Recent Trends in Therapeutic Application Of Engineered Blood Purification Materials For Kidney Disease
Jul 10, 2023
Abstract
Blood purification is a commonly used method to remove excess metabolic waste in the blood in renal replacement therapy. The sufficient removal of these toxins from blood can reduce complications and improve survival lifetime in dialysis patients. However, the current biological blood purification materials in clinical practice are not ideal, where there is an unmet need for producing novel materials that have better biocompatibility, reduced toxicity, and, in particular, more efficient toxin clearance rates and a lower cost of production. Given this, this review has carefully summarized newly developed engineered different structural biomedical materials for blood purification in terms of types and structure characteristics of blood purification materials, the production process, as well as interfacial chemical adsorption properties or mechanisms. This study may provide a valuable reference for fabricating a user-friendly purification device that is more suitable for clinical blood purification applications in dialysis patients.
Keywords
Blood purification, Renal replacement therapy, Engineered biomaterials, Adsorbent, Polymeric composite membrane.

Click here to know what is the Cistanche
Introduction
Uremic toxins often accumulate in patients with compromised kidney function, including those with end-stage kidney disease (ESKD). This leads to serious illness, with renal replacement therapies as the only solution for survival. Many studies found reduced mortality and reports of a better quality of life among kidney recipients; however, demand outstrips availability, where only 25% of ESKD patients receive a kidney [1]. Worldwide, it is estimated that about 280 patients per million undergo regular hemodialysis or peritoneal dialysis treatment, while the five-year survival of these people is between 13% and 60% lower than people in the general population of similar ages [1–3]. Owing to population aging, as well as the increased prevalence of diabetes and hypertension, the incidence of ESKD is expected to rise over the next decades [4]. What’s more, the accumulation of uremic retention solutes at high concentrations is associated with adverse outcomes in dialysis patients, including high mortality and low overall health-related quality of life [5–8].

Creatinine is a major uremic toxin, and its assemblage in the blood causes a series of toxic symptoms that can reduce kidney function and consequently accelerate renal decline [9]. Protein-bound uremic toxins (PBUTs) are small molecules that primarily bind to the transport protein, human serum albumin in blood [10], and are involved in the generation of reactive oxygen species (ROS) [11]. While PBUTs are especially known to be associated with adverse/toxic effects, including cardiovascular disease [12–14], progression of kidney failure [15], and mortality [5], their renal clearance mechanisms and roles in uremic pathophysiology remain unclear [16]. Frequently, p-cresyl sulfate (PCS), indoxyl sulfate (IS), 3- Carboxy-4-methyl-5-propyl-2-furanpropionic acid (CMPF), and indole-3-acetic acid (IAA) are the most discriminating biomarkers of uremia and are considered to be prototype protein-bound uremic toxins that can bind more than 90% of plasma proteins. All four of these PBUTs have an aromatic ring and ionic functional group and can form non-covalent bonds, such as Van der Waals forces and hydrogen bonds, as well as electrostatic and hydrophobic interactions [16]. With PBUTs, there is a balance between both protein-bound and unbound forms in their secretion and circulation, and the inherent clearance of unbound toxins is largely dependent on renal tubular secretion via specific basolateral organic anion transporters (OATs). The pathway for the production and clearance of protein PBUTs is shown in Fig. 1 [16]. Uremic toxicity is linked to endothelial dysfunction and immune dysfunction, which causes inflammation and activation of innate immune effectors through the induction of a pro-inflammatory state that involves Toll-like receptors and inflammatory cytokines [14]. Thus, the sufficient removal of these toxins from the blood increases the efficacy of dialysis, which in turn increases the survival rate in ESKD patients. The basic mechanism of hemodialysis is shown in Fig. 2. However, the elimination of excess metabolites using conventional extracorporeal renal replacement therapies via semipermeable porous polymeric membranes has not currently been effective in clinical use. In particular, the PBUTs are difficult to remove through hemodialysis because each of these molecules possesses an aromatic moiety and ionic functional group that allows for binding to several adsorption sites on human serum albumin [17–20]. It has been reported that less than a 35% reduction ratio of both PCS and IS is eliminated during high-flux hemodialysis [11].

Given hemodialysis as a life-sustaining extracorporeal treatment for kidney failure, where large numbers of ESKD patients need long-term dialysis, it is necessary to prepare materials with simple and low-cost fabrication methods. Although the efficiency and selectivity properties of dialysis membranes have improved in recent decades, traditional dialysis treatment remains an inconvenient, time-consuming, and expensive process [21–23]. Recently, several biomedical blood purification materials have been developed to increase the toxin removal rate, some of which also possess characteristics like less toxicity and low production cost. In this article, we summarize these results by category to offer information for further research about dialysis materials with improved properties.

Cistanche tubulosa
Biomedical materials for toxin removal-Adsorbents
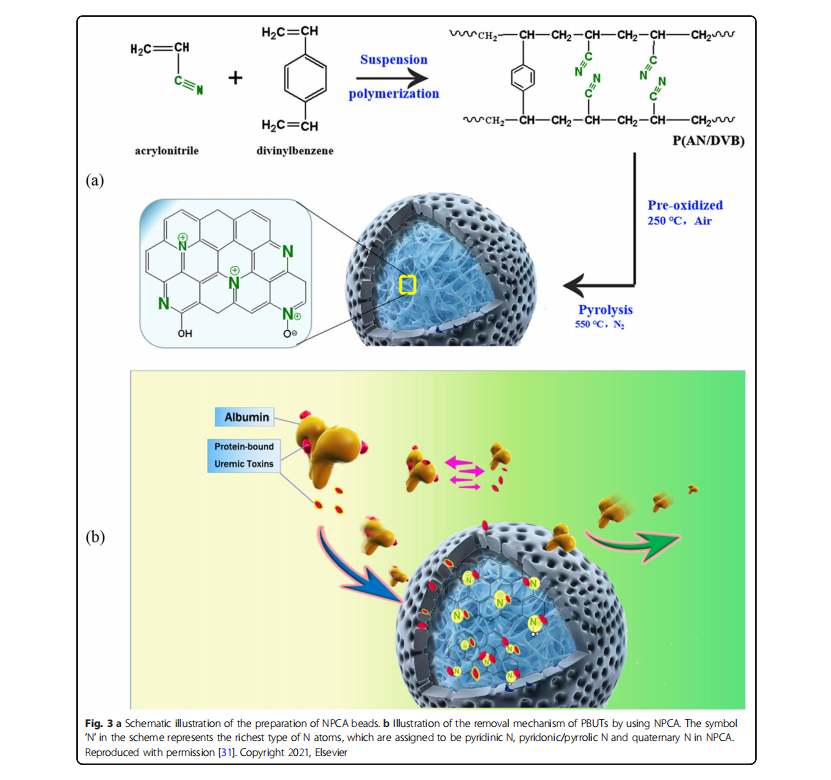
Activated carbon (AC) and zeolite are two common adsorbents applied in purification to increase ultrafiltration properties. AC has a long track record in detoxification systems as an adsorptive particle because it can adsorb a broad range of solutes [24–26]. It possesses a vast pore size but is not size-selective. AC exhibits a high adsorption capacity for uremic toxins, but it also simultaneously removes other useful molecules. Reducing the diameter of a particle is a feasible way to raise the outer surface area and thus increase accessible active sites and fast binding. However, the application of suspensions with powdered activated carbon requires a membrane filter to keep the sorbents suspended and prevent any particle contact with blood, which usually limits the amount of sorbent volume and inevitably results in lower concentrations of sorbents [27]. Compared with AC, carbon nanotubes (CNTs) possess higher surface area, larger aspect ratio, and better adsorption performance for uremic toxins, and thus they are considered the more suitable material for the design of a highly efficient blood purification membrane [28–30]. Liu et al. fabricated nitrogen-containing porous carbon adsorbent (NPCA) beads that had added advantages in terms of safe, and effective clearance of PBUTs, and possessed satisfactory in vitro hemocompatibility. NPCA was prepared via pyrolyzing a cross-linked porous acrylonitrile/divinylbenzene copolymer beads (Fig. 3a) [31]. The NPCA beads showed higher adsorption rates of PBUTs (IS, PCS, and IAA; 45%, 44%, and 95%) and an equivalent adsorption performance towards the middle-molecular-weight toxins (PTH and IL-6) in human plasma compared with HA- 130/MG-150 (a commercial adsorbent used in clinic). The PBUTs removal mechanism of NPCA is ascribed to competition between nitrogen functional groups on NPCA and proteins for PBUTs binding via the electrostatic interactions, and it has no strong relationship with the pore structure (Fig. 3b) [31].
