Novel Target To Improve AKI-CKD Transition
Jul 12, 2023
Acute kidney injury (AKI) is a common complication in many hospitalized patients and is associated with high morbidity and mortality. Survivors of acute kidney injury often develop chronic kidney disease (CKD) due to the irreversible loss of tubular cells during the acute phase of AKI and their persistent loss thereafter. Increased adaptive metabolism of remaining tubular cells affects stress response pathways, promotes tubulointerstitial inflammation and fibrosis, and ultimately promotes tubular cell death, ie, CKD progression. Tubulointerstitial hypoxia is a hallmark of the AKI-CKD transition in the context of increased metabolism of remaining tubular cells, as these cells decrease in number, injury, and renal remodeling lead to microvascular dysfunction and reduction.

Click to cistanche herba for kidney disease
High mobility group box 1 (HMGB1) is a nuclear protein. When acetylated, HMGB1 can be translocated into the cytoplasm to regulate cellular activities. During necrosis or the release of neutrophil extracellular traps, HMGB1 can reach the extracellular space and induce pro-inflammatory effects via CXCR4 as a chemokine or as a damage-associated molecular pattern (DAMP). Multiple studies have documented the role of HMGB1 as a DAMP in models of acute tissue injury, including the kidney. However, the distinct intracellular and extracellular roles of HMGB1 remain unclear. To explore how HMGB1 participates in AKI in renal tubular epithelial cells, in March 2023, the team of Hans-Joachim Anders from the University of Munich in Germany published a paper entitled "Tubular Epithelial Cell HMGB1 Promotes AKI-CKD Transition by Sensitizing Cycling Tubular Cells to Oxidative Stress: A Rationale for Targeting HMGB1 during AKI Recovery" article, using renal tubular epithelial cells to specifically knock out Hmgb1 mice and a variety of HMGB1 antagonists, found that intracellular HMGB1 reduces the recovery of tubular cells in response to prolonged ischemic stimulation ability, indicating that intracellular HMGB1 is a potential target to promote renal remodeling and improve long-term prognosis in AKI.
First, to determine the expression of HMGB1 in normal kidney, the authors analyzed the single-nucleus RNA sequencing data set and found that in the normal kidney of mice and humans, HMGB1 was widely expressed in tubular epithelial cells, mesenchymal cells, and podocytes, It is also expressed in macrophages and to a lesser extent in endothelial cells. In ischemia-reperfusion injury (IRI) mice, HMGB1 was downregulated early (4 h and 12 h) and returned to near basal levels 48 h after injury. Analysis of single-cell RNA-sequencing data of cells in the urine of critically ill patients with AKI revealed ubiquitous expression of HMGB1. Thus, HMGB1 is expressed in multiple cell types before and during the onset of AKI (Fig. 1).
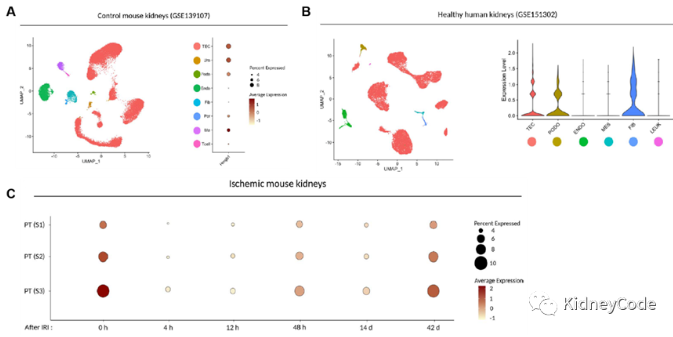
Figure 1 Expression of HMGB1 in mouse and human kidney
Previous studies using HMGB1-neutralizing antibodies or HMGB1 inhibitors have shown that extracellular HMGB1 has a pro-inflammatory role in the acute and subacute phases after AKI. Neutralizing antibodies target only extracellular HMGB1, while small molecule antagonists such as glycyrrhizic acid (Gly) and ethyl pyruvate (EP) also target intracellular HMGB1. After AKI, plasma HMGB1 levels peaked early and then declined. However, plasma HMGB1 levels remained higher than normal at day 30, consistent with continued cell death in the kidney. Gly reduced glomerular filtration rate (GFR) and restored renal function on day 4, but did not significantly reduce blood HMGB1 levels. At day 30 after reperfusion, Gly treatment resulted in better proximal tubule repair and significantly reduced renal injury scores. EP pretreatment also restored GFR on day 4 and persisted until day 30, while significantly reducing blood HMGB1 levels. In addition, EP also resulted in better repair of proximal and distal tubules and reduced renal injury scores. However, neither treatment affected initial GFR levels. Taken together, the renoprotective effects of Gly and EP follow the early injury stage (Fig. 2).
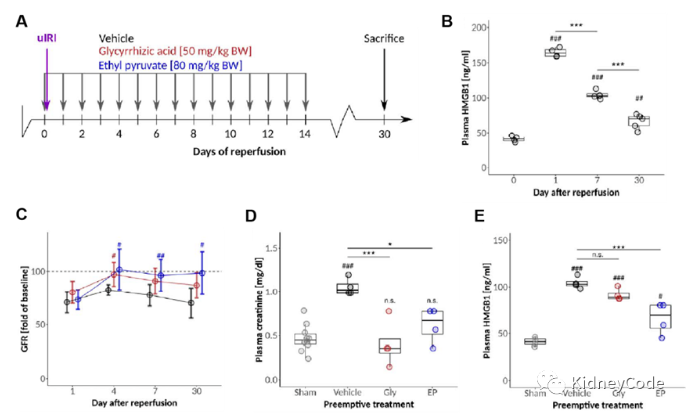
Figure 2 HMGB1 inhibitors Gly and EP can inhibit AKI-CKD transition
To explore the role of HMGB1 in tubular epithelial cells, the authors constructed Pax8-Cre; Hmgb1fl/fl mice, which can induce tubular cell-specific knockout of Hmbg1 after the completion of mouse kidney development and growth. Compared with control Hmgb1fl/fl mice, the authors did not find obvious abnormalities in renal function and histology after Hmbg1-specific knockout. Further exploring the effect of Hmgb1-specific knockout on AKI-CKD transition, the authors found that Pax8-Cre; Hmgb1fl/fl mice had no significant difference in the early injury level 7 days after AKI compared with Hmgb1fl/fl mice, but at GFR began to recover after 7 days. On day 30 after reperfusion, Pax8-Cre; Hmgb1fl/fl mice had no significant difference in the weight of both sides of the kidney, the proximal tubule recovered better, the interstitial matrix deposition was significantly reduced, and the expression of Ngal was reduced. These results suggest that tubular HMGB1 promotes AKI-CKD transition under persistent renal hypoxia (Fig. 3).

