI) network was drawn through the String database and Cytoscape software. Next, KEGG pathway analysis was conducted using the Bioconductor platform. Macromolecular docking was performed using Autodock Vina. Based on screening criteria, 6 active ingredients, and 85 potential target genes were obtained from Cistanche deserticola. Through an online database, 6737 SCI-related targets and 74 intersection targets of Cistanche deserticola deserticola and SCI were obtained. In the active ingredient target network, β- Glutosterol, Quercetin, and Arachidonic acid are important active ingredients. In the PPI network, HIF1A, FOS, AR, RELA, EGFR, and CCD1 are core targets. KEGG analysis shows that the main pathway of Cistanche deserticola to treat SCI involves PI3K-Akt, TNF, and MAPK signaling pathways. In Macromolecular docking, the active compound Quercetin of Cistanche deserticola has good affinity with the core target. Finally, in vitro experimental verification shows that The treatment of Cistanche deserticola deserticola on SCI may be mainly through its active ingredient Quercetin to inhibit inflammation and oxidation, thereby improving the microenvironment of SCI It provides a new idea for further clinical application of Cistanche deserticola in the treatment of SCI.

Effects of Cistanche-Tonifying kidney
Click here to view Cistanche for Kidney disease products
【Ask for more】 Email:cindy.xue@wecistanche.com / Whats App: 0086 18599088692 / Wechat: 18599088692
Keywords: Spinal cord injury; Cistanche deserticola; Molecular mechanisms; Network pharmacology; Inflammatory response; oxidation reaction
Spinal cord injury (SCI) is a Mortal wound of the central nervous system. Due to the limited repair ability of the central nervous system, more than 27 million SCI patients worldwide are currently struggling with motor and sensory disorders [1]. There are complex pathological changes after spinal cord injury, in which the number of residual neurons and persistent inflammatory reactions often determine the final prognosis. After primary trauma, local bleeding leads to inflammatory cells Vasoactive peptides and cytokines flow into the spinal cord. Activation of proapoptotic signaling pathways, changes in cell permeability, and ischemic damage lead to the loss and demyelination of a large number of functional neurons, completely destroying the local spinal microenvironment [2]. Inflammatory cells released after vascular destruction produce inflammatory cytokines, such as TNF- α And IL-1 β, These cytokines continue to play an inflammatory role in the injured area after the subacute phase [3]. A series of reactions triggered by inflammation after injury, coupled with the destruction of the blood-spinal cord barrier, gradually aggravate spinal cord swelling and further mechanical compression, which will also lead to subsequent secondary spinal cord injury [4]. Therefore, after local injury, adjusting the microenvironment to promote the repair of the injured spinal cord is the key to SCI treatment.

Benefits of cistanche tubulosa-Tonifying kidney
Traditional Chinese medicine can be used to improve the stability of the spinal cord microenvironment. Cistanche deserticola Herba is known as "desert ginseng". Cistanche deserticola has a wide range of pharmacological effects, which can reduce free radical activity, inhibit Lipid peroxidation, anti-tumor, anti-inflammatory and other effects, and can be used for the treatment of many diseases. Research shows that Cistanche deserticola has therapeutic effects on SCI [5-6]. However, the mechanism of Cistanche deserticola in treating SCI is not very clear。
Modern pharmacology research shows that traditional Chinese medicine has complex mechanisms and multi-way interactions in the treatment of diseases. Network pharmacology can clarify the potential mechanism of traditional Chinese medicine in treating SCI by building a "drug Active ingredient target disease" network [7]. Using network pharmacology and Macromolecular docking technology, explore the potential mechanism of Cistanche deserticola Active ingredient Quercetin in inhibiting inflammation and oxidation, regulating the balance of the microenvironment, and treating SCI, Thus providing a new therapeutic drug for spinal cord injury.

Desert living cistanche-Tonifying kidney
1 Material and Methods
1.1 Database and Software
The database and software used are shown in Table 1
Table 1 Databases and software

1.2 Effective active ingredients and target prediction of Cistanche deserticola deserticola
The Active ingredient and targets of Cistanche deserticola were obtained by reference [8]
1.3 Screening of disease targets
With the keyword "neutral core injury", collect targets from DrugBank, PharmaGkb, GeneCards, and OMIM databases, remove duplicate targets, and screen drug targets and disease targets with Venny 2.1.0 to obtain potential targets of Cistanche deserticola for treating SCI.
1.4 Construct a network diagram of "Chinese medicine - Active ingredient - target - disease"
Intersecting potential targets is imported into Cytoscape software to build a network diagram of "Chinese medicine - Active ingredient - targets - diseases".
1.5 PPI network construction
The PPI network is constructed using a String database and Cytoscape software. During this process, potential intersection targets are input into the String database to obtain protein interactions. Cytoscape software can display the interactions of the PPI network and obtain the core targets based on the degree values.
1.6 KEGG pathway enrichment analysis
Utilizing the Bioconductor platform( http://bioconductor.org/biocLite.R )KEGG pathway analysis was performed on target genes. At the critical point of P<0.05, corresponding bar charts were drawn.