Unlike amorphous AC, crystalline adsorbents can allow for direct structural characterization that can assist in understanding the interactions between an adsorbent and a toxin, which is critical for the design of superior adsorbent materials. Zeolite is one of the best alum inosilicates applied for numerous molecular sieves and possesses high resistance in chemical and thermal processes [32]. Zeolites are non-toxic, stable in aqueous solution, and not degraded under physiological conditions. Moreover, different types of microporous zeolite have channel systems in different sizes that can selectively adsorb some uremic toxins [33–36], and they can be found naturally or produced synthetically. These features make zeolites a potential material for artificial kidney applications. The size and shape of zeolite particles are important for creatinine uptake ability when incorporated inside the membrane [37, 38]. For instance, microparticles have better performance on creatinine adsorption than nanoparticles. Meanwhile, compared with rod nanoparticles, spherical nanoparticles are a better choice to incorporate into the electrospinning polymer fibers for improving creatinine clearance rate [37].

Cistanche supplement
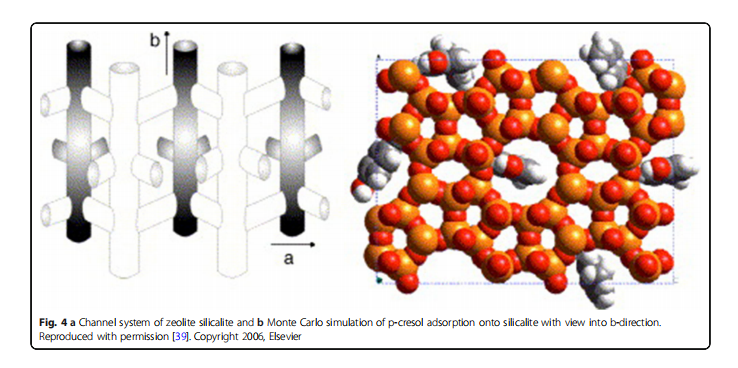
Wernet et al. investigated the elimination of uremic toxins using zeolites of different structural types [33]. They concluded that the adsorption properties of zeolites not only depend on the size of the channels but also the interactions between the adsorbates and the zeolite lattices. More specifically, zeolite silicalite (MFI) demonstrates strong adsorption of p-cresol (about 60% p-cresol in solution with concentrations close to those found in uremic patients), which is attributed to channel opening/size effect and hydrogen bonding interactions. The mechanisms of adsorption are shown in Fig. 4 (e.g., the adsorption of p-cresol onto silicalite zeolite) [39]. MFI possesses less equilibration time and a higher adsorption level of p-cresol than cellulose-based membranes and synthetic membranes [39]. Besides, it is possible to selectively eliminate 75% of creatinine in solution by an acidic mordenite (MOR), which is basically due to electrostatic interactions between the Ofunctional group and the Brønstedt sites present in the MOR pores [33]. The high adsorption of uric acid on ion-exchanged stilbites (STIs), such as Ca-STI, K-STI, and Na-STI, can be attributed to the electrostatic interaction between a cation and the negative atom of a polar molecular bond. However, there is a potential way to increase the adsorption properties of STIs through the formation of strong covalent interactions using cations with an affinity for uremic toxins [33]. Bergé-Lefranc et al. further demonstrated that pure silica MFI possesses a better capacity for the removal of p-cresol than aluminosilicate MFIs (Si/Al=30, compensating charges with H+, Na+, K+, and Mg2+) [34]. They found that compared with in solution, p-cresol affinities for the zeolites are generally reduced in human serum (obtained from dialysis patients) because proteins obstructed the pore system. Thus, zeolites can only be employed in the ultra-filtrate without direct contact with serum albumin [35].
Metal-organic framework (MOF) is one kind of novel hybrid material, which has high thermal and chemical stability and has been demonstrated to be more effective than AC or mesoporous silica materials, due to its ultrahigh porosity and active sites [40–42]. And BET surface area and internal MOF cage diameter have been previously hypothesized as key parameters in the adsorption capacity of MOFs [43]. MOF is made of metal ions and organic linkers through coordinate bounds to form a 1D, 2D, or 3D structure [40, 44]. MOFs have quickly gained traction in applications for, but are not limited to, bioactive compound separation [45], water purification [46, 47], drug delivery [48], and gas separation [49]. Nevertheless, the usage of MOFs in artificial kidney applications is still in the early stages. Furthermore, MOFs possess exceptional tenability, and, unlike other classes of crystalline materials, they can be systematically studied and incorporated into a host of functionalities [50].

Cistacnhe extract
Abdelhameed et al. reported on one type of Zr-based MOF that was grown within cotton fabric composite, which can be regenerated by sonication using methanol, while its efficiency in creatinine removal only reduces by 16% (98% vs. 82%) after three generation cycles [51]. This in-situ composite was directly formatted without fabricating UiO-66-(COOH)2 and instead used 1,2,4,5- benzene tetracarboxylic dianhydride, zirconium tetrachloride, and cotton fabric [51]. The UiO-66- (COOH)2@cotton fabric composite adsorbs creatinine through weak interactions between bonding sites of MOF and function groups of creatinine. The reaction mechanism is demonstrated in Fig. 5. Besides, it had been reported that functionalizing UiO-66 with isovalent substituents such as -NH2, -OH, and SO2H considerably improved adsorption capacity by changing the electronic properties of MOF [52, 53]. Klaudia et al. synthesized the series of UiO-66 materials varying with the final content of amino groups by changing the H2BDC/ H2BDC-NH2 ratio and adding hydrochloric acid (HCl) during modulated synthesis [52]. They confirmed that UiO-66-NH2(75%) (with 75 mol% -amino groups) and UiO-66-NH2(75%)12.5%HCl performed better adsorption capacity for hippuric acid and 3-indoleacetic acid, revealing an analogous adsorption capacity to NU-1000, meanwhile, the prepared UiO-66-NH2 did not still showed any cytotoxic effect.