Figure 3 Knockout of HMGB1 in renal tubular epithelial cells inhibits AKI-CKD transition
To explore the effect of intracellular HMGB1 on the survival of primary tubular cells under ischemic stress, the authors simulated acute injury by isolating tubular cells, exposed the cells to H2O2 for 24 h to simulate the continuous oxidative stress after injury, and found that in the early stage of injury Deletion of HMGB1 had no significant effect on the resilience of cells to stress, but Hmgb1 knockout cells showed stronger resilience to prolonged oxidative stress. In addition, the authors also found that the loss or inhibition of the intracellular function of HMGB1 has a renal protective effect through Hmgb1 knockout or Gly/EP combined with HMGB1 neutralizing antibody 2G7. These results suggest that HMGB1 renders tubular epithelial cells more vulnerable to oxidative stress through mechanisms other than the DAMP function of extracellular HMGB1 during the early necroinflammatory phase of AKI. HMGB1 promotes the AKI-CKD transition that occurs in the late/subacute phase of AKI , when a small fraction of tubular epithelial cells proliferate and repair damaged tubular cells. In addition to pretreatment, postoperative administration of HGMB1 inhibitors also improved AKI-CKD transition (Fig. 4).
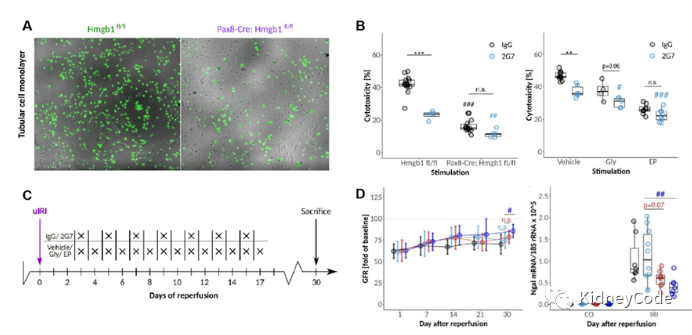
Figure 4 HMGB1 affects the sensitivity of tubular cells to prolonged oxidative stress
In this work, the authors explored the role of HMGB1 in AKI-CKD using the Hmgb1 tubular-specific knockout mouse model and mouse primary clls, and found that HMGB1 increases the sensitivity of tubular epithelial cells to continuous oxidative stress, to promote the transformation of AKI-CKD, suggesting that HMGB1 may be used as a target for the treatment of AKI-CKD transformation, and verified the improvement effect of HMGB1 inhibitors on AKI-CKD transformation. However, the specific mechanism by which HMGB1 plays this role is still unclear, and further research is needed to better target therapy.
How does Cistanche treat kidney disease?
Cistanche is a traditional Chinese herbal medicine used for centuries to treat various health conditions, including kidney disease. It is derived from the dried stems of Cistanche deserticola, a plant native to the deserts of China and Mongolia. The main active components of cistanche are phenylethanoid glycosides, echinacoside, and acteoside, which have been found to have beneficial effects on kidney health.