Cistanche powder-Tonifying kidney
1.7 Macromolecular docking
Select the core target and Active ingredient of Cistanche deserticola, and use Autodock Vina for Macromolecular docking analysis. The smaller the Binding energy (affinity), the more stable the interaction between the core protein and the active component.
1.8 Validation of anti-inflammatory and antioxidant activities at the cellular level
1.8.1 Anti-inflammatory activity verification at the cell level: (1) PC12 cells (purchased from the cell bank of Chinese Academy of Sciences in Shanghai), Quercetin (purchased from Shanghai Yuanye Biotechnology Co., Ltd.), the culture medium is a complete medium (90% DMEM medium+10% Fetal bovine serum+1% double antibody), PC12 cells in the culture bottle are cultured at 37 ℃ in the incubator with 5% CO2 saturation humidity, and the cells used in the experiment are in the logarithmic growth phase (2) Effect of Quercetin on LPS induced PC12 cell injury: cells were cultured in 96 well plates (5 × 104/mL). The experiment was divided into three groups: the control group (without any treatment) and the model group (5 μ G/mL, LPS) and Quercetin group (LPS+Quercetin, in which the concentration of Quercetin was 5, 25, and 50, respectively μ Quercetin group: pretreated with Quercetin of different concentrations for 2h; Then add the model group to 5 μ G/mL, LPS, Quercetin group added 5 μ G/mL, LPS continued to incubate for 6 hours; Discard the old culture medium and incubate it with CCK-8 reagent. Measure its absorbance (OD490 value) using an enzyme-linked immunosorbent assay. Calculate cell survival rate. Survival rate=(OD experimental group OD blank group)/(OD control group OD blank group) × 100% (3) ELISA method for detecting relevant oxidation indicators: After processing according to the above method, three parallel groups were set up for each concentration. After 24 hours of cultivation, the supernatant of each group of cells was collected, and the ELISA method (according to the instructions) was used to detect IL-6 and TNF in the supernatant- α Detect the content of IL-8.
1.8.2 Validation of antioxidant activity at the cellular level: (1) Cell density of 5 × Inoculate 104 cells/mL into 96 well plates for cultivation. The experiment was divided into three groups: the control group (without any treatment) and model group (500 cells) μ Mol/L H2O2) and the Quercetin group (H2O2+Quercetin, where the concentration of Quercetin is 5, 12.5 and 25 respectively μ Mol/L). Add 500 to the model group μ Mol/L H2O2, Quercetin group was pretreated with Quercetin of different concentrations for 2h, and 500 μ Mol/L H2O2, and the last Quercetin groups with different concentrations continued to culture for 6 h; Discard the old culture medium, add MTS reagent (purchased from Shanghai Zeye Biological) for incubation, and use an enzyme-linked immunosorbent assay to measure the absorbance value at 490 nm to determine OD. Calculate cell survival rate. Survival rate=(OD experimental group OD blank group)/(OD control group OD blank group) × 100% (2) ELISA method for detecting related oxidation indicators: After processing according to the above method, three parallel groups were set up for each concentration. After 24 hours of cell culture, the content of SOD, MDA, and ROS in the supernatant was measured. ROS level was detected using the Elabscience assay kit, the MDA level was detected using the MDA assay kit (Shanghai Fusheng Industrial Co., Ltd.), and the SOD level was detected using the Abcam assay kit according to the instructions.

Cistanche extract powder-Tonifying kidney
2 Results
2.1 Screening of effective active ingredients and targets in Cistanche deserticola deserticola
Through screening all the effective active ingredients and related targets of Cistanche deserticola deserticola in the TCMSP database, 75 Active ingredients and 85 targets were found. With OB ≥ 30% and DL ≥ 0.18 as the criteria for screening, a total of 6 Active ingredients were obtained. See Table 2 for details.
Table 2 Main active ingredients of cistanches herba in the treatment of SCI


Fig.1 Identification of potential targets of cistanches herba in the treatment of SCI