Kato et al. observed the adsorption behavior of cresyl sulfate in a series of Zr6-based MOFs with varying topology, connectivity, and linker structure, including UiO-66, UiO-67, UiO-NDC, PCN-608-OH, NU-901, NU-1000, NU-1010, NU-1200 and MOF-808 [50]. These zirconium-based MOFs possess comparable surface areas and pore sizes. Among them, NU-1000 exhibits the highest toxin removal efficiency, where more than 70% of p-cresyl sulfate, 98% of indoxyl sulfate, and hippuric acid in solution, as well as about 93% of p-cresyl sulfate, can be removed from human serum albumin. This can be attributed to the highly hydrophobic adsorption sites that are sandwiched by two pyrene linkers, as well as the hydrogen bonding between the hydroxyl groups on the Zr6 nodes and the ionic functional groups of the adsorbates [50]. Two sites of p-cresyl sulfate on NU-1000 are depicted in Fig. 6. These two locations approximately have the same occupancy, where electrostatic interactions with hydroxyl groups on the Zr6 nodes and π-π interactions with pyrene-based linkers are important factors in adsorption ability. However, Cuchiaro et al. pointed out that the application of NU-1000 is limited as it is commercially unavailable, and iron is a desirable alternative to zirconium [43]. Cuchiaro et al. synthesized MIL-100(Fe) and MOF-808, both of which have the same organic linker with MOF-808 but MIL-100 (Fe) is less toxic due to its iron-based metal nodes [43]. They found p-cresyl sulfate uptake for MIL-100(Fe) was three times greater than that for MOF-808, which was less correlated to BET surface area, pore window size, cage diameter, and number of aromatic carbons in the organic linker, indicating that metal-iron interactions may be occurring more favorably in MIL-100(Fe) than MOF-808.

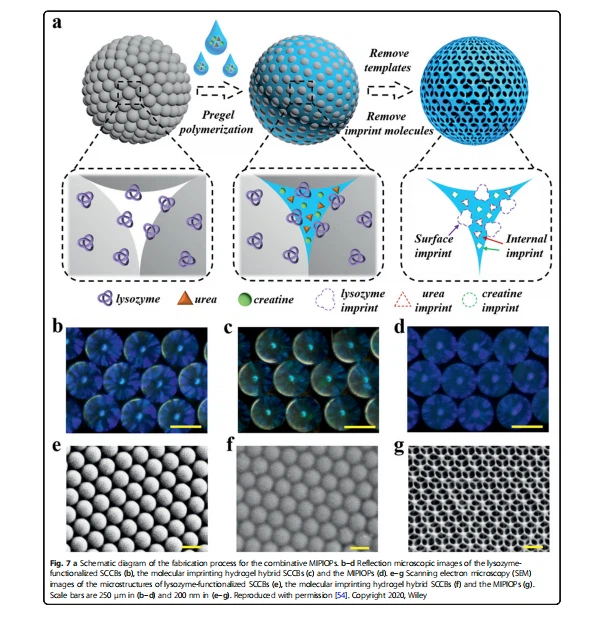
Inspired by the self-purification ability of the kidney, Chen et al. proposed a new adsorbent called molecular imprinted polymer inverse opal particles (MIPIOPs) [54]. Here, the fluidic environment is used to resolve the dilemma of insufficient contact between adsorbent materials and target molecules. The MIPIOPs are embedded in a microfluidic chip with a herringbone mixer and the scale can be readily amplified to accommodate a large number of MIPIOPs for purification. Moreover, the herringbone channels can generate chaotic advection of the fluid, and therefore improve the mixing and adsorption efficiency between the target biomolecules and MIPIOPs. The MIPIOPs are fabricated through a combinative imprinting process (Fig. 7). The silica colloidal crystal beads (SCCBs) were first fabricated, and then the silica nanoparticles at the surface of the SCCBs adsorbed the lysozyme through electrostatic interaction. The following step filled the void spaces between the silica nanoparticles of the lysozyme-functionalized SCCBs using a prequel mixture of methacrylate gelatin (GelMA), polyethylene glycol diacrylate (PEGDA), urea, and creatinine. Then, the SCCBs and the imprint molecules, including lysozyme, urea, and creatinine, were removed from the pre-gel polymer. The resultant MIPIOPs possess multiple molecular binding sites for lysozyme at the surface, while the urea and creatinine are in the interior. The MIPIOPs have unique features and display good blood compatibility, and it has been demonstrated that their adsorption capacity is maintained stably after being reused five times [54]. Moreover, due to highly ordered 3D porous structures, they are imparted with photonic band gap properties that allow for monitoring and self-reporting of the state of adsorption.
Cyclodextrins (CDs) are toroidal-shaped cyclic oligosaccharides composed of 6-8 D-glucose units (α, β, γ), with a hydrophilic exterior and a relatively hydrophobic inner cavity, which can encapsulate different low molecular weight lipophilic guests or macromolecules [55, 56]. The cup-shaped β-CD is composed of 7 glucose units and is most commonly applied due to its sensitive recognition of diverse hydrophobic molecules by forming a CD-guest complex. Li et al. proposed for the first time that adding crosslinked poly-β-cyclodextrins (PCD) into the dialysate of the exterior dialyzer for improving the mass-transfer efficiency of indoxyl sulfate (IS) (Fig. 8a) [56]. PCD was synthesized by a cross-link between β-CD and epichlorohydrin (ECH). β-CD first dissolved in NaOH solution, then ECH was added into the mixture, which was subsequently stirred at 30 °C for 2 h. According to the original study, the maximum binding capacity of PCD for IS is about 45 mg g-1 and a 21% increase in removal rate has been achieved in the simulated dialysis experiment with the introduction of PCD. The mechanism of IS binding to PCD may be that the indole ring of IS is accommodated in the cavity of β-CD through hydrophobic interaction and hydrogen bonding [56]. This new strategy exerts no negative impact on the dialysis membrane and is safe for clinical application since PCD has been proven with a low hemolysis rate and could not traverse the membrane to contact with the blood (hydrodynamic diameter of polymer is 9nm). Li et al. further compared the adsorption capacity of poly-α-cyclodextrins, poly-β- cyclodextrins and poly-γ-cyclodextrins, and poly-β- cyclodextrins performed best with the maximum cresol sulfate (PCS) binding capacity (263 mg g-1) [57]. They found that the clearance of PCS in plasma through once-through mode (Fig. 8b) is more effective than recycled mode (96% vs. 43%), which attributed to the PCS concentration difference between plasma and dialysate increase with the removal of PCS. And the PCS (96%), hippuric acid (98%), and quinolinic acid (97%) in plasma were removed in PCD added dialysate system, further elucidating the broad-spectrum PBUT removal properties of PCD [57].