Kidney disease, also known as renal disease, refers to a condition in which the kidneys are not functioning properly. This can result in a buildup of waste products and toxins in the body, leading to various symptoms and complications. Cistanche may help treat kidney disease ase through several mechanisms.
Firstly, cistanche has been found to have diuretic properties, meaning it can increase urine production and help eliminate waste products from the body. This can help relieve the burden on the kidneys and prevent the buildup of toxins. By promoting diuresis, cistanche may also help Reduce high blood pressure, a common complication of kidney disease.

Moreover, cistanche has been shown to have antioxidant effects. Oxidative stress, caused by an imbalance between the production of free radicals and the body's antioxidant defenses, plays a key role in the progression of kidney disease. ies help neutralize free radicals and reduce Oxidative stress, thereby protecting the kidneys from damage. The phenylethanoid glycosides found in cistanche have been particularly effective in scavenging free radicals and inhibiting lipid peroxidation.
Additionally, cistanche has been found to have anti-inflammatory effects. Inflammation is another key factor in the development and progression of kidney disease. Cistanche's anti-inflammatory properties help reduce the production of pro-inflammatory cytokines and inhibit the activation of inflammation mandatory pathways, thus alleviating inflammation in the kidneys.
Furthermore, cistanche has been shown to have immunomodulatory effects. In kidney disease, the immune system can be dysregulated, leading to excessive inflammation and tissue damage. Cistanche helps regulate the immune response by modulating the production and activity of immune cells, such as T cells and macrophages. This immune regulation helps reduce inflammation and prevent further damage to the kidneys.
Moreover, cistanche has been found to improve renal function by promoting the regeneration of renal tubes with cells. Renal tubular epithelial cells play a crucial role in the filtration and reabsorption of waste products and electrolytes. In kidney disease, these cells can be damaged, leading to damaged renal function. Cistanche's ability to promote the regeneration of these cells helps restore proper renal function and improve overall kidney health.

In addition to these direct effects on the kidneys, cistanche has been found to have beneficial effects on other organs and systems in the body. This holistic approach to health is particularly important in kidney disease, as the condition often affects multiple organs and systems. che has been shown to have protective effects on the liver, heart, and blood vessels, which are commonly affected by kidney disease. By promoting the health of these organs, cistanche helps improve overall kidney function and prevent further complications.
In conclusion, cistanche is a traditional Chinese herbal medicine used for centuries to treat kidney disease. Its active components have diuretic, antioxidant, anti-inflammatory, immunomodulatory, and regenerative effects, which help improve renal function and protect the kidneys from further damage. , cistanche has beneficial effects on other organs and systems, making it a holistic approach to treating kidney disease.