Fig.2 Traditional Chinese Medicine disease target map

Fig.3 Traditional Chinese Medicine-component disease targets regulation network

Fig.4 The core targets of SCI targets in cistanches herba therapy PPI network diagram
2.2 Draw a Venn diagram
Using the disease database, 6737 disease target genes related to spinal cord injury were screened out, and the Venn diagram (Figure 1) was drawn. The target genes corresponding to the six effective active ingredients finally obtained from Cistanche deserticola deserticola were intersection processed, and 74 common gene targets were obtained. Venn diagram was drawn using Venny2.1 software, as shown in Figure 2
2.3 Build a network of "Chinese medicine - Active ingredient - targets - diseases"
Cytoscape was used to analyze 74 common targets and build an interaction network of "Chinese medicine Active ingredient target disease" (Figure 3), including 168 edges and 81 nodes. It can be seen from the network diagram that Quercetin is connected with 68 targets, β- Glutosterol is linked to 15 targets, threazinolide is linked to 3 targets, Arachidonic acid is linked to 2 targets, and Liriodendron chinese resin phenol B dimethyl ether is linked to 1 target, indicating that all six Active ingredients of Cistanche deserticola can act on disease targets, of which Quercetin is the most closely related to disease and occupies the core position in the network diagram
2.4 Key Target Protein Interaction (PPI) Network and Obtaining Core Targets
The PPI network consists of 74 nodes and 108 edges, with an average node degree of 2.92 (Figure 4a). The degree values of the first six targets are represented by a Bar chart (Figure 4b). Among them, the degree values of FOS, EGFR, RELA, AR, CCD1, and HIF1A are 26, 22, 22, 18, 18, and 18 respectively, which are considered core targets. Cistanche deserticola may play an important role in protecting SCI through them
2.5 KEGG pathway enrichment analysis
The enrichment analysis of the KEGG pathway showed that 124 pathways were enriched through 74 potential key targets, including chemical carcinogenesis receptor activation, TNF-1 signaling pathway, PI3K-Akt signaling pathway, and MAPK signaling pathway. The abscissa is the number of genes and the ordinate is the name of the pathway, as shown in Figure 5. It can be concluded that Cistanche deserticola plays a role in treating spinal cord injury by participating in the regulation of multiple biological processes through multiple pathways.
2.6 Drug composition core target Macromolecular docking
The core component Quercetin in Cistanche deserticola was selected to perform Macromolecular docking with FOS, CCND1, HIF1A, EGFR, RELA, and AR respectively. The 3D structure of quercetin and the core target protein structure was imported into AutoDock 4.2.6 software for Macromolecular docking. The molecular affinity is shown in Table 3 and the docking structure is shown in Figure 6. The results show that the representative compound Quercetin has good docking activity with the target. AR is most stable in binding to Quercetin (-38.22 kJ/mol), The second is EGFR (-34.44 kJ/mol). It is confirmed that Quercetin can be used as an Active ingredient in the treatment of spinal cord injury
2.7 Validation of anti-inflammatory and antioxidant activities at the cellular level.
2.7.1 Effect of Quercetin on the viability of PC12 cells injured by H2O2: Compared with the control group, the survival rate of cells in the H2O2 intervention model group decreased to 50% (# # # P<0.001); Compared with the model group, the cell survival rate after adding Quercetin gradually increased with the increase of Quercetin concentration, and Quercetin 50 μ The survival rate of the mol/L group reached the highest, with a statistically significant difference (* * * P<0.001), as shown in Figure 7a.
2.7.2 Effects of Quercetin on the contents of MDA and ROS and the activity of SOD in PC12 cells injured by H2O2: Compared with the control group, the activity of SOD in the model group decreased, and the contents of MDA and ROS increased, with statistical significance (P<0.001); In the Quercetin group, different concentrations of Quercetin could improve the expression of SOD, ROS, and MDA in injured PC12 cells, with a statistically significant difference (* P<0.05, * * P<0.01, * * * P<0.001) in a concentration-dependent manner (Figure 7b-d).
2.7.3 Quercetin improved the survival rate of LPS-induced PC12 cells: compared with the control group, the survival rate of LPS-induced PC12 cells in the model group decreased to about 50% (# # # P<0.001); Compared with the model group, the Quercetin group improved the survival rate of PC12 cells induced by LPS (* P<0.05, * * P<0.01, * * * P<0.001) in a dose-dependent manner (Fig. 8a).
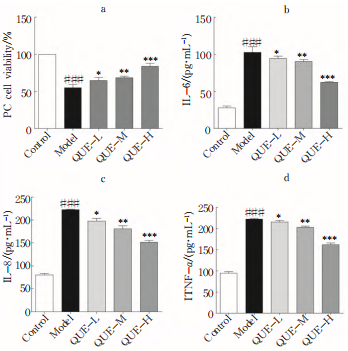
2.7.4 Quercetin reduced the inflammatory level of PC12 cells induced by LPS: compared with the control group, IL-6, IL-8, and TNF in the model group- α The content increased, and the differences were statistically significant (# # # P<0.001); Compared with the model group, Quercetin reduced the inflammatory level of PC12 cells induced by LPS, including IL-6, IL-8, and TNF- α (* P<0.05, * * P<0.01, * * * P<0.001), and in a concentration dose-dependent manner (Figure 8b-d).
Fig.5 Bubble diagram of KEGG enrichment analysis