MXenes are a family of two-dimensional carbides and nitrides of transition metals with the general structure Mn+1XnTx (M is an early transition metal, such as Ti, V, Nb, etc.; n+1=1-3; X is C and/or N; Tx represents the surface terminations, such as O, OH, F, and/or Cl) [58, 59]. MXenes have a unique combination of properties, including hydrophilic due to functionalized surfaces and stable colloidal solutions in water owing to high negative zeta potential, and have been extensively researched in the biomedical field in recent years [58]. Ti3C2Tx is the first reported and most studied member of MXenes and is known to efficiently adsorb urea. And Ti3C2Tx has been strongly demonstrated to have relatively high biocompatibility and low biotoxicity in previous in vivo studies [60]. In the work of Zhao et al., Ti3C2Tx (Ti3C2- F, Ti3C2-O, Ti3C2-OH, fabricated from precursor Ti3AlC2 using 10 wt % hydrofluoric acid) was used as adsorbents in aqueous solution, and it performed the rapid adsorption rate and higher adsorption capacity towards creatinine and uric acid compared with conventional activated carbon [59]. The high affinity between Ti3C2Tx and creatinine in the adsorption process is ascribed to hydrophilic surface terminations of Ti3C2Tx and intraparticle diffusion of creatinine between the Ti3C2Tx layer. In the uric acid adsorption process, however, the high affinity might come from hydrogen bonding (TiOH … N) and van der Waals interactions. What’s more, considering that the Ti3C2Tx effectively adsorb urea, creatine, and uric acid, as well as electrolyte cations (K+, Ca2+, Mg2+, etc.) can also occupy the active sites of Ti3C2Tx, Zhao et al. proposed that Ti3C2Tx has the potential to be used as an efficient sorbent for the regeneration of dialysate. More recently, Wang et al. prepared Ti3C2Tx nanosheet by delaminating the etched Ti3C2Tx in deaerated water by ultrasonication and discovered for the first time that the ultrahigh removal capability of Ti3C2Tx towards IL-6, which demonstrated 13.4 times than that of traditional activated carbon and much faster removal rate [61]. The main mechanism for adsorption is the formation of hydrogen bonding between MXene and IL-6 (TI-X … H-N-C=O), as well as the immobilization of IL-6 on the surface of MXene nanosheets.

Cistanche capsules
Conclusion
In conclusion, we summarized the novel biomaterial for blood purification that has been reported in recent years. These biomaterials were roughly divided into 3 categories, including adsorbents, polymeric composite membranes, and nanomaterials. Adsorbents, such as zeolites, activated carbon, and CTNs, are commonly incorporated inside polymer materials to enhance ultrafiltration capacity. When adsorptive particles are dispersed throughout the polymer matrix, the removal efficiency of uremic toxins can be notably improved by combining adsorption and diffusion of uremic retention solutes, which has been validated in studies related to MMM and f-CNT/ PES composite membranes. Due to the large surface area and porosity, nanomaterials and MOFs both exhibit favorable adsorption abilities. In addition, the fabrication of polymeric nanofibers is cost-effective and Zr-based MOFs have good reusability.
Permeation is a vital property of a membrane used for hemodiafiltration since a low removal rate of toxins makes the patients suffer through long periods of dialysis that are higher in expenditure. When the concentration of free toxin on the dialysate side is maintained at low levels, there is a continuous diffusion driving force over the whole hemodialyzer length, especially for PBUTs [130, 131]. As mentioned in this paper, materials with large surface areas and porosity are promising candidates for producing hemodialysis membranes. Embedding adsorptive particles in a polymer membrane matrix is also an efficacious method for achieving a more efficient clearance rate of uremic toxins, which is owing to the maintenance of a concentration difference. There are also a few adsorbents whose adsorption ability is less correlated with pore structure, such as NPCA beads. Therefore, it is crucial to analyze the respective molecular structure of adsorbent and uremic toxins, as well as the interaction within them.
Dialysis is a long-term and life-sustaining treatment for ESKD patients, and nearly 500 L of tap water is needed to obtain pure dialysis water for a single hemodialysis session, which is high in both cost and energy consumption [132]. Therefore, the simple and low-cost manufacturing method of dialysis material is of great significance for its clinical application. In addition, microbiological biofilm caused by water stagnation will still form when the tubing system is inadequate or there is improper machine maintenance [133, 134]. The bacterial growth and lysis in the water purification system, and thus pyrogens could be transferred into the blood of patients, which causes micro-inflammatory status, leading to cardiovascular side effects or acute side effects, like fever and muscular cramps [133–139]. Several sorbent systems for endotoxins (e.g., lipopolysaccharide) removal have been reported, such as functionalized nanoparticles [140], activated carbon [26, 141], the addition of a PS-poly (ethylene glycol) copolymer (PS-PEG) and bleach sterilization [133]. Additionally, the MMM offers superior endotoxin removal and acts as a safety barrier that avoids inflammatory responses without decreases in uremic toxins removal [71]. In general, biomaterials with long-term and high ultrafiltration, favorable biocompatibility, and cost-effective fabrication methods are ideal candidates for blood purification. Even if the new biomaterials mentioned in this review have improved adsorption capacity and biocompatibility, there is still much work to be done that focuses on the modification of materials so that they are suitable for clinical application.
References
1. Webster AC, Nagler EV, Morton RL, Masson P. Chronic kidney disease. Lancet. 2017;389(10075):1238–52.
2. Tonelli M, Wiebe N, Knoll G, Bello A, Browne S, Jadhav D, et al. Systematic review: kidney transplantation compared with dialysis in clinically relevant outcomes. Am J Transplant. 2011;11(10):2093–109.
3. Nordio M, Limido A, Maggiore U, Nichelatti M, Postorino M, Quintaliani G. Survival in patients treated by long-term dialysis compared with the general population. Am J Kidney Dis. 2012;59(6):819–28.
4. Liyanage T, Ninomiya T, Jha V, Neal B, Patrice HM, Okpechi I, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015;385(9981):1975–82.
5. Bammens B, Evenepoel P, Keuleers H, Verbeke K, Vanrenterghem Y. Free serum concentrations of the protein-bound retention solute p-cresol predict mortality in hemodialysis patients. Kidney Int. 2006;69(6):1081–7.
6. Cheung AK, Rocco MV, Yan G, Leypoldt JK, Levin NW, Greene T, et al. Serum beta-2 microglobulin levels predict mortality in dialysis patients: results of the home study. J Am Soc Nephrol. 2006;17(2):546–55.
7. Ito S, Osaka M, Higuchi Y, Nishijima F, Ishii H, Yoshida M. Indoxyl sulfate induces leukocyte-endothelial interactions through up-regulation of selection. J Biol Chem. 2010;285(50):38869–75.
8. Duranton F, Cohen G, De Smet R, Rodriguez M, Jankowski J, Vanholder R, et al. Normal and pathologic concentrations of uremic toxins. J Am Soc Nephrol. 2012;23(7):1258–70.
9. Panasyuk-Delaney T, Mirsky VM, Wolfbeis OS. Capacitive creatinine sensor based on a photographed molecularly imprinted polymer. Electroanalysis. 2002;14(3):221–4.
10. Vanholder R, De Smet R, Glorieux G, Argiles A, Baurmeister U, Brunet P, et al. Review on uremic toxins: classification, concentration, and interindividual variability. Kidney Int. 2003;63(5):1934–43.
11. Itoh Y, Ezawa A, Kikuchi K, Tsuruta Y, Niwa T. Protein-bound uremic toxins in hemodialysis patients measured by liquid chromatography/tandem mass spectrometry and their effects on endothelial ros production. Anal Bioanal Chem. 2012;403(7):1841–50.
12. Lin CJ, Pan CF, Liu HL, Chuang CK, Jayakumar T, Wang TJ, et al. The role of protein-bound uremic toxins on peripheral artery disease and vascular access failure in patients on hemodialysis. Atherosclerosis. 2012;225(1):173–9.
13. Wu PH, Lin YT, Chiu YW, Baldanzi G, Huang JC, Liang SS, et al. The relationship of indoxyl sulfate and p-cresyl sulfate with target cardiovascular proteins in hemodialysis patients. Sci Rep. 2021;11(1):3786.
14. Falconi CA, Junho CVDC, Fogaça-Ruiz F, Vernier ICS, Da Cunha RS, Stinghen AEM, et al. Uremic toxins: an alarming danger concerning the cardiovascular system. Front Physiol. 2021;12:686249.
15. Wu IW, Hsu KH, Lee CC, Sun CY, Hsu HJ, Tsai CJ, et al. P-cresyl sulfate and indoxyl sulfate predict progression of chronic kidney disease. Nephrol Dial Transplant. 2011;26(3):938–47.
16. Daneshamouz S, Eduok U, Abdelrasoul A, Shoker A. Protein-bound uremic toxins (PBUTS) in chronic kidney disease (CKD) patients: production pathway, challenges and recent advances in renal PBUTS clearance. NanoImpact. 2021;21:100299.
17. Ghuman J, Zunszain PA, Petitpas I, Bhattacharya AA, Otagiri M, Curry S. Structural basis of the drug-binding specificity of human serum albumin. J Mol Biol. 2005;353(1):38–52.
18. Brettschneider F, Toelle M, von der Giet M, Passlick-Deetjen J, Steppan S, Peter M, et al. Removal of protein-bound, hydrophobic uremic toxins by a combined fractionated plasma separation and adsorption technique. Artif Organs. 2013;37(4):409–16.
19. Berge-Lefranc D, Chaspoul F, Cerini C, Brunet P, Gallice P. Thermodynamic study of indoxyl sulfate interaction with human serum albumin and competitive binding with p-cresyl sulfate. J Therm Anal Calorim. 2014;115(3):2021–6.
20. Yu S, Schuchardt M, Toelle M, van der Giet M, Zidek W, Dzubiella J, et al. Interaction of human serum albumin with uremic toxins: a thermodynamic study. RSC Adv. 2017;7(45):27913–22.
21. Baboolal K, McEwan P, Sondhi S, Spiewanowski P, Wechowski J, Wilson K. The cost of renal dialysis in a UK setting--a multicentre study. Nephrol Dial Transplant. 2008;23(6):1982–9.
22. Harris A, Cooper BA, Li JJ, Bulfone L, Branley P, Collins JF, et al. Cost-effectiveness of initiating dialysis early: a randomized controlled trial. Am J Kidney Dis. 2011;57(5):707–15.
23. Young BA, Chan C, Blagg C, Lockridge R, Golper T, Finkelstein F, et al. How to overcome barriers and establish a successful home HD program. Clin J Am Soc Nephrol. 2012;7(12):2023–32.
24. Dinh DC, Recht NS, Hostetter TH, Meyer TW. Coated carbon hemoperfusion provides limited clearance of protein-bound solutes. Artif Organs. 2008; 32(9):717–24.
25. Ash SR. Sorbents in treatment of uremia: a short history and a great future. Semin Dial. 2009;22(6):615–22.
26. Gun'Ko VM, Betz WR, Patel S, Murphy MC, Mikhalovsky SV. Adsorption of lipopolysaccharide on carbon sieves. Carbon. 2006;44(7):1258–62.
27. Ash SR, Sullivan TA, Carr DJ. Sorbent suspensions vs. Sorbent columns for extracorporeal detoxification in hepatic failure. Ther Apher Dial. 2006;10(2):145–53.
28. Ye C, Gong Q, Lu F, Liang J. Adsorption of uraemic toxins on carbon nanotubes. Sep Purif Technol. 2007;58(1):2–6.
29. Lu C, Su F. Adsorption of natural organic matter by carbon nanotubes. Sep Purif Technol. 2007;58(1):113–21.
30. Spitalsky Z, Tasis D, Papagelis K, Galiotis C. Carbon nanotube-polymer composites: chemistry, processing, mechanical and electrical properties. Prog Polym Sci. 2010;35(3):357–401.
31. Liu Y, Peng X, Hu Z, Yu M, Fu J, Huang Y. Fabrication of a novel nitrogen-containing porous carbon adsorbent for protein-bound uremic toxins removal. Mater Sci Eng C Mater Biol Appl. 2021;121:111879.
32. Soldatkin OO, Soy E, Errachid A, Jaffrezic-Renault N, Akata B, Soldatkin AP, et al. Influence of composition of zeolite/enzyme nanobiocomposites on analytical characteristics of urea biosensor based on ion-selective field-effect transistors. Sens Lett. 2011;9(6SI):2320–6.
33. Wernert V, Schäf O, Ghobarkar H, Denoyel R. Adsorption properties of zeolites for artificial kidney applications. Micropor Mesopor Mat. 2005;83(1): 101–13.
34. Bergé-Lefranc D, Pizzala H, Paillaud JL, Schäf O, Vagner C, Boulet P, et al. Adsorption of small uremic toxin molecules on mfi-type zeolites from aqueous solution. Adsorption. 2008;14(2):377–87.
35. Bergé-Lefranc D, Vagner C, Calaf R, Pizzala H, Denoyel R, Brunet P, et al. In vitro elimination of protein-bound uremic toxin p-cresol by mfi-type zeolites. Micropor Mesopor Mat. 2012;153:288–93.
36. Namekawa K, Tokoro Schreiber M, Aoyagi T, Ebara M. Fabrication of zeolite– polymer composite nanofibers for removal of uremic toxins from kidney failure patients. Biomater Sci-Uk 2014;2(5):674–9.
37. Lu L, Chen C, Samarasekera C, Yeow JTW. Influence of zeolite shape and particle size on their capacity to adsorb uremic toxin as powders and as fillers in membranes. J Biomed Mater Res B Appl Biomater. 2017;105(6):1594–601.
38. Tantekin-Ersolmaz SB, Atalay-Oral C, Tatlier M, Erdem-Senatalar A, Schoeman B, Sterte J. Effect of zeolite particle size on the performance of polymer zeolite mixed matrix membranes. J Membrane Sci. 2000;175(2):285–8.
39. Wernert V, Schäf O, Faure V, Brunet P, Dou L, Berland Y, et al. Adsorption of the uremic toxin p-cresol onto hemodialysis membranes and microporous adsorbent zeolite silicalite. J Biotechnol. 2006;123(2):164–73.
40. Furukawa H, Cordova KE, O'Keeffe M, Yaghi OM. The chemistry and applications of metal-organic frameworks. Science. 2013;341(6149):974.
41. Haque E, Jun JW, Jhung SH. Adsorptive removal of methyl orange and methylene blue from aqueous solution with a metal-organic framework material, iron terephthalate (mof-235). J Hazard Mater. 2011;185(1):507–11.
42. Fletcher AJ, Thomas KM, Rosseinsky MJ. Flexibility in metal-organic framework materials: impact on sorption properties. J Solid State Chem. 2005;178(8):2491–510.
43. Cuchiaro H, Thai J, Schaffner N, Tuttle RR, Reynolds M. Exploring the parameter space of p-cresyl sulfate adsorption in metal-organic frameworks. ACS Appl Mater Interfaces. 2020;12(20):22572–80.
44. Tan J, Civalleri B. Metal-organic frameworks and hybrid materials: from fundamentals to applications. Crystengcomm. 2015;17(2):197–8.
45. Abdelhameed RM, Abdel-Gawad H, Taha M, Hegazi B. Separation of bioactive chamazulene from chamomile extract using the metal-organic framework. J Pharmaceut Biomed. 2017;146:126–34.