Table 3 Affinity of molecular docking
Fig.6 Molecular models of com‐ pounds binding to the predicted target proteins

Fig.7 Validation of anti-inflammatory activity at the cellular level

Fig.8 Validation of antioxidant activity at the cellular level
3 Discussion
Spinal cord injury (SCI) can lead to loss of motor function and sensory function. Currently, Western medicine mainly focuses on drugs, cell transplantation, extracellular vesicles, tissue engineering, cell reprogramming, and rehabilitation. For example, the main role of methylprednisolone is to regulate neuroinflammation after spinal cord injury, which can achieve certain therapeutic effects. However, due to dose-related side effects, the use of high-dose MP to treat SCI is controversial [9], Exploring the pathogenesis of SCI and finding safe and effective therapeutic drugs is an urgent issue to be addressed There are complex pathological changes after spinal cord injury. The main difficulty of effective treatment of SCI is to inhibit the microenvironment. Severe oxidative stress and inflammation will interfere with the stability of the spinal cord microenvironment. Therefore, the local microenvironment is the key to the treatment of spinal cord injury. At present, potential drugs that regulate the damaged microenvironment and promote neuroprotection have attracted more and more attention. Experiments show that Cistanche deserticola can treat SCI, However, the mechanism of action is still unclear. In order to further explore the mechanism of Cistanche deserticola in treating SCI and provide more evidence for clinical treatment, the main active ingredients and targets of Cistanche deserticola and the possible signal pathways of Cistanche deserticola in treating SCI were predicted through network pharmacology, and finally verified through Macromolecular docking and in vitro experiments In the active ingredient target network diagram, Quercetin β- Glutosterol and Arachidonic acid are at the core of the network diagram, which indicates that they are the main components of Cistanche deserticola deserticola in the treatment of SCI. Among them, Quercetin is a kind of flavonoid. Pharmacological studies show that Quercetin has a variety of pharmacological activities, including anti-tumor, anti-inflammatory, antioxidant, etc. [10]. Because it is an effective free radical scavenger (antioxidant), Quercetin can treat diseases caused by oxidative stress and inflammation β- Glutosterol belongs to phytosterol, which has the effects of lowering blood fat, anti-inflammatory, antioxidant, and immune regulation [11]. Arachidonic acid is a Polyunsaturated fatty acid. Studies have found that Arachidonic acid has anti-inflammatory effects, and has a certain important role in preventing diabetes, cardiovascular disease, and cancer [12] In short, Quercetin β- Glutosterol and Arachidonic acid may be the main pharmacodynamic basis of Cistanche deserticola deserticola in treating SCI PPI network shows that EGFR, CCND1, HIF1A, FOS, RELA, and AR may be the targets of Cistanche deserticola deserticola for the treatment of SCI. Epidermal growth factor receptor (EGFR) is a transmembrane glycoprotein and one of the four members of the ErbB family of Tyrosine kinase receptors. Davinder et al found that a new EGFR inhibitor can play an anti-inflammatory role through the PI3K Akt pathway [13].
Li et al. showed that EGFR antibody functionalized collagen scaffold can promote the differentiation of neural precursor cells to repair spinal cord injury [14]. Cell Cyclin D1 is a key regulator of the cell cycle. Its biological function lies in connecting external growth factors, signal transduction and cell cycle regulation, and regulating cell proliferation, differentiation, and apoptosis [15] Wang et al. found that epigenetic silencing of CyclinD1 using bone marrow mesenchymal stem cells may accelerate the repair of spinal cord injury in rats [16]. Hypoxia-inducible factor-1 (HIF-1) plays a key role in cell adaptation to changes in oxygen supply and can serve as a transcription factor to alter gene expression and adapt to hypoxic environments through metabolic changes [17]. HIF-1 and protein von Hippel Lindau (pVHL) Proline hydroxylase (PHD) and factor inhibiting HIF (FIH) constitute a tight and orderly regulatory network. Therefore, EGFR, CCND1, and HIF1A may play an important role in the protection of Cistanche deserticola deserticola against SCI In the KEGG