46. Mon M, Bruno R, Ferrando-Soria J, Armentano D, Pardo E. Metal-organic framework technologies for water remediation: towards a sustainable ecosystem. J Mater Chem A. 2018;6(12):4912–47.
47. Abdelhameed RM, Abdel-Gawad H, Elshahat M, Emam HE. Cu–btc@cotton composite: design and removal of ethion insecticide from water. RSC Adv. 2016;6(48):42324–33.
48. Huxford RC, Della Rocca J, Lin W. Metal-organic frameworks as potential drug carriers. Curr Opin Chem Biol. 2010;14(2):262–8.
49. Rodenas T, Luz I, Prieto G, Seoane B, Miro H, Corma A, et al. Metal–organic framework nanosheets in polymer composite materials for gas separation. Nat Mater. 2015;14(1):48–55.
50. Kato S, Otake K, Chen H, Akpinar I, Buru CT, Islamoglu T, et al. Zirconium-based metal–organic frameworks for the removal of protein-bound uremic toxin from human serum albumin. J Am Chem Soc. 2019;141(6):2568–76.
51. Abdelhameed RM, Rehan M, Emam HE. The figuration of zr-based mof@cotton fabric composite for potential kidney application. Carbohyd Polym. 2018; 195:460–7.
52. Dymek K, Kurowski G, Kuterasinski L, Jedrzejczyk R, Szumera M, Sitarz M, et al. In search of effective auto-66 metal-organic frameworks for artificial kidney application. ACS Appl Mater Inter. 2021;13(38):45149–60.
53. Zhu J, Wu L, Bu Z, Jie S, Li B. Polyethyleneimine-modified UiO-66-nh2 (zr) metal-organic frameworks: preparation and enhanced co2 selective adsorption. ACS Omega. 2019;4(2):3188–97.
54. Chen H, Bian F, Sun L, Zhang D, Shang L, Zhao Y. Hierarchically molecular imprinted porous particles for biomimetic kidney cleaning. Adv Mater. 2020; 32(52):2005394.
55. Mejia-Ariza R, Grana-Suarez L, Verboom W, Huskens J. Cyclodextrin-based supramolecular nanoparticles for biomedical applications. J Mater Chem B. 2017;5(1):36–52.
56. Li J, Han L, Liu S, He S, Cao Y, Xie J, et al. Removal of indoxyl sulfate by water-soluble poly-cyclodextrins in dialysis. Colloid Surface B. 2018;164:406–13.
57. Li J, Han L, Xie J, Liu S, Jia L. Multi-sites poly cyclodextrin adsorbents for removal of protein-bound uremic toxins combining with hemodialysis. Carbohyd Polym. 2020;247:116665.
58. Gogotsi Y, Anasori B. The rise of mxenes. ACS Nano. 2019;13(8):8491–4.
59. Zhao Q, Seredych M, Precetti E, Shuck CE, Harhay M, Pang R, et al. Adsorption of uremic toxins using ti3c2tx men for dialysate regeneration. ACS Nano. 2020;14(9):11787–98.
60. Dai C, Lin H, Xu G, Liu Z, Wu R, Chen Y. Biocompatible 2d titanium carbide (mxenes) composite nanosheets for ph-responsive mri-guided tumor hyperthermia. Chem Mater. 2017;29(20):8637–52.
61. Wang T, Sun X, Guo X, Zhang J, Yang J, Tao S, et al. Ultraefficiently calming cytokine storm using ti (3) c (2) t (x) mxene. Small Methods. 2021;5(5): e2001108.
62. Tian H, Tang Z, Zhuang X, Chen X, Jing X. Biodegradable synthetic polymers: preparation, functionalization, and biomedical application. Prog Polym Sci. 2012;37(2):237–80.
63. Weber V, Linsberger I, Hauner M, Leistner A, Leistner A, Falkenhagen D. Neutral styrene-divinylbenzene copolymers for adsorption of toxins in liver failure. Biomacromolecules. 2008;9(4):1322–8.
64. Jiang X, Xiang T, Xie Y, Wang R, Zhao W, Sun S et al. Functional polyethersulfone particles for the removal of bilirubin. Journal of Materials Science: Materials in Medicine 2015;27(2):28.
65. Lu L, Samarasekera C, Yeow JTW. The creatinine adsorption capacity of electrospun polyacrylonitrile (PAN)-zeolite nanofiber membranes for potential artificial kidney applications. J Appl Polym Sci 2015;132(34): 42418- 26.
66. Lu L, Yeow JTW. An adsorption study of indoxyl sulfate by zeolites and polyethersulfone-zeolite composite membranes. Mater Design. 2017;120: 328–35.
67. Tijink MS, Wester M, Sun J, Saris A, Bolhuis-Versteeg LA, Saiful S, et al. A novel approach for blood purification: mixed-matrix membranes combining diffusion and adsorption in one step. Acta Biomater. 2012;8(6):2279–87.
68. Tijink MSL, Wester M, Glorieux G, Gerritsen KGF, Sun J, Swart PC, et al. Mixed matrix hollow fiber membranes for removal of protein-bound toxins from human plasma. Biomaterials. 2013;34(32):7819–28.
69. Pavlenko D, van Geffen E, van Steenbergen MJ, Glorieux G, Vanholder R, Gerritsen KGF et al. New low-flux mixed matrix membranes offer superior removal of protein-bound toxins from human plasma. Sci Rep 2016;6:34429.
70. Geremia I, Pavlenko D, Maksymow K, Rueth M, Lemke HD, Stamatialis D. Ex vivo evaluation of the blood compatibility of mixed matrix hemodialysis membranes. Acta Biomater. 2020;111:118–28.
71. Geremia I, Bansal R, Stamatialis D. In vitro assessment of mixed matrix hemodialysis membrane for achieving endotoxin-free dialysate combined with high removal of uremic toxins from human plasma. Acta Biomater. 2019;90:100–11.
72. Nie C, Ma L, Xia Y, He C, Deng J, Wang L, et al. Novel heparin-mimicking polymer brush grafted carbon nanotube/pes composite membranes for safe and efficient blood purification. J Membrane Sci. 2015;475:455–68.
73. Zhang Y, Lim CT, Ramakrishna S, Huang Z. Recent development of polymer nanofibers for biomedical and biotechnological applications. J Mater Sci Mater Med. 2005;16(10):933–46.
74. Ramakrishna S, Fujihara K, Teo W, Yong T, Ma Z, Ramaseshan R. Electrospun nanofibers: solving global issues. Mater Today. 2006;9(3):40–50.
75. Huang Z, Zhang YZ, Kotaki M, Ramakrishna S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos Sci Technol. 2003;63(15):2223–53.
76. Fu GD, Xu LQ, Yao F, Zhang K, Wang XF, Zhu MF, et al. Smart nanofibers from combined living radical polymerization, “click chemistry”, and electrospinning. ACS Appl Mater Inter. 2009;1(2):239–43.
77. Kim Y, Ebara M, Aoyagi T. A smart nanofiber web that captures and releases cells. Angew Chem Int Ed. 2012;51(42):10537–41.
78. Wang XY, Drew C, Lee SH, Senecal KJ, Kumar J, Samuelson LA. Electrospinning technology: a novel approach to sensor application. J Macromol Sci Pure Appl Chem. 2002; A39(10):1251–8.
79. Barhoum A, Pal K, Rahier H, Uludag H, Kim IS, Bechelany M. Nanofibers as new-generation materials: from spinning and nano-spinning fabrication techniques to emerging applications. Appl Mater Today. 2019;17:1–35.
80. Peh P, Lim NSJ, Blocki A, Chee SML, Park HC, Liao S, et al. Simultaneous delivery of highly diverse bioactive compounds from blend electrospun fibers for skin wound healing. Bioconjugate Chem. 2015;26(7):1348–58.
81. Qi R, Guo R, Shen M, Cao X, Zhang L, Xu J, et al. Electrospun poly (lactic-co-glycolic acid)/halloysite nanotube composite nanofibers for drug encapsulation and sustained release. J Mater Chem. 2010;20(47):10622–9.
82. Salalha W, Kuhn J, Dror Y, Zussman E. Encapsulation of bacteria and viruses in electrospun nanofibres. Nanotechnology. 2006;17(18):4675–81.
83. Luo Y, Nartker S, Miller H, Hochhalter D, Wiederoder M, Wiederoder S, et al. Surface functionalization of electrospun nanofibers for detecting e. Coli o157:h7 and body cells in a direct-charge transfer biosensor. Biosens Bioelectron. 2010;26(4):1612–7.
84. Esmaeili A, Haseli M. Electrospinning of thermoplastic carboxymethyl cellulose/poly (ethylene oxide) nanofibers for use in drug-release systems. Mat Sci Eng C-Mater. 2017;77:1117–27.
85. Han D, Steckl AJ. Triaxial electrospun nanofiber membranes for controlled dual release of functional molecules. ACS Appl Mater Inter. 2013;5(16):8241–5.
86. Bahramimehr F, Esmaeili A. Producing hybrid nanofiber-based on /pan/ fe3o4/zeolite/nettle plant extract/urease and a deformed coaxial natural polymer to reduce toxicity materials in the blood of dialysis patients. J Biomed Mater Res A. 2019;107(8):1736–43.
87. Irfan M, Idris A, Yusof NM, Khairuddin NFM, Akhmal H. Surface modification and performance enhancement of nano-hybrid f-mwcnt/pvp90/pes hemodialysis membranes. J Membrane Sci. 2014;467:73–84.
88. Niyogi S, Hamon MA, Hu H, Zhao B, Bhowmik P, Sen R, et al. Chemistry of single-walled carbon nanotubes. Accounts Chem Res. 2002;35(12):1105–13.
89. Raravikar NR, Schadler LS, Vijayaraghavan A, Zhao Y, Wei B, Ajayan PM. Synthesis and characterization of thickness-aligned carbon nanotube-polymer composite films. Chem Mater. 2005;17(5):974–83.
90. Valcárcel M, Cárdenas S, Simonet BM, Moliner-Martínez Y, Lucena R. Carbon nanostructures as sorbent materials in analytical processes. TrAC Trends Analyt Chem. 2008;27(1):34–43.
91. Tsuge M, Takahashi K, Kurimoto R, Fulati A, Uto K, Kikuchi A, et al. Fabrication of water-absorbing nanofiber meshes toward an efficient removal of excess water from kidney failure patients. Fibers. 2019;7(5):39.
92. Khan I, Saeed K, Khan I. Nanoparticles: properties, applications, and toxicities. Arab J Chem. 2019;12(7):908–31.
93. Wang L, Zhang Y, Li Y, Chen J, Lin W. Recent advances in engineered nanomaterials for acute kidney injury theranostics. Nano Res. 2021;14(4):920–33.
94. Stamopoulos D, Bouziotis P, Benaki D, Kotsovassilis C, Zirogiannis PN. Utilization of nanobiotechnology in hemodialysis: mock-dialysis experiments on homocysteine. Nephrol Dial Transplant. 2008;23(10):3234–9.
95. Ma Y, Cai F, Li Y, Chen J, Han F, Lin W. A review of the application of nanoparticles in the diagnosis and treatment of chronic kidney disease. Bioact Mater. 2020;5(3):732–43.
96. Cheah W, Ishikawa K, Othman R, Yeoh F. Nanoporous biomaterials for uremic toxin adsorption in artificial kidney systems: a review. J Biomed Mater Res B Appl Biomater. 2017;105(5):1232–40.
97. Abidin MNZ, Goh PS, Ismail AF, Said N, Othman MHD, Hasbullah H, et al. Highly adsorptive oxidized starch nanoparticles for efficient urea removal. Carbohyd Polym. 2018;201:257–63.
98. Cabello-Alvarado C, Andrade-Guel M, Pérez-Alvarez M, Cadenas-Pliego G, Cortés-Hernández DA, Bartolo-Pérez P, et al. Graphene nanoplatelets modified with amino groups by ultrasonic radiation of variable frequency for potential adsorption of uremic toxins. NanomaterialsBasel. 2019;9(9):1261.
99. Andrade-Guel M, Ávila-Orta CA, Cadenas-Pliego G, Cabello-Alvarado CJ, Pérez-Alvarez M, Reyes-Rodríguez P, et al. Synthesis of nylon 6/modified carbon black nanocomposites for application in uric acid adsorption. Materials. 2020;13(22):5173.
100. Korsvik C, Patil S, Seal S, Self WT. Superoxide dismutase mimetic properties exhibited by vacancy-engineered ceria nanoparticles. Chem Commun. 2007; 10:1056–8.
101. Pirmohamed T, Dowding JM, Singh S, Wasserman B, Heckert E, Karakoti AS, et al. Nanoceria exhibit redox state-dependent catalase mimetic activity. Chem Commun. 2010;46(16):2736–8.
102. Xue Y, Luan Q, Yang D, Yao X, Zhou K. Direct evidence for hydroxyl radical scavenging activity of cerium oxide nanoparticles. J Phys Chem C. 2011; 115(11):4433–8.
103. Ni D, Wei H, Chen W, Bao Q, Rosenkrans ZT, Barnhart TE, et al. Ceria nanoparticles meet hepatic ischemia-reperfusion injury: the perfect imperfection. Adv Mater. 2019;31(40):1902956.
104. Fleming RE, Ponka P. Iron overload in human disease. New Engl J Med. 2012;366(4):348–59.
105. Hamilton JL, Kizhakkedathu JN. Polymeric nanocarriers for the treatment of systemic iron overload. Mol Cell Ther. 2015;3:3.
106. Kang H, Han M, Xue J, Baek Y, Chang J, Hu S et al. Renal clearable nano chelators for iron overload therapy. Nat Commun 2019;10(1):5134.
107. Mobarra N, Shanaki M, Ehteram H, Nasiri H, Sahmani M, Saeidi M, et al. A review on iron chelators in treatment of iron overload syndromes. Int J Hematol Oncol Stem Cell Res. 2016;10(4):239–47.
108. Shapiro SM. Bilirubin toxicity in the developing nervous system. Pediatr Neurol. 2003;29(5):410–21.
109. Peng Z, Yang Y, Luo J, Nie C, Ma L, Cheng C, et al. Nanofibrous polymeric beads from aramid fibers for efficient bilirubin removal. Biomater Sci-Uk. 2016;4(9):1392–401.
110. Yang M, Cao K, Sui L, Qi Y, Zhu J, Waas A, et al. Dispersions of aramid nanofibers: a new nanoscale building block. ACS Nano. 2011;5(9):6945–54.
111. Zhao C, Xue J, Ran F, Sun S. Modification of polyethersulfone membranes - a review of methods. Prog Mater Sci. 2013;58(1):76–150.