pathway network analysis, key targets are mainly involved in chemical carcinogenesis receptor activation, TNF, PI3K-Akt, HIF-1, p53, and MAPK signaling pathways. It suggests that the mechanism of Cistanche deserticola in treating SCI may be related to these anti-inflammatory and antioxidant pathways. Mitogen-activated protein kinases (MAPKs) are serine Threonine kinases, which mediate intracellular signal transduction related to multiple cell activities such as cell proliferation, differentiation, survival, death, and transformation [21] Luo et al. found that asiaticoside alleviates the impact of spinal cord injury through antioxidant and anti-inflammatory effects, as well as inhibiting the p38-MAPK mechanism [22]. The PI3K/Akt signaling pathway is currently recognized as a signaling pathway that regulates cell activity and proliferation ability. Wang Chunyan found that the repair effect of low-frequency pulsed electromagnetic fields (LPEMFs) on local antioxidant stress in spinal cord injury rats is closely related to the activation of the PI3K/Akt signaling pathway [23] Shultz et al. have shown that Minocycline hydrochloride regulates p38 Mitogen-activated protein kinase (MAPK) and Phosphatidylinositol 3-kinase (PI3K)/Akt signaling pathways and inhibits Matrix metalloproteinase (MMPs) targeting multiple secondary injury mechanisms in spinal cord injury through its anti-inflammatory, antioxidant and anti-apoptotic properties [21].
Tumor necrosis factor (TNF) is the main mediator of cell apoptosis, inflammation, and immunity. TNF can mediate inflammatory responses and regulate immune function. Studies have shown that the interaction between TNF and TNF receptor-1 (TNF-R1) activates multiple signal transduction pathways, including inflammation-related NF- κ B and MAPK signaling pathways [22]. So far, most participants in the TNF pathway have been verified by biochemical and genetic means, thus providing a rich source of potential drug targets for the development of new-generation anti-inflammatory drugs. There are complex pathological changes after spinal cord injury, and the secondary injury mechanism involves multiple pathways, which can also interact with each other, similar to a huge reticular structure. It is not meaningful to study a single pathway or Signaling molecule, so, Traditional Chinese medicine network pharmacology starts from the connection between pathways, studies their correlation, and provides new targets for clinical treatment.
References
1. Li L M, Zhang M, Mu J F, et al. Transplantation of human mesenchymal stem-cell-derived exosomes immobilized in an adhesive hydrogel for effective treatment of spinal cord injury[J]. Nano Lett, 2020, 20(6): 4 298-4 305.
2. Choo A M, Liu J, Lam C K, et al. Contusion, dislocation, and distraction: Primary hemorrhage and membrane permeability in distinct mechanisms of spinal cord injury[J]. Journal of Neurosurgery, 2007, 6: 255-266.
3. Pineau I, Lacroix S. Proinflammatory cytokine synthesis in the injured mouse spinal cord: Multiphasic expression pattern and identification of the cell types involved[J]. J Comp Neurol, 2007, 500: 267-285.
4. Zhang H, Xiang Z, Duan X, et al. Antitumor and anti-inflammatory effects of oligosaccharides from Cistanche deserticola extract on spinal cord injury[J]. International Journal of Biological Macromolecules, 2018, 15(124): 360-367.
5. Hao D C, Xiao P G. Network pharmacology: A rosetta stone for traditional Chinese Medicine[J]. Drug Dev Res, 2014, 75: 299-312.
6. Fehlings M G, Cho N. Methylprednisolone for the treatment of acute spinal cord injury: Counterpoint[J]. Contemporary Neurosurgery, 2015, 37(14): 1-6.
7. Li Y, Yao J Y, Han C Y, et al. Quercetin, inflammation and immunity [J]. Nutrients, 2016, 8: 167-181.
8. Rashed K. Beta-sitosterol medicinal properties: A review article[J]. International Journal of Science Inventions Today, 2020, 9(4): 208-212.
9. Tallima H, Rudi E I. Arachidonic acid: Physiological roles and potential health benefits: A review[J]. Journal of Advanced Research, 2018, 11: 33-41.
10. Davinder S, Attri K B, Kaur G R, et al. Review on EGFR inhibitors: Critical updates[J]. Mini Reviews in Medicinal Chemistry, 2016, 16(14): 1 134-1 166.
11. Li X R, Xiao Z F, Han J, et al. Promotion of neuronal differentiation of neural progenitor cells by using EGFR antibody functionalized collagen scaffolds for spinal cord injury repair[J]. Biomaterials, 2013, 34(21): 5 107-5 116.