112. Goldberg AL. Protein degradation and protection against misfolded or damaged proteins. Nature. 2003;426(6968):895–9.
113. Furie B, Furie BC. Mechanisms of disease: mechanisms of thrombus formation. New Engl J Med. 2008;359(9):938–49.
114. Arepally GM. Heparin-induced thrombocytopenia. Blood. 2017;129(21):2864– 72.
115. Ratner BD. Blood compatibility - a perspective. J Biomat Sci-Polym E. 2000; 11(11):1107–19.
116. Mao C, Qiu YZ, Sang HB, Mei H, Zhu AP, Shen J, et al. Various approaches to modify biomaterial surfaces for improving hemocompatibility. Adv Colloid Interface. 2004;110(1-2):5–17.
117. Werner C, Maitz MF, Sperling C. Current strategies towards hemocompatible coatings. J Mater Chem. 2007;17(32):3376–84.
118. Huang J, Xue J, Xiang K, Zhang X, Cheng C, Sun S, et al. Surface modification of polyethersulfone membranes by blending triblock copolymers of methoxyl poly (ethylene glycol)-polyurethane-methoxyl poly (ethylene glycol). Colloid Surface B. 2011;88(1):315–24.
119. Liu X, Xu Y, Wu Z, Chen H. Poly(n-vinylpyrrolidone)-modified surfaces for biomedical applications. Macromol Biosci. 2013;13(2):147–54.
120. Li X, Wang M, Wang L, Shi X, Xu Y, Song B, et al. Block copolymer-modified surfaces for conjugation of biomacromolecules with control of quantity and activity. Langmuir. 2013;29(4):1122–8.
121. Modi A, Verma SK, Bellare J. Hydrophilic zif-8 decorated go nanosheets improve biocompatibility and separation performance of polyethersulfone hollow fiber membranes: a potential membrane material for bioartificial liver application. Mater Sci Eng C. 2018;91:524–40.
122. Said N, Abidin MNZ, Hasbullah H, Ismail AF, Goh PS, Othman MHD, et al. Iron oxide nanoparticles improved biocompatibility and removal of middle molecule uremic toxin of polysulfone hollow fiber membranes. J Appl Polym Sci. 2019;136(48):48234.
123. Wang L, Gong T, Brown Z, Randle C, Guan Y, Ye W, et al. Ascidian-inspired heparin-mimetic magnetic nanoparticles with potential for application in hemodialysis as recycling anticoagulants. ACS Biomater Sci Eng. 2020;6(4): 1998–2006.
124. Cheng C, Sun S, Zhao C. Progress in heparin and heparin-like/mimicking polymer-functionalized biomedical membranes. J Mater Chem B. 2014;2(44): 7649–72.
125. Nie S, Xue J, Lu Y, Liu Y, Wang D, Sun S, et al. Improved blood compatibility of polyethersulfone membrane with a hydrophilic and anionic surface. Colloid Surface B. 2012;100:116–25.
126. Li L, Cheng C, Xiang T, Tang M, Zhao W, Sun S, et al. Modification of polyethersulfone hemodialysis membrane by blending citric acid grafted polyurethane and its anticoagulant activity. J Membrane Sci. 2012;405:261–74.
127. Wang LR, Qin H, Nie SQ, Sun SD, Ran F, Zhao CS. Direct synthesis of heparin-like poly (ether sulfone) polymer and its blood compatibility. Acta Biomater. 2013;9(11):8851–63.
128. Nie S, Tang M, Cheng CS, Yin Z, Wang L, Sun S, et al. Biologically inspired membrane design with a heparin-like interface: prolonged blood coagulation, inhibited complement activation, and bio-artificial liver-related cell proliferation. Biomater Sci-Uk. 2014;2(1):98–109.
129. Ma L, Qin H, Cheng C, Xia Y, He C, Nie C, et al. Mussel-inspired self-coating at macro-interface with improved biocompatibility and bioactivity via dopamine-grafted heparin-like polymers and heparin. J Mater Chem B. 2014;2(4):363–75.
130. Meyer TW, Peattie JWT, Miller JD, Dinh DC, Recht NS, Walther JL, et al. Increasing the clearance of protein-bound solutes by the addition of a sorbent to the dialysate. J Am Soc Nephrol. 2007;18(3):868–74.
131. Patzer J. Principles of bound solute dialysis. Ther Apher Dial. 2006;10(2):118– 24.
132. Agar JWM. Green dialysis: the environmental challenges ahead. Semin Dialysis. 2015;28(2):186–92.
133. Madsen B, Britt DW, Ho C, Henrie M, Ford C, Stroup E, et al. Hemodialysis membrane surface chemistry as a barrier to lipopolysaccharide transfer. J Appl Polym Sci. 2015;132(4155021).
134. Lonnemann GR. The quality of dialysate: an integrated approach. Kidney Int. 2000;5876:S112–9.
135. Susantitaphong P, Riella C, Jaber BL. Effect of ultrapure dialysate on markers of inflammation, oxidative stress, nutrition, and anemia parameters: a meta-analysis. Nephrol Dial Transpl. 2013;28(2):438–46.
136. Gorbet MB, Sefton MV. Endotoxin: the uninvited guest. Biomaterials. 2005; 26(34):6811–7.
137. Schepers E, Glorieux G, Eloot S, Hulko M, Boschetti-de-Fierro A, Beck W, et al. Assessment of the association between increasing membrane pore size and endotoxin permeability using a novel experimental dialysis simulation set-up. BMC Nephrol. 2018;19(1):1–0.
138. Glorieux G, Hulko M, Speidel R, Brodbeck K, Krause B, Vanholder R. Looking beyond endotoxin: A comparative study of pyrogen retention by ultrafilters used for the preparation of sterile dialysis fluid. Sci Rep 2014;4:6390.
139. van Tellingen A, Grooteman M, Schoorl M, Bartels P, Schoorl M, van der Ploeg T, et al. Intercurrent clinical events are predictive of plasma c-reactive protein levels in hemodialysis patients. Kidney Int. 2002;62(2):632–8.
140. Darkow R, Groth T, Albrecht W, Lutzow K, Paul D. Functionalized nanoparticles for endotoxin binding in aqueous solutions. Biomaterials. 1999;20(14):1277–83.
141. Murphy MC, Patel S, Phillips GJ, Davies JG, Lloyd AW, Gun'Ko VM, et al. Adsorption of inflammatory cytokines and endotoxin by mesoporous polymers and activated carbons. In: RodriguezReinoso F, McEnaney B, Rouquerol J, Unger K, editors. Studies in surface science and catalysis; 2002. p. 515–20.
Cui Gao1 , Qian Zhang1 , Yi Yang1,2,3, Yangyang Li4,5 and Weiqiang Lin1,3
1 Kidney Disease Center, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310003, Zhejiang, China.
2 Department of Nephrology, The Fourth Affiliated Hospital, Zhejiang University School of Medicine, Yiwu 322000, Zhejiang, China.
3 International Institutes of Medicine, The Fourth Affiliated Hospital, Zhejiang University School of Medicine, Yiwu 322000, Zhejiang, China.
4 Key Laboratory of Women’s Reproductive Health Research of Zhejiang Province, Women’s Hospital, Zhejiang University School of Medicine, Hangzhou 310006, Zhejiang, China.
5 Cancer Center, Zhejiang University, Hangzhou 310058, Zhejiang, China.